Abstract
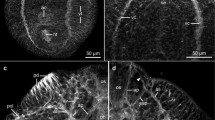
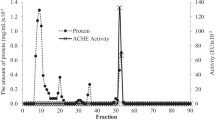
Various parasitic nematodes secrete acetylcholinesterase (AChE). In this study, the localization of AChE in the nematode Nippostrongylus brasiliensis and the secretory forms of AChE in culture fluid were examined. A thiocholine method revealed that AChE activity was localized in the subventral glands, which have a secretory and excretory function via a duct connected to the excretory pore. By electron microscopy, AChE activity was found mainly in the matrix of secretory granules, and sometimes in the Golgi apparatus in the subventral gland cells. These results show that nematode AChE is produced and stored in the subventral glands. Monoclonal antibodies against AChE of human erythrocytes or electric rays also bound to the nematode subventral gland, suggesting immuno-cross-reactivity of AChE among these species. When AChE activity in the nematode excretory-secretory product was examined by SDS polyacrylamide gel electrophoresis combined with the thiocholine method, intense activity was demonstrated as a single band at 74kDa. Immunoblot analysis showed specific recognition of this molecule by IgE and IgG1 antibodies, but not by IgG2a antibody, in nematode-infected rat sera. These results indicate that the nematode AChE molecule produced in and secreted from the subventral glands is antigenic for the production of IgE/IgG1 in host animals.
Similar content being viewed by others
References
Arpagaus M, Richier P, Berge JB, Toutant JP (1992) Acetylcholinesterases of the nematode Steinernema carpocapsae. Characterization of two types of amphiphilic forms differing in their mode of membrane association. Eur J Biochem 207: 1101–1108
Blackburn CC, Selkirk ME (1992) Characterisation of the secretory acetylcholinesterases from adult Nippostrongylus brasiliensis. Mol Biochem Parasitol 53:79–88
Burt JS, Ogilvie BM (1975) In vitro maintenance of nematode parasites assayed by acetylcholinesterase and allergen secretion. Exp Parasitol 38:75–82
Edwards AJ, Burt JS, Ogilvie BM (1971) The effect of immunity upon some enzymes of the parasitic nematode, Nippostrongylus brasiliensis. Parasitology 62:339–347
Espinoza B, Tarrab-Hazdai R, Silman I, Arnon R (1988) Acetylcholinesterase in Schistosoma mansoni is anchored to the membrane via covalently attached phosphatidylinositol. Mol Biochem Parasitol 29:171–179
Johnson CD, Stretton AOW (1980) Neural control of locomotion in Ascaris: anatomy electrophysiology, and biochemistry. In: Zuckerman B (ed) Nematodes as biological models. Academic Press, London, pp 159–195
Karnovsky MJ, Roots L (1964) A “direct-coloring” thiocholine method for cholinesterases. J Histochem Cytochem 12:219–221
Lee DL (1970) The fine structure of the excretory system in adult Nippostrongylus brasiliensis (nematode) and a suggested function for the ‘excretory glands’. Tissue Cell 2:225–231
Lewis PR, Shute CCD (1966) The distribution of cholinesterase in cholinergic neurons demonstrated with the electron microscope. J Cell Sci 1:381–390
Massoulié J (1980) The polymorphism of cholinesterases and its physiological significance. Trends Biochem Sci 5:160–164
Massoulié J, Bon S (1982) The molecular forms of cholinesterase and acetylcholinesterase in vertebrates. Annu Rev Neurosci 5:57–106
McLaren DJ, Burt JS, Ogilvie BW (1974) The anterior glands of adult Necator americanus (Nematoda: stronglyloidea).II. Cytochemical and functional studies. Int J Parasitol 4:39–46
McLean IW, Nakane PK (1974) Periodate-lysine-paraformaldehyde fixative: a new fixative for immunoelectron microscopy. J Histochem Cytochem 22:1077–1083
Ogilvie BM, Jones VE (1971) Nippostrongylus brasiliensis: a review of immunity and the host parasite relationship in the rat. Exp Parasitol 29:138–177
Ogilvie BM, Rothwell TLW, Bremner KC, Schnitzerling HJ, Nolan J, Keith RK (1973) Acetylcholinesterase secretion by parasitic nematodes. I. Evidence for secretion of the enzyme by a number of species. Int J Parasitol 3:589–597
Philipp M (1984) Acetylcholinesterase secreted by intestinal nematodes: a reinterpretation of its putative role of “biochemical holdfast”. Trans R Soc Trop Med Hyg 78:138–139
Rathaur S, Müller S, Maizels RM, Walter RD (1992) Identification of circulating parasite acetylcholinesterase in human and rodent filariasis. Parasitol Res:78:671–676
Sanderson BE (1969) Acetylcholinesterase activity in Nippostrongylus brasiliensis (Nematoda). Comp Biochem Physiol 29:1207–1213
Small DH (1989) Acetylcholinesterases: zymogens of neuropeptide processing enzymes? Neuroscience 29:241–249
Somogyi P, Chubb IW, Smith AD (1975) A possible structural basis for the extracellular release of acetylcholinesterase. Proc R Soc Lond [Biol] 191:271–283
Weise C, Kreienkamp HJ, Raba R, Aaviksaar A, Hucho F (1990) The active site and partial sequence of cobra venom acetylcholinesterase. J Protein Chem 9:53–57
Yamada M, Nakazawa M, Matumoto Y, Arizono N (1991) IgE antibody production in rats against multiple components of excretory-secretory products of the nematode Nippostrongylus brasiliensis. Immunology 72:104–108
Yamada M, Nakazawa M, Arizono N (1993) IgE and IgG2a antibody responses are induced by different antigen groups of the nematode Nippostrongylus brasiliensis in rats. Immunology 78:298–302
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakazawa, M., Yamada, M., Uchikawa, R. et al. Immunocytochemical localization of secretory acetylcholinesterase of the parasitic nematode Nippostrongylus brasiliensis . Cell Tissue Res 280, 59–64 (1995). https://doi.org/10.1007/BF00304511
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304511