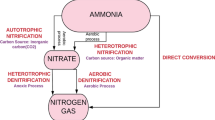
Abstract
This paper describes both qualitative and quantitative aspects of simultaneous autotrophic nitrification and heterotrophic denitrification by, respectively, the nitrifierNitrisomonas europaea and either of the denitrifiersPseudomonas denitrificans orParacoccus denitrificans co-immobilized in double-layer gel beads. The system is based on the establishment of well-defined oxic and anoxic zones within the cell supports and on physical separation of the nitrifying and denitrifying populations. Nitrification and denitrification rates were obtained from measured bulk concentrations and head-space analysis. The latter analyses showed that ammonia was primarily converted into molecular nitrogen. Nitrous oxide was not detected. High nitrogen removal rates (up to 5.1 mmol N m−3 gel s−1) were achieved in continuous reactors under aerobic conditions. The overall rate of nitrogen removal was controlled by the nitrifying step. The approach followed is, in principle, also suitable to the coupling of other oxidative and reductive bioprocesses having complementary metabolic routes. Two-stage bioconversion processes can be thus conducted as if single-staged, which results in more compact reactor systems.
Similar content being viewed by others
References
Beer D de, Van den Heuvel JC, Ottengraaf SPP (1993) Microelectrode measurements of the activity distribution in nitrifying bacterial aggregates. Appl Environ Microbiol 59:573–579
Beunik H, Rehm HJ (1988) Synchronous anaerobic and aerobic degradation of DDT by an immobilized mixed culture system. Appl Microbiol Biotechnol 29:72–80
Beunik H, Rehm HJ (1990) Coupled reductive and oxidative degradation of 4-chloro-2-nitrophenol by a co-immobilized mixed culture system. Appl Microbiol Biotechnol 34:108–115
EPA (US Environmental Protection Agency) (1975) Process design manual for nitrogen control. USEPA, Washington D.C., USA.
Field JA, Stams AJ, Kato M, Schraa G (1995) Enhanced biodegradation of aromatic pollutants in cocultures of anaerobic and aerobic bacterial consortia. Antonie van Leeuwenhoek 67:47–77
Gardin H, Pauss A (1994) Co-immobilization of aerobic and anaerobic bacterial communities in a mixed κ-carrageenan/gelatin gel for the degradation of 2,4,6-trichlorophenol. In: Lacroïx C, Vuillemard J-C (eds) Proceedings of International Workshop on Bioencapsulation. IV. Quebec, Canada. pp 93–97
Gerritse J, Gottschal JC (1992) Mineralization of the herbicide 2,3,6-trichlorobenzoic acid by a coculture of aerobic and anaerobic bacteria. FEMS Microbiol Ecol 10:89–98
Ginkel CG van, Tramper J, Luyben KChAM, Klapwijk A (1983) Characterization ofNitrosomonas europaea immobilized in calcium alginate. Enzyme Microb Technol 5:297–303
Gupta SK, Raja SM, Gupta AB (1994) Simultaneous nitrification-denitrification in a rotating biological cactor. Environ Technol 15:145–153
Hooijmans CM, Geraats SGM, Van Niel EWJ, Robertson LA (1990a) Determination of growth and coupled nitrification/ denitrification by immobilizedThiosphaeara pantotropha using measurement and modeling of oxygen profiles. Biotechnol Bioeng 36:931–939
Hooijmans CM, Ras C, Luyben KChAM (1990b) Determination of oxygen profiles in biocatalyst particles by means of a combined polarographix oxygen microsensor: measurement and modeling of oxygen profiles. Enzyme Microb Technol 12:178–183
Hunik JH, Bos CG, Van Den Hoogen P, De Gooijer CD, Tramper J (1994) Co-immobilizedNitrosomonas europaea andNitrobacter agilis cells: validation of a dynamic model for simultaneous substrate conversion and growth in κ-carrageenan gel beads. Biotechnol Bioeng 43:1153–1163
Kokufuta E, Shimoshashi M, Nakamura I (1987) Continuous column denitrification using polyelectrolyte complex-entrappedParacoccus denitrificans cells. J Ferment Technol 65:359–361
Kurosawa H, Tanaka H (1990) Advances in immobilized cell culture: development of a co-immobilized mixed culture system of aerobic and anaerobic micro-organisms. Process Biochem Int 25:189–196
Nilsson I, Ohlson S (1982) Columnar denitrification of water by immobilized cells. Appl Microbiol Biotechnol 14:86–90
Nilsson I, Ohlson S, Haggstrom L, Molin N, Mosbach K (1980). Denitrification of water using immobilizedPseudomonas denitrificans cells. Eur J Appl Microbiol Biotechnol 10:261–274
Robertson LA, Kuenen JG (1990) Combined heterotrophic nitrification and aerobic denitrification inThiosphaera pantotropha and other bacteria. Antonie van Leeuwenhoek 57:139–152
Robertson LA, Corneline R, De Vos P, Hadioetomo R, Kuenen JG (1989) Aerobic denitrification in various heterotrophic nitrifiers. Antonie van Leeuwenhoek 56:289–299
Santos VAPM dos, Tramper J, Wijffels RH, Tramper J (1992) Integrated nitrification and denitrification with immobilized micro-organisms. In: Melo LF et al (eds) Biofilms-Science and technology. Kluwer Academic Publishers, Dordrecht, The Netherlands, pp 449–453
Santos VAPM dos, Tramper J, Wijffels RH (1993) Simultaneous nitrification and denitrification using immobilized micro-organisms. Biomater Artif Cells Immobilization Biotech 21:317–322
Santos VAPM dos, Marchal JL, Tramper J, Wijffels RH (1996) Modeling and evaluation of an integrated nitrogen removal with co-immobilized micro-organisms in double-layer gel beads. Biotechnol Prog (in press)
Schlegel HG (1986)General Microbiology. Cambridge University Press, Cambridge
Tijhuis L (1994) Biofilm airlift suspension reactor: biofilm formation, detachement and heterogeneity. PhD thesis, TU Delft, The Netherlands
Tramper J (1984) Nitrification and denitrification by immobilized bacteria. In: Proceedings of the 3rd European Congress on Biotechnology, VCH Verlag, Weinheim, vol IV, pp 363–368
Tramper J (1987) Nitrification and denitrification by immobilized viable cells. In: Laskin AI et al. (eds) Enzyme Engineering vol 8. New York Academy of Sciences, New York, pp 362–366
Van Niel EWJ (1991) Nitrification by heterotrophic denitrifiers and its relation to autotrophic nitrification. PhD Thesis, TU Delft, The Netherlands
Varseveld HG van (1979) Environmental factors on the efficiency of energy conservation inParacoccus denitrificans. PhD Thesis, Free University of Amsterdam, The Netherlands
Vishniac W, Santer M (1957) The thiobacilli. Bacteriol Rev 21:195–213
Wijffels RH, Tramper J (1989) Performance of growingNitrosomonas europaea cells immobilized in κ-carrageenan, Appl Microbiol Biotechnol 32:108–112
Wijffels RH, Schukking A, Tramper J (1990) Characterization of a denitrifying bacterium immobilized in κ-carrageenan, Appl Microbiol Biotechnol 34:399–403
Wijffels RH, de Gooijer CD, Kortekaas S, Tramper J (1991) Growth and substrate consumption ofNitrobacter agilis cells immobilized in κ-carrageenan. Part 2. Model evaluation. Biotechnol Bioeng. 38:232–240
Wijffels RH, de Gooijer CD, Schepers AW, Beuling EE, Mallée LF, Tramper J (1995) Dynamic modelling of immobilizedNitrosomonas europaea: implementation of diffusion limitation over expanding micro-colonies. Enzyme Microb Technol 17:462–471
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
dos Santos, V.A.P.M., Bruijnse, M., Tramper, J. et al. The magic-bead concept: an integrated approach to nitrogen removal with co-immobilized micro-organisms. Appl Microbiol Biotechnol 45, 447–453 (1996). https://doi.org/10.1007/BF00578454
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00578454