Summary
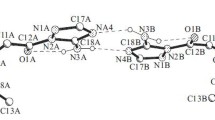
The configurations of four of the six asymmetric centers of stachysolone have been established. This diterpenoid belongs to compounds with the trans (5α, 10β) linkage of the A/B rings.
Similar content being viewed by others
Literature cited
D. P. Popa, T. M. Orgiyan, Z. Samek, and L. Dolejs, Khim. Prirodn. Soedin., 295 (1972).
R. Misra, R. C. Pandey, and Sukh Dev, Tetrahedron Lett., 2681 (1968).
R. Misra and Sukh Dev, Tetrahedron Lett., 2685 (1968).
T. J. King, S. Rodrigo, and S. C. Wallwork, Chem. Comm., D. 683 (1969).
T. J. King and S. Rodrigo, Chem. Comm., 575 (1967).
G. Snatzke and H. W. Fehlhaber, Tetrahedron,20, 1243 (1964).
G. Snatzke, “α,β- and β-γ-unsaturated ketones,” in: Optical Rotatory Dispersion and Circular Dichroism in Organic Chemistry, P. Crabbe (editor), Holden-Day, San Francisco (1965).
C. Djerassi, R. Riniker, and B. Riniker, J. Amer. Chem. Soc.,78, 6378 (1956).
C. Djerassi and D. Marshall, J. Amer. Chem. Soc.,80, 3986 (1958).
L. H. Zalkow, F. X. Markley, and C. Djerassi, J. Amer. Chem. Soc.,81, 2914 (1959).
A. E. Lippman, E. W. Foltz, and C. Djerassi, J. Amer. Chem. Soc.,77, 4364 (1955).
C. Djerassi, W. Closson, and A. E. Lippman, J. Amer. Chem. Soc.,78, 3163 (1956).
C. Djerassi, R. Riniker, and B. Riniker, J. Amer. Chem. Soc.,78, 6362 (1956).
R. G. Curtis, I. Heilbron, E. R. H. Jones, and G. F. Woods, J. Chem. Soc., 457 (1953).
Additional information
Institute of Chemistry, Academy of Sciences of the Moldavian SSR. Translated from Khimiya Prirodnykh Soedinenii, No. 6, pp. 735–738, November–December, 1972.
Rights and permissions
About this article
Cite this article
Popa, D.P., Orgiyan, T.M. The stereochemistry of stachysolone. Chem Nat Compd 8, 717–719 (1972). https://doi.org/10.1007/BF00564591
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00564591