Summary
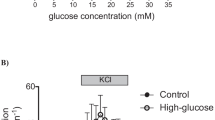
HIT is a hamster-derived beta-cell line which in contrast to normal beta cells that only express the high Km GLUT-2 glucose transporter, also expresses the low Km glucose transporter GLUT 1. In HIT cells the abnormal glucose transport mechanism is associated with a marked shift to the left of the glucose-induced insulin release dose-response curve. We have used this cell model to investigate whether changes in glucose transport affect the glucose-induced insulin release. HIT cells were first incubated with a concentration of cytochalasin B (0.4 μmol/l) that selectively inhibits the GLUT-1 but not the GLUT-2 transporter. The consequences of blocking glucose phosphorylation and insulin release were studied. Exposure to 0.4 μmol/l cytochalasin B for 1 h caused a selective loss of the low Km transport: the calculated Vmax of GLUT 1 was reduced from 1726±98 to 184±14 pmol · mg protein−1 5 min−1 (mean±SEM, n=6, p<0.005), while no major difference in the high Km (GLUT-2) transport was observed. In cytochalasin B exposed HIT cells the glucose phosphorylating activity (due to hexokinase and glucokinase) was unaffected. In these cells, however, the dose-response curve of glucose-induced insulin release was significantly shifted to the right: the 50% of maximal response (increment over baseline) was reached at an average glucose concentration of 2.9±0.2 mmol/l (vs 0.6±0.01 mmol/l in control HIT cells mean±SE, n=5, p<0.05) and the maximal effect was reached at 11.0 mmol/l glucose (vs 2.8 mmol/l in control HIT cells p<0.005). These results are consistent with the hypothesis that the affinity of the glucose transport system may contribute to determination of the glucose threshold concentration that triggers insulin secretion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Matschinsky FM (1990) Glucokinase as glucose sensor and metabolic signal generator in pancreatic β-cells and hepatocytes. Diabetes 39: 647–652
Purrello F, Buscema M, Rabuazzo AM et al. (1993) Glucose modulates the glucose transporter affinity, the glucokinase activity, and the secretory response in rat pancreatic islets. Diabetes 42: 199–205
Purrello F, Buscema M, Vetri M et al. (1991) Glucose regulates both glucose transport and the glucose transporter gene expression in a hamster-derived pancreatic Beta-cell line (HIT). Diabetologia 34: 366–369
Shimizu T, Knowles BB, Matschinsky FM (1988) Control of glucose phosphorylation and glucose usage in clonal insulinoma cells. Diabetes 37: 563–568
Johnson JH, Newgard CB, Milburn LJ, Lodish HF, Thorens B (1990) The high Km glucose transporter of islets of Langerhans is functionally similar to the low affinity transporter of liver and has identical primary sequence. J Biol Chem 265: 6548–6551
Clark SA, Burnham BL, Chick WL (1990) Modulation of glucose-induced insulin secretion from a rat clonal β-cell line. Endocrinology 127: 2779–2788
Yasunami Y, Funakoshi A, Ono J, Miyazaki K, Jimi A, Konomi K (1987) In vitro study of cultured human insulinoma cells: evidence of abnormal sensitivity to glucose. J Clin Endocrinol Metab 65: 110–115
Brant AM, McCoid S, Thomas HM et al. (1992) Analysis of the glucose transporter content of islet cell lines — implications for glucose-stimulated insulin release. Cell Signal 4: 641–650
Seino Y, Yamamoto T, Inoue K et al. (1993) Abnormal facilitative glucose transporter gene expression in human islet cell tumors. J Clin Endocrinol Metab 76: 75–78
Epstein PN, Boschero AC, Atwater I, Xingang C, Overbeek PA (1992) Expression of yeast hexokinase in pancreatic beta cells of transgenic mice reduces blood glucose, enhances insulin secretion, and decreases diabetes. Proc Natl Acad Sci USA 89: 12038–12042
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rabuazzo, A.M., Buscema, M., Vinci, C. et al. Inhibition of the high-affinity glucose transporter GLUT 1 affects the sensitivity to glucose in a hamster-derived pancreatic beta cell line (HIT). Diabetologia 36, 1204–1207 (1993). https://doi.org/10.1007/BF00401067
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00401067