Summary
In the superposition eyes of the sphingid moth Deilephila and the neuropteran Ascalaphus, adjustment to different intensities is subserved by longitudinal migrations of screening pigment in specialized pigment cells. Using ophthalmoscopic techniques we have localized the light-sensitive trigger that controls pigment position.
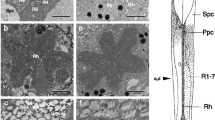
In both species, local illumination of a small spot anywhere within the eye glow of a dark-adapted eye evokes local light adaptation in the ommatidia whose facets receive the light. Details of the response pattern demonstrate that a distal light-sensitive trigger is located axially in the ommatidium, just beneath the crystalline cone, and extends with less sensitivity deep into the clear zone. The distal trigger in Deilephila was shown to be predominantly UV sensitive, and a UV-absorbing structure, presumably the distal trigger, was observed near the proximal tip of the crystalline cone.
In Ascalaphus we also found another trigger located more proximally, which causes local pigment reaction in the ommatidia whose rhabdoms are illuminated (the centre of the eye glow). The light-sensitive trigger for this response appears to be the rhabdom itself.
Similar content being viewed by others
References
Bernard GD, Owens ED, Voss Hurley A (1984) Intracellular optical physiology of the eye of the pyralid moth Amyelois. J Exp Zool 229:173–187
Gogala M (1967) Die spektrale Empfindlichkeit der Doppelaugen von Ascalaphus macaronius Scop. (Neuroptera, Ascalaphidae). Z Vergl Physiol 57:232–243
Hamdorf K, Höglund G (1981) Light induced screening pigment migration independent of visual cell activity. J Comp Physiol 143:305–309
Hamdorf K, Höglund G, Juse A (1986) Ultra-violet and blue induced migration of screening pigment in the retina of the moth Deilephila elpenor. J Comp Physiol A 159:353–362
Hamdorf K, Höglund G, Juse A, Stusek P (1989) Effect of neurotransmitters on movement of screening pigment in insect superposition eyes. Z Naturforsch 44c: 992–998
Höglund G (1966) Pigment migration, light screening and receptor sensitivity in the compound eye of nocturnal Lepidoptera. Acta Physiol Scand, suppl 282:1–56
Höglund G, Struwe G (1970) Pigment migration and spectral sensitivity in the compound eye of a moth. Z Vergl Physiol 67:229–237
Höglund G, Hamdorf K, Rosner G (1973) Trichromatic visual system in an insect and its sensitivity control by blue light. J Comp Physiol 86:265–279
Juse A, Stusek P, Hamdorf K (1989) The properties of photoreconvertible fluorophore systems in insect eyes resemble those of quinones. J Comp Physiol A 165:789–800
Kirschfeld K, Franceschini N (1969) Ein Mechanismus zur Steuerung des Lichtflusses in den Rhabdomeren des Komplexauges von Musca. Kybernetik 6:13–22
Kolb G, Autrum H (1974) Selektive Adaptation und Pigmentwanderung in den Sehzellen des Bienenauges. J Comp Physiol 94:1–6
Kunze P (1972) Comparative studies of arthropod superposition eyes. Z Vergl Physiol 76:347–357
Land MF (1987) Screening pigment migration in a sphingid moth is triggered by light near the cornea. J Comp Physiol A 160:355–357
Nilsson D-E (1989) Optics and evolution of the compound eye. In: Stavenga DG, Hardie R (eds) Facets of vision. Springer, Berlin Heidelberg New York, pp 30–73
Nilsson D-E, Howard J (1989) Intensity and polarization of the eyeshine in butterflies. J Comp Physiol A 166:51–56
Nilsson D-E, Land MF, Howard J (1988) Optics of the butterfly eye. J Comp Physiol A 162:341–366
Nilsson D-E, Henrekson I, Järemo A-C (1989) Pupil control in compound eyes: more than one mechanism in moths. In: Sing N, Strausfeld NJ (eds) Neurobiology of sensory systems. Plenum, New York, pp 17–22
Schlecht P (1979) Colour discrimination in dim light: an analysis of photoreceptor function in the moth Deilephila. J Comp Physiol 129:257–267
Schlecht P, Juse A, Höglund G, Hamdorf K (1987) Photoreconvertible fluorophore systems in rhabdomeres, Semper cells and corneal lenses in the compound eye of the blowfly. J Comp Physiol A 161:227–243
Schneider L, Gogala M, Draslar K, Langer H, Schlecht P (1978) Feinstruktur und Schirmpigment-Eigenschaften der Ommatidien des Doppelauges von Ascalaphus (Insecta, Neuroptera). Cytobiologie 16:274–307
Snyder AW (1979) Physics of vision in compound eyes. In: Autrum H (ed) Handbook of sensory physiology, vol VII/6A. Springer, Berlin Heidelberg New York, pp 225–314
Stavenga DG (1979) Pseudopupils of compound eyes. In: Autrum H (ed) Vision in invertebrates (Handbook of sensory physiology, Vol VII/6A). Springer, Berlin Heidelberg New York, pp 357–439
Stavenga DG, Kuiper JW (1977) Insect pupil mechanisms: I — On the pigment migration in the retinula cells of Hymenoptera (suborder Apocrita). J Comp Physiol 113:55–72
Stavenga DG, Numan JAJ, Tinbergen J, Kuiper JW (1977) Insect pupil mechanisms: II. Pigment migration in the retinula cells of butterflies. J Comp Physiol 113:73–93
Warrant EJ, McIntyre PD (1990) Limitations to resolution in superposition eyes. J Comp Physiol A 167:785–803
Welsch B (1977) Ultrastruktur und funktionelle Morphologie der Augen des Nachtfalters Deilephila elpenor (Lepidoptera, Sphingidae). Cytobiologie 14:378–400
Weyrauther E (1986) Do retinula cells trigger the screening pigment migration in the eye of the moth Ephestia kühniella? J Comp Physiol A 159:55–60
White RH, Banister MJ, Bennett RR (1983) Spectral sensitivity of screening pigment migration in the compound eye of Manduca sexta. J Comp Physiol 153:59–66
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nilsson, D.E., Hamdorf, K. & Höglund, G. Localization of the pupil trigger in insect superposition eyes. J Comp Physiol A 170, 217–226 (1992). https://doi.org/10.1007/BF00196904
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00196904