Summary
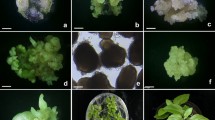
Protoplasts isolated from a totipotent embryogenic cell suspension culture of Zea mays L. (cultivar ‘Dekalb XL82’) underwent sustained cell divisions when cultured in liquid as well as agarose media. Optimal colony formation (5%) occurred in a liquid medium containing 0.5 mg/l 2,4-dichlorophenoxyacetic acid (2,4-D). A soft and unorganized callus was formed when the protocolonies were transferred to agar solidified suspension maintenance medium. Compact, organized and yellow to pale green folded structures and somatic embryos were formed upon subsequent transfer of this callus to a low 2,4-D medium. Clusters of somatic embryos germinated precociously but no plants were recovered.
Similar content being viewed by others
References
Chu CC, Wang CC, Sun CS, Hsu C, Yin KC, Chu CY, Bi FY (1975) Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Sci Sinica 18:659–668
Chourey PS, Zurawski DB (1981) Callus formation from protoplasts of a maize cell culture. Theor Appl Genet 59:341–344
Fromm M, Taylor LP, Walbot V (1985) Expression of genes transferred into monocot and dicot plant cells by electroporation. Proc Natl Acad Sci USA 82:5824–5828
Fromm M, Taylor LP, Walbot V (1986) Stable transformation of maize after gene transfer by electroporation. Nature 319:791–793
Kao KN, Michayluk MR (1975) Nutritional requirements for growth of Vicia hajastana cells and protoplasts at a very low population density in liquid media. Planta 126:105–110
Lu C, Vasil V, Vasil IK (1981) Isolation and culture of protoplasts of Panicum maximum Jacq. (Guinea grass): somatic embryogenesis and plantlet formation. Z Pflanzenphysiol 104:311–318
Ludwig SR, Somers DA, Peterson WL, Pohlman RF, Zarowitz MA, Gengenbach BG, Messing J (1985) High frequency callus formation from maize protoplasts. Theor Appl Genet 71:344–350
Nagata T, Takebe I (1970) Cell wall regeneration and cell division in isolated tobacco mesophyll protoplasts. Planta 92:301–308
Ozias-Akins P, Ferl RJ, Vasil IK (1986) Somatic hybridization in the Gramineae: Pennisetum americanum (L.) K. Schum. (pearl millet) + Panicum maximum Jacq. (Guinea grass). Mol Gen Genet 203:365–370
Srinivasan C, Vasil IK (1986) Plant regeneration from protoplasts of sugarcane (Saccharum officinarum L.). J Plant Physiol 126:41–48
Tabaeizadeh Z, Ferl RJ, Vasil IK (1986) Somatic hybridization in the Gramineae: Saccharum officinarum L. (sugarcane) and Pennisetum americanum (L.) K. Schum. (pearl millet). Proc Natl Acad Sci USA 83:5616–5619
Vasil V, Vasil IK (1980) Isolation and culture of cereal protoplasts. 2. Embryogenesis and plantlet formation from protoplasts of Pennisetum americanum. Theor Appl Genet 56:97–99
Vasil IK, Vasil V (1986) Regeneration in cereal and other grass species. In: Vasil IK (ed) Cell culture and somatic cell genetics of plants, vol 3. plant regeneration and genetic variability. Academic Press, Orlando, pp 121–150
Vasil V, Vasil IK (1986) Plant regeneration from friable embryogenic callus and cell suspension cultures of Zea mays L. J Plant Physiol 124:399–408
Vasil V, Wang D, Vasil V (1983) Plant regeneration from protoplasts of Napier grass (Pennisetum purpureum Schum.). Z Pflanzenphysiol 111:233–239
Yamada Y, Yang ZQ, Tang DT (1986) Plant regeneration from protoplast-derived callus of rice (Oryza sativa L.). Plant Cell Rep 5:85–88
Author information
Authors and Affiliations
Additional information
Communicated by P. L. Pfahler
Rights and permissions
About this article
Cite this article
Vasil, V., Vasil, I.K. Formation of callus and somatic embryos from protoplasts of a commercial hybrid of maize (Zea mays L.). Theoret. Appl. Genetics 73, 793–798 (1987). https://doi.org/10.1007/BF00289381
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00289381