Conclusions
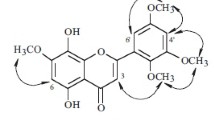
The following flavonoids have been isolated from propolis and identified: acacetin (I), 7,4′-dimethoxyflavone (II), kaempferide (III), rhamnocitrin (IV), 5,7-dihydroxy-3,4′-dihydroxyflavone (V), 3,5-dihydroxy-7,4′-dimethoxyflavone (VI), (−)-pinostrobin (VII), and (−)-5-hydroxy-7,4′-dimethoxyflavanone (VIII).
Similar content being viewed by others
References
P. Lavie, Ann. Inst. Natl. Rech. Agron., Sér Cbis 3 103, 201, 1960.
P. Lavie, Compt. rend.,248, 455, 1959.
V. P. Kivalkina, “Propolis: its antimicrobial and medicinal properties,” Dissertation, Kazan, 1964.
Austrian patent nos. 167862 and 172063; C. A.,46, 9810, 1952.
L. A. Lindenfelser, Am. Bee J.,107, 90, 130, 1967.
V. R. Villanueva, D. Bogdanovsky, M. Barbier, M. Gonnet, and P. Lavie, Ann. Inst. Pasteur, 106, 292, 1964.
H. Erdtman, L. Novotný, and M. Romanuk, Tetrah., Suppl., no. 8, 71, 1966.
E. L. Ghisalberti, P. R. Jefferies, and C. I. Stacey, Austral. J. Chem.,20, 1049, 1967.
F. Bohlmann and C. Zdero, Tetrah. Let., 3239, 1967.
R. G. Wilson, J. H. Bowie, and D. H. Williams, Tetrah.,24, 1407, 1968.
L. Jurdi, in: T. A. Geissman, (editor) The Chemistry of Flavonoid Compounds, Oxford, 107–155, 1962.
H. M. Lynch, T. M. O'Toole, and T. S. Wheeler, J. Chem. Soc., 2063, 1952.
F. M. Dean, Naturally Occurring Oxygen Ring Compounds, London, 357, 1963.
Y. Asahina and M. Inubuse, Ber.,61, 1514, 1928.
W. Gaffield and A. C. Waiss, Chem. Communs., 29, 1968.
Y. Asahina, J. Shinoda, and M. Inubuse, J. Pharm. Soc., Japan, 47, 133, 1927.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 5, No. 6, pp. 476–482, 1969
Rights and permissions
About this article
Cite this article
Popravko, S.A., Gurevich, A.I. & Kolosov, M.N. Flavonoid components of propolis. Chem Nat Compd 5, 397–401 (1969). https://doi.org/10.1007/BF00568574
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00568574