Summary
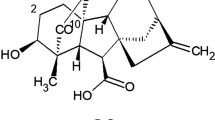
1. By means of preparative paper chromatography, an isomer of gibberelin A3, the 1,3-lactone of 2β, 3α, 7α-trihydroxy-1β-methyl-8-methylene-Δ4-gibbene-1α, 10β-dicarboxylic acid(II) has been isolated from the culture liquid ofFusarium moniliforme grown in a medium containing sunflower oil and ammonium nitrate.
2. The formation of this product under fermentation conditions takes place hydrolytically by a nonenzymatic mechanism.
3. A study of the physiological activity of the 1,3-lactone (II) has shown that the change in the position of the lactone cycle in ring A leads to a considerable decrease of the growth activity of this substance with respect to dwarf pea and lettuce, while it is retained with respect to cucumbers (as compared with the activity of gibberellin A3).
Similar content being viewed by others
References
B. E. Cross, J. F. Grove, and A. Morrison, J. Chem. Soc., 2498, 1961.
T. P. C. Mulholland, Brit. Pat. no. 919 205, 20 Feb., 1963.
J. Kuhr, Folia microbiol., 7, 358, 1962.
G. W. Elson, D. F. Jones, J. MacMillan, and P. J. Suter, Phytochemistry 3, 93, 1964.
E. P. Serebryakov, et al., KhPS [Chemistry of Natural Compounds], 55, 1966.
G. S. Muromtsev, et al., Fiziologiya rastenii, 11, 506, 1964.
G. S. Muromtsev, Uspekhi sovremennoi biologii, 57, 477, 1964.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 2, No. 2, pp. 114–120, 1966
Rights and permissions
About this article
Cite this article
Muromtsev, G.S., Agnistikova, V.N., Lupova, L.M. et al. Gibberellins and substances related to them. Chem Nat Compd 2, 89–93 (1966). https://doi.org/10.1007/BF00567542
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00567542