Abstract
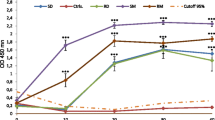
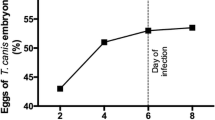
In the present study the behavior and pathogenicity of second-stage larvae ofToxocara canis were examined in different mouse strains with special emphasis on the major histocompatibility complex (MHC). Mice of the inbred strains BALB, C3H, C57BL, and DBA and the outbred strain NMRI were infected orally with 1000 second-stage larvae ofT. canis. The clinical behavior of the animals: the numbers of larvae detected in the liver, lungs, brain and musculature; the hematological and serological parameters; and histological sections were examined. In mice of the BALB strain, no death occurred during the entire period of the investigation and the pattern of body-weight development of infected and uninfected animals was almost identical. The highest larval counts in the brain of all strains were found in BALB mice. The percentage of eosinophils in the blood of BALB mice increased after the 8th week postinfection, whereas it decreased in the other strains. Histological and pathophysiological changes developed to a lesser extent in this strain than in the other strains. In mice of the strains C3H, C57BL, DBA, and NMRI, deaths occurred from the 4th week postinfection onward. The infected animals lost weight in comparison with the uninfected controls; the numbers of larvae found in the brains of infected mice of the above-mentioned strains were lower than those detected in the BALB strain. There is no evidence that mechanical damage caused by migrating larvae in the brain tissue is mainly responsible for symptoms of central nervous toxocariasis. Likewise, the assumption that the MHC is involved in the allergicinflammatory response in the brain could not be proven: infected mice of the BALB and DBA strains reacted completely differently, although both are equipped with the same MHC haplotype.
Similar content being viewed by others
References
Abraham D, Grieve RB (1990) Genetic control of murine responses to larvalDirofilaria immitis J Parasitol 76: 523–528
Baermann G (1917) Eine einfache Methode yur Auffindung von Ankylostomum-(Nematoden)/Larven in Erdproben. Tijdschr Diergeneesk 57: 131–137
Dunsmore JD, Thompson RCA, Bates IA (1983) The accumulation ofToxocara canis larvae in the brains of mice Int J Parasitol 13: 517–521
Else K, Wakelin, D (1988) The effects of H-2 and non-H-2 genes on the expulsion of the nematodeTrichuris muris from inbred and congenic mice. Parasitology 96: 543–550
Else K, Wakelin D (1990) Genetically-determined influences on the ability of poor responder mice to respond to immunization againstTrichuris muris. Parasitology 100: 479–489
Horn G (1983) Verhalten und Pathogenität der Larven vonToxocara canis Werner 1782 (Anisakidae) bei ganzkörperbestrahlten Mäusen. Veterinary medicine dissertation, University of Hannover
Kayes SG, Oaks JA (1978) Development of the granulomatous response in murine toxocariasis. Initial events. Am J Pathol 93: 277–294
Kennedy MW (1990) Resistance to parasitic nematodes—how is the MHC involved. Parasitol Today 6: 374–375
Kennedy MW, Gordon AMS, Tomlinson LA, Fureshi F (1987) Genetic (major histocompatibility complex? control of antibody repertoire to the second stage antigens ofAscaris. Parasite Immunol 9: 269
Kennedy MW, McIntosh AE, Blair AJ, McLaughlin D (1990a) MHC (RT1) restriction of the antibody repertoire to infection with the nematodeNippostrongylus brasiliensis in the rat. Immunology 71: 317–322
Kennedy MW, Tomlinson LA, Fraser EM, Christie JF (1990b) The specificity of the antibody response to internal antigens ofAscaris: heterogeneity in infected humans, and MHC (H-2) control of the repertoire in mice. Clin Exp Immunol 80: 219–224
Kennedy MW, Fraser EM, Christie JF (1991) MHC class II (I-A) region control of the IgE antibody repertoire to the ABA-I allergen of the nematodeAscaris Immunology 72: 577–579
Levine HS, Silverman PH (1969) Cultivation ofAscaris suum larvae in supplemented and unsupplemented chemically defined media. J Parasitol 55: 17–21
Lohmann K (1989) Verhalten und Pathogenität der Larven vonToxocara canis Verner 1782 (Anisakidae) in der Maus. Zentralbl Veterinarmed [B] 36: 609–618
Mitruka BM, Rawnsley HM (1981) Clinical biochemical and hematological reference values in normal experimental animals and normal humans, 2nd edn. Masson, New York, p 59
Peppersack T (1981) Zur Pathogenität der Larven vonToxocara canis Werner 1782 (Anisakidae) bei der Maus. Veterinary medicine dissertation, University of Hannover
Sonnen P (1981) Zur Wirkung verschiedener Benzimidazolcarbamate auf somatische Larven vonAncylostoma caninum Ercolani 1859 (Ancylostomidae) undToxocara canis Werner 1782 (Anisakidae). 1. Untersuchungen an der weißen Maus. Zentralbl Veterinarmed [B] 28: 226–240
Sugane K, Oshima T (1984) Interrelationship of eosinophilia and IgE antibody production to larval ES antigen inToxocara canis infected mice. Parasite Immunol 6: 409–420
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Epe, C., Sabel, T., Schnieder, T. et al. The behavior and pathogenicity ofToxacara canis larvae in mice of different strains. Parasitol Res 80, 691–695 (1994). https://doi.org/10.1007/BF00932955
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00932955