Abstract
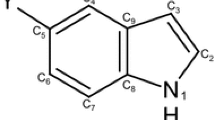
The nitration and bromination of 6-methylindolo[2,3-b]quinoxaline take place in the 9 position. The reactivity indexes were calculated within the framework of the Hückel MO and CNDO (complete neglect of differential overlap) methods. Only the π-charge distribution is in satisfactory agreement with the experimental data on electrophilic substitution in the investigated compound.
Similar content being viewed by others
Literature cited
A. V. Ivashchenko and V. M. Dziomko, Usp. Khim., 46, 244 (1977).
A. R. Katritzky (editor), Physical Methods in the Chemistry of Heterocyclic Compounds, Academic Press (1963).
W. Borsche and N. Jacobs, Chem. Ber., 47, 354 (1914).
H. O. Calvery, C. R. Noller, and R. Adams, J. Am. Chem. Soc., 47, 3059 (1925).
R. Silverstein, G. Bassler, and T. Morrill, Spectroscopic Identification of Organic Compounds, Wiley (1974).
K. Nakanishi, Infrared Spectra and Structure of Organic Compounds [Russian translation], Mir, Moscow (1965), p. 61.
R. L. Ellis, G. Kuehnlenz, and H. H. Jaffe, Theor. Chim. Acta, 26, 131 (1972).
V. M. Ryaboi and I. N. Fundlyer, Algoritmy Programmy, No. 3, P 001396, 48 (1975).
M. M. Kaganskii, G. G. Dvoryantseva, I. V. Sokolova, and V. I. Danilov, Khim. Geterotsikl. Soedin., No. 1, 118 (1975).
E. M. Evleth, Theor. Chim. Acta, 16, 22 (1970).
G. M. Badger and P. Nelson, J. Chem. Soc., No. 10, 3930 (1962).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 840–842, June, 1984.
Rights and permissions
About this article
Cite this article
Grachev, V.T., Ivashchenko, S.P., Ivanova, O.A. et al. Electrophilic substitution and electronic structure of indolo[2,3-b]quinoxaline. Chem Heterocycl Compd 20, 687–689 (1984). https://doi.org/10.1007/BF00508686
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00508686