Summary
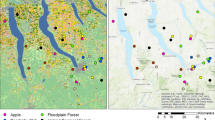
The caterpillars of Jalmenus evagoras are tended by ants as they feed upon Acacia trees. In the area of Brisbane, Australia, J. evagoras require ants of the Iridomyrmex anceps species group; predation and parasitism are so intense that larvae and pupae deprived of attendant ants cannot survive (Pierce 1983). We investigated the efficiency with which J. evagoras locate and exploit the “host ant” resource by sampling 737 quadrats in 30 sampling grids and six study sites containing appropriate host plants; ants were collected at baits located in the center of each quadrat. J. evagoras was found in all habitats where I. anceps cooccurred with host Acacia. Nine of the ten sampling grids which had three or more I. anceps/Acacia “host” quadrats also had colonies of J. evagoras present (or immediately adjacent), including sites as far as 35 km apart. Of 19 sampling grids on which “host” quadrats were rare (i.e., less than three quadrats), none had J. evagoras (P<0.001). Within sample grids, I. anceps was distributed indepedently from Acacia trees, suggesting that they are not dependent for their survival on either Acacia or on J. evagoras. Within montane pasture habitats, I. anceps and at least one other ground-dwelling Iridomyrmex species were distributed in mutually exclusive “ant mosaic” territories which were stable during a one month period. I. anceps did not colonize or tend pupae of J. evagoras experimentally placed in adjacent territories of a different, nontending species of Iridomyrmex, demonstrating the integrity of territory boundaries. Sampling of ants in Acacia trees revealed that, in the absence of J. evagoras, Iridomyrmex workers are not common above ground level, and that their numbers decline in larger trees (P=0.02). In I. anceps territories, eight of nine J. evagoras pupae placed in trees over 3.0 m tall were not found after 24 h whereas all ten controls placed in low trees were found and tended (P=0.00012). This may explain why J. evagoras tends to oviposit in trees less than 2.0 m tall. An alternative hypothesis, that smaller trees have higher content of total nitrogen, and are threfore more nutritious, was not supported. We conclude that the local distribution and host tree selection by J. evagoras is dependent upon the distribution, patchiness, and foraging behavior of the host ant, I. anceps, and its spatial overlap with a number of species of host Acacia.
Similar content being viewed by others
References
Atsatt PR (1981a) Ant-dependent foodplant selection by the mistletoe butterfly Ogyris amaryllis (Lycaenidae). Oecologia 48:60–63.
Atsatt PR (1981b) Lycaenid butterflies and ants: selection for enemy free space. Am. Nat. 118:638–654.
Common IFB, Waterhouse DF (1981) Butterflies of Australia. Angus and Robertson Publ., Sydney.
Elgar MA, Pierce NE (1988) Mating success and fecundity in an ant-tended lycaenid butterfly. In: Reproductive success: studies of selection and adaptation in contrasting breeding systems. Ed. by T.H. Clutton-Brock, University of Chicago Press, Chicago, IL (in press).
Jackson DA (1984) Ant distribution patterns in a Cameroonian cocoa plantation: investigation of the ant mosaic hypothesis. Oecologia 62:318–324.
Jeffries MJ, Lawton JH (1984) Enemy free space and the structure of ecological communities. Biol. J. Linn. Soc. 23:269–286.
Kitching RL (1983) Myrmecophilous organs of the larvae and pupae of the lycaenid butterfly, Jalmenus evagoras (Donovan). J. Nat. Hist. 17:471–481.
Leston D (1978) A neotropical and mosaic. Ann. Ent. Soc. Amer. 71:649–653.
Pierce NE (1983) The ecology and evolution of symbiosis between lycaenid butterflies and ants. Ph.D. Thesis, Harvard University, Cambridge, MA.
Pierce NE (1984) Amplified species diversity: A case study of an Australian lycaenid butterfly and its attendant ants. Symp. R. Entomol. Soc. Lond. 11:197–200.
Pierce NE (1985) Lycaenid butterflies and ants: selection for nitrogen fixing and other protein rich food plants. Am. Nat. 125:888–895.
Pierce NE, Elgar MA (1985) The influence of ants on host selection by Jalmenus evagoras, a myrmecophilous lycaenid butterfly. Behav. Ecol. and Sociobiol. 16:209–222.
Pierce NE, Easteal S (1986) The selective advantage of attendant ants for the larvae of a lycaenid butterfly, Glaucopsyche lygdamus. J. Anim. Ecol. 55:451–462.
Pierce NE, Kitching RL, Buckley RC, Taylor MFJ, Benbow KF (1987) The costs and benefits of cooperation between the Australian lycaenid butterfly, Jalmenus evagoras, and its attendant ants. Behav Ecol Sociobiol 21:237–248
Pierce NE, Mead PS (1981) Parasitoids as selective agents in the symbiosis between lycaenid butterfly larvae and ants. Science 211:1185–1187.
Price WP, Bouton CE, Gross P, McPheron BA, Thompson JN, Weis AE (1981) Interactions among three trophic levels. Influence of plants on interactions between herbivores and their natural enemies. Ann. Rev. Ecol. Syst. 11:41–65.
Sokal RR, Rohlf FJ (1981) Biometry. W.H. Freeman. San Francisco, CA.
Strong DR, Lawton JH, Southwood TRE (1984) Insect on plants. Harvard U. Press, Cambridge, Mass.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smiley, J.T., Atsatt, P.R. & Pierce, N.E. Local distribution of the lycaenid butterfly, Jalmenus evagoras, in response to host ants and plants. Oecologia 76, 416–422 (1988). https://doi.org/10.1007/BF00377037
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00377037