Summary
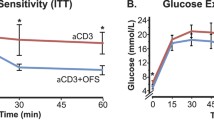
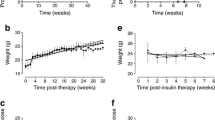
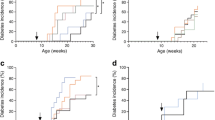
Oral administration of the synthetic immunomodulating drug quinoline-3-carboxamide (Linomide) in the drinking water to 5-week-old female non-obese diabetic (NOD) mice resulted in complete protection from insulitis and maintenance of normal glucose tolerance for over 40 weeks (impaired glucose tolerance: treated n=2 of 18; control n=17 of 18, p<0.0001). Delayed administration of the drug at 16 weeks resulted in slowing of the progression to diabetes when assessed at 42 weeks (treated with diabetes n=7 of 25; control with diabetes 25 of 43, p<0.0234). No gross changes of immune system cell phenotype or function were observed in the Linomide-treated group. Adoptive transfer of spleen and lymph node cells from treated female NOD mice into sub-lethally irradiated male recipients failed to transfer diabetes, whereas a similar transfer of cells obtained from untreated age-matched controls resulted in diabetes in all secondary recipients (diabetes in control group n=12 of 13; in Linomide group n=0 of 11, p<0.0001). Linomide pretreatment of the secondary recipients also inhibited the transfer of diabetes (diabetes in pretreated group n=2 of 9, control group n=12 of 13, p<0.015), as did adoptive co-transfer of cell mixtures obtained from treated female NOD mice, free of diabetes, and from diabetic NOD female mice (diabetes in Linomide group n=4 of 9; in control group 7 of 7, p<0.0337). Our data indicate that Linomide-treated NOD mice generate immune cells with the capacity to downregulate responses to beta-cell antigens, apparently through immunoregulation rather then antigen non-specific immunosuppression. Based on our findings and considering the lack of severe side effects of orally administered Linomide in man, this new compound should be considered as a potential drug for treatment of insulin-dependent diabetes mellitus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- IDDM:
-
Insulin-dependent diabetes mellitus
- NK:
-
natural killer
- IL:
-
interleukin
- NOD:
-
non-obese diabetic
- IPGTT:
-
intraperitoneal glucose tolerance test
- FITC:
-
fluorescein-5-isothiocyanate
- DTAF:
-
5(4,6 dichlorotriazinyl) aminofluorescein
- FACS:
-
fluorescein activated cell sorting
- PHA:
-
phythaemagglutinin
- Con A:
-
concanavalin A
- SEA:
-
staphylococcal extract A
- SEB:
-
staphylococcal extract B
- SRBC:
-
sheep erythrocytes
- HBSS:
-
Hanks buffered salt solution
- LPS:
-
lipopolysaccharide
- MLR:
-
mixed lymphocyte reaction
- DTH:
-
delayed-type, hypersensitivity
- BCG:
-
Bacillus Calmette Guerrin
- APC:
-
antigen presenting cell
- TNF-α:
-
tumor necrosis factor-α
References
Kalland T, Alm G, Standhalske T (1985) Augmentation of mouse natural killer cell activity by LS 2616, a new immunomodulator. J Immunol 134:3956–3961
Kalland T (1990) Regulation of natural killer progenitors. Studies with a novel immunomodulator with distinct effects at the precursor level. J Immunol 144:4472–4476
Stalhandske T, Kalland T (1986) Effects of the novel immunomodulator LS 2616 on the delayed-type hypersensitivity reaction to Bordetella pertussis in the rat. Immunopharmacology 11:87–92
Larson EL, Joki A, Standhalske T (1987) Mechanisms of action of the new immunomodulator LS2616 on T-cell responses. Int J Immunopharmacol 9:425–431
Tarkowski A, Gunnarsson K, Stalhandske T (1986) Effects of LS-2616 administration upon the autoimmune disease of (NZB×NZW) F1 hybrid mice. Immunology 59:589–594
Tarkowski A, Gunnarsson K, Nilsson LA, Lindholm L, Stalhandske T (1986) Successful treatment of autoimmunity in MRL/1 mice with LS-2616, a new immunomodulator. Arthr Rheum 29:1405–1409
Bjork J, Klienau S (1989) Paradoxical effects of LS-2616 (Linomide) treatment in the type II collagen arthritis model in mice. Agents and Actions 27:319–321
Karussis DM, Lehmann D, Slavin S et al. (1993) Successful treatment of chronic-relapsing experimental autoimmune encephalomylitis by the synthetic immunomodulator linomide (quinoline-3-carboxamide). Proc Natl Acad Sci USA 90:6400–6404
Charlton B, Bacelj A, Mandel TE (1988) Administration of silica particles or anti-Lyt2 antibody prevents beta-cell destruction in NOD mice given cyclophosphamide. Diabetes 37:930–935
Slavin S, Strober S, Fuks Z, Kaplan HS (1977) Induction of specific tissue transplantation tolerance using fractionated total lymphoid irradiation in adult mice: long-term survival of allogeneic bone marrow and skin grafts. J Exp Med 146:34–48
Weiss L, Kedar E, Weigensberg M, Slavin S (1982) Natural cell-mediated cytotoxicity in mice treated with total lymphoid irradiation (TL1). Cell Immunol 70:188–195
Slavin S, Strober S (1979) Induction of allograft tolerance after total lymphoid irradiation (TLI): development of suppressor cells of the mixed leukocyte reaction (MLR). J Immunol 123:942–946
Tarcic N, David CS, Naor D (1989) Auto-delayed-type hypersensitivity induced in immunodeficient mice with modified self antigens. Immunology 67:184–190
Yagi H, Matsumoto M, Suzuki S et al. (1991) Possible mechanism of the preventive effect of BCG against diabetes mellitus in NOD mouse. I. Generation of suppressor macrophages in spleen cells of BCG-vaccinated mice. Cell Immunol 138:130–141
Sadelain MW, Qin HY, Lauzon J, Singh B (1990) Prevention of type I diabetes in NOD mice by adjuvant immunotherapy. Diabetes 39:583–589
McInerney MF, Pek SB, Thomas DW (1991) Prevention of insulitis and diabetes onset by treatment with complete Freund's adjuvant in NOD mice. Diabetes 40:715–725
Ulaeto D, Lacy PE, Kipnis DM, Kanagawa O, Unanue ER (1992) A T-cell dormant state in the autoimmune process of nonobese diabetic mice treated with complete Freund's adjuvant. Proc Natl Acad Sci USA 89:3927–3931
Qin H-Y, Sadelain MWJ, Hitchon C, Lauzon J, Singh B (1993) Complete Freund's adjuvant-induced T cells prevent the development and adoptive transfer of diabetes in nonobese diabetic mice. J Immunol 150:2072–2080
Serreze DV, Gaskins HR, Leiter EH (1993) Defects in the differentiation and function of antigen presenting cells in NOD/It mice. J Immunol 150:2534–2543
Cirulli V, Halban PA, Rouiller D (1993) Tumor necrosis factor-α modifies adhesion properties of rat islet B cells. J Clin Invest 91:1868–1876
Mandrup-Poulsen T, Bendtzen K, Dinarello CA, Nerup J (1987) Human tumor necrosis factor potentiates human interleukin-1-mediated rat pancreatic Β-cell cytotoxicity. J Immunol 139:4077–4082
Campell IL, Iscalo A, Harrison LC (1988) IFN-γ and tumor necrosis factor-α: cytotoxicity to murine islets of Langerhans. J Immunol 141:2325–2329
Pukel C, Baquerizo H, Rabinovitch A (1988) Destruction of rat islet cell layers by cytokines: synergistic interactions of interferon-γ, tumor necrosis factor, lymphotoxin and interleukin-1. Diabetes 37:133–136
Jacob CO, Also S, Michie SA, McDevitt HO, Acha-Orbea H (1990) Prevention of diabetes in nonobese diabetic mice by tumor necrosis factor (TNF): similarities between TNF-alpha and interleukin 1. Proc Natl Acad Sci USA 87:968–972
Yamada K, Takane N, Otabe S, Inada C, Inoue M, Nonaka K (1993) Pancreatic Β-cell-selective production of tumor necrosis-alpha induced by interleukin-1. Diabetes 42:1026–1031
Gonzalo JA, Gonzalez- Garcia A, Kalland T, Hedlund G, Martinez-A C, Kroemer G (1993) Linomide, a novel immunomodulator that inhibits death in four different models of septic shock. Eur J Immunol 23:2372–2374
Bengtsson M, Simonsson B, Carlsson K et al. (1992) Stimulation of NK cell, T cell, and monocyte functions by the novel immunomodulator Linomide after autologous bone marrow transplantation. A pilot study in patients with acute myeloid leukemia. Transplantation 53:882–888
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gross, D.J., Sidi, H., Weiss, L. et al. Prevention of diabetes mellitus in non-obese diabetic mice by Linomide, a novel immunomodulating drug. Diabetologia 37, 1195–1201 (1994). https://doi.org/10.1007/BF00399792
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00399792