Summary
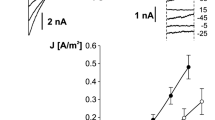
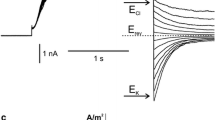
This study is concerned with the characterization of the ionic currents in the vacuolar membrane (tonoplast) of plant cells. Voltage patch-clamp experiments at the whole vacuole and single channel levels were employed to study the effects of cytoplasmic chloride on the tonoplast inward rectifying currents of sugar beet cultured cells. Whole vacuole experiments showed that removal of cytoplasmic chloride induced a decrease in the level of the inward currents, an effect that was reversed upon returning to control levels of cytoplasmic chloride. Substitution of cytoplasmic chloride by any other anion (organic or inorganic) resulted in a reduction in the level of the inward currents. At a given negative tonoplast potential, the inward currents showed a linear relationship with the concentration of cytoplasmic chloride between 10 and 100 mM, with the slope of these relationships increasing as the potential was made more negative. Single channel experiments showed that reduction of cytoplasmic chloride changed the gating mechanism of the channels without affecting the single channel conductance. Reduction of cytoplasmic chloride caused a decrease in the open probability of the tonoplast cation channels by reducing their mean open time and by inducing the appearance of an additional closed state.
Similar content being viewed by others
References
Blumwald, E., Poole, R.J. 1987. Salt adaptation in suspension cultures of sugar beet. Induction of Na+/H+ antiport activity in tonoplast vesicles by salt. Plant Physiol. 83:884–887
Boller, T., Wiemken, A. 1986. Dynamics of vacuolar compartmentation. Annu. Rev. Plant Physiol. 37:137–164
Colombo, R., Cerana, R., Lado, P., Peres, A. 1988. Voltage-dependent channels permeable to K+ and Na+ in the membrane of Acer pseudoplatanus. J. Membrane Biol. 103:227–236
Coyaud, L., Kurkdjian, A., Kado, T., Hedrich, R. 1987. Ion channels and ATP-driven pumps involved in ion transport across the tonoplast of sugar beet vacuoles. Biochim. Biophys. Acta 902:263–268
Critchley, C. 1985. The role of chloride in Photosystem II. Biochim. Biophys. Acta 811:33–46
Dietz, K.J., Jäger, R., Kaiser, G., Martinoia, E. 1989. Amino acid transport across the tonoplast of vacuoles isolated from barley mesophyll protoplasts. Plant Physiol. 92:123–129
Flowers, T.J. 1988. Chloride as a nutrient and as an osmoticum. In: Advances in Plant Nutrition. P.B. Tinker and A. Läuchli, editors. Vol. 3, pp. 55–78. Praeger, New-York
Getz, H.P. 1987. Accumulation of sucrose in vacuoles released from isolated beet root protoplasts by both direct sucrose uptake and UDP-glucose-dependent group translocation. Plant Physiol. Biochem. 25:573–579
Hamill, O.P., Marty, A., Neher, E., Sakmann, B., Sigworth, F.J. 1981. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pfluegers Arch. 391:85–100
Hamilton, K.L., Eaton, D.C. 1985. Single-channel recordings from amiloride-sensitive epithelial sodium channel. Am. J. Physiol. 249:C200-C207
Hedrich, R., Flügge, U.I., Fernandez, J.M. 1986. Patch-clamp studies of ion transport in isolated plant vacuoles. FEBS Lett. 204:228–232
Hedrich, R., Neher, E. 1987. Cytoplasmic calcium regulates voltage-dependent ion channels in plant vacuoles. Nature 329:833–836
Kolb, H.A., Köhler, K., Martinoia, E. 1987. Single potassium channels in membranes of isolated mesophyll barley vacuoles. J. Membrane Biol. 95:163–169
Labarca, P., Coronado, R., Miller, C. 1980. Thermodynamic and kinetic studies of the gating behaviour of a K+-selective channel from the sarcoplasmic reticulum membrane. J. Gen. Physiol. 76:397–424
Maathuis, F.J.M., Prins, H.B.A. 1990. Patch clamp studies on root cell vacuoles of a salt-tolerant and a salt-sensitive Plantago species. Regulation of channel activity by salt stress. Plant Physiol. 92:23–28
McManus, O.B., Magleby, K.L. 1988. Kinetic states and modes of single large-conductance calcium-activated potassium channels in cultured rat skeletal muscle. J. Physiol. 402:79–120
Moczydlowski, E., Latorre, R. 1983. Gating kinetics of Ca2+-activated K+ channels from rat muscle incorporated into planar lipid bilayers. J. Gen. Physiol. 82:511–542
Palmer, L.G., Frindt, G. 1986. Amiloride-sensitive Na channels from the apical membrane of the rat cortical collecting tubule. Proc. Natl. Acad. Sci. USA 63:2767–2770.
Palmer, L.G., Frindt, G. 1987. Effects of cell Ca and pH on Na channels from rat cortical collecting tubule. Am. J. Physiol. 253:F333-F339
Pantoja, O., Dainty, J., Blumwald, E. 1989. Ion channels in vacuoles from halophytes and glycophytes. FEBS Lett. 255: 92–96
Pantoja, O., Dainty, J., Blumwald, E. 1990. Tonoplast ion channels from sugar beet cell suspensions. Inhibition by amiloride and its analogs. Plant Physiol. 94:1788–1794
Rea, P.A., Sanders, D. 1987. Tonoplast energization: Two H+ pumps, one membrane. Physiol. Plant. 71:131–141
Schumaker, K.S., Sze, H. 1985. A Ca+-H+ antiport system driven by the proton electrochemical gradient of a tonoplast H+-ATPase from oat roots. Plant Physiol. 79:1111–1117
Sze, H. 1985. H+-translocating ATPases: Advances using membrane vesicles. Annu. Rev. Plant Physiol. 36:175–208
Willmer, C.M. 1983. Stomata. Longman, London—New York
Author information
Authors and Affiliations
Additional information
This work was supported by the National Science and Engineering Research Council of Canada.
Rights and permissions
About this article
Cite this article
Pantoja, O., Dainty, J. & Blumwald, E. Cytoplasmic chloride regulates cation channels in the vacuolar membrane of plant cells. J. Membarin Biol. 125, 219–229 (1992). https://doi.org/10.1007/BF00236435
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00236435