Abstract
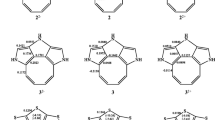
A thermodynamic criterion for aromatic and conjugative interactions is proposed. Enthalpies of stepwise hydrogenation of, for example, three double bonds are compensated for strain energy changes during hydrogenation. Strain energies are calculated by molecular mechanics. If the compensated values show a monotonic increase from bond 3 to bond 1, the molecule is conjugatively stabilized. If the initial rise is sharp followed by a constant ΔH h for bonds 2 and 1 and the molecule is cyclic, stabilization is aromatic. If the compensated ΔH h decreases, the interaction is destabilizing. By this set of criteria, biquinacene is unstabilized, triquinacene is homoaromatically stabilized, hexaquinacene is homoconjugatively stabilized, and cis,cis,cis-1,4,7-cyclononatriene is homoaromatically destabilized. New experimental data are presented for the biquinacenes (bicycloocta-1,7-diene and its hydrogenation products) and the hexaquinacenes.
Similar content being viewed by others
References and notes
McEwan, A. B.; Schleyer, P. v-R.J. Org. Chem. 1986,51, 4357.
Garratt, P. J.Aromaticity; John Wiley: New York, 1986.
Scott L. T.; Cooney, M. J.; Rogers, D. W.; Dejroongruang, K.J. Am. Chem. Soc. 1988,110, 7244.
Liebman, J. F., Paquette, L. A., Peterson, J. B., Rogers, D. W.J. Am. Chem. Soc. 1986,108, 8267.
Miller, M. A.; Schulman J. M.; Disch, R. L.J. Am. Chem. Soc. 1988,110, 7681.
Dewar, M. S.; Holder, A. J.J. Am. Chem. Soc. 1989,111, 5384.
Roth, W. R.; Bang, W. B.; Goebel, P.; Sass, R. L.; Turner, R. B.; Yu, A. P.J. Am. Chem. Soc. 1964,86, 3178.
Paquette, L. A.; Galatsis, P.J. Org. Chem. 1989,54, 5039.
Pedley, J. B.; Naylor, R. D.; Kirby, S. P.Thermochemical Data of Organic Compounds; Chapman and Hall: New York, 1986.
Allinger, N. L.; Dodziuk, H.; Rogers, D. W.; Naik, S. N.Tetrahedron 1982,38, 1593.
Conant, J. B.; Kistiakowsky, G. B.Chem. Rev. 1937,20, 181.
Lindner, H. J.Tetrahedron 1981,37, 535.
Schulman, J. M.; Disch, R. L.; Sabio, M. J.J. Am. Chem. Soc. 1984,106, 7696.
Kao, J.; Allinger, N. L.J. Am. Chem. Soc. 1977,99, 975,'85 modification: QCPE 318 MMP2(85); Quantum Chemistry Program Exchange; Indiana University, Department of Chemistry; Bloomington, IN.
Burkert U.; Allinger, N. L.Molecular Mechanics, ACS Publ. No. 177; American Chemical Society: Washington, DC, 1982.
Anet, F. A. L.; Yivari, I.J. Am. Chem. Soc. 1977,99, 6496.
Osawa, E.J. Am. Chem. Soc. 1979,101 5523.
Loggins, S. A. M.S. Thesis, Long Island University, 1989, Brooklyn, NY.
By subtracting one or two times ΔH h, (hexene) from the ΔH h of the diene or triene. (An empirical correction of −0.7 kcal mol−1 per double bond was made to correct data obtained in glacial acetic acid to gas-phase values.)
Turner, R. B.; Mallon, B. J.; Tichy, M., von E. Doering, W. Roth, W. R.; Schroeder, G.J. Am. Chem. Soc. 1973,95, 8605.
Rogers, D. W.; Crooks, E. L.J. Chem. Thermodyn. 1983,15, 1087.
Rogers D. W.; Dejroongruang, K.J. Chem. Thermodyn. 1988,20, 675.
Rogers, D. W.; Dagdagan, O. A.; Allinger, N. L.J. Am. Chem. Soc. 1979,101, 671.
Rogers, D. W.J. Phys. Chem. 1979,83, 2430.
Roth, W. R.; Lennartz, H.-W.Chem. Ber. 1980,113, 1806.
Supelco 60-m wide-bore capillary SE 30.
Chickos, J. Personal communication (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rogers, D.W., Loggins, S.A., Samuel, S.D. et al. Homoaromaticity and homoconjugation in the quinacenes: Biquinacene, triquinacene, and hexaquinacene. Struct Chem 1, 481–489 (1990). https://doi.org/10.1007/BF00671235
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00671235