Summary
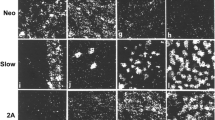
Transitions in myosin patterns were examined in situ by immunofluorescence in two fast muscles of the developing chicken, the pectoralis and the posterior latissimus dorsi. Myosin isoforms were localized using stage-specific monoclonal antibodies against the heavy chain of pectoralis myosin. Two antibodies (12C5 and 10H10) recognize adult and late embryonic myosin. They reacted weakly with both the pectoralis and posterior latissimus dorsi at 10 days in ovo, but intensely at 18 days in ovo. Both muscles were completely unreactive with an adult-specific antibody (5C3), indicating that the staining with 12C5 and 10H10 at 18 days in ovo reflects embryonic myosin. Thus two different embryonic isoforms are expressed sequentially in each muscle. Both 12C5 and 10H10 reacted weakly again with these muscles after hatching. The reappearance of a strong positive response to both antibodies, at 28 days in the pectoralis and after 60 days in the posterior latissimus dorsi, correlated well with the first appearance of a response to the adult-specific antibody, 5C3, signalling the beginning of the adult pattern. Both muscles reacted strongly with an antibody (5B4) specific for ‘neonatal’ myosin between 18 days in ovo and 60 days after hatching. In the pectoralis, embryonic was replaced by neonatal myosin in most fibres by 14 days after hatching; by 28 days, both adult and neonatal myosin were expressed in most fibres; and in the adult, neonatal myosin was replaced entirely by the adult isoform. In contrast, many fibres in the posterior latissimus dorsi still expressed both embryonic and neonatal myosins up to at least 60 days post-hatch, and the remaining fibres expressed the neonatal isoform; the neonatal isoform was present in some fibres even in the adult posterior latissimus dorsi. We have therefore demonstrated in situ four different heavy chain isoforms in two different fast muscles. ‘Early embryonic’, ‘late embryonic’, ‘neonatal’ and eventually ‘adult’ isoforms are expressed in each muscle and more than one isoform often coexists in the same fibre.
Similar content being viewed by others
References
AADER, D., MASAKI, T. & FISCHMAN, D. A. (1982) Immunochemical analysis of myosin heavy chain during myogenesis in vivo and in vitro. J. Cell Biol. 95, 763–70.
AANDMAN, E. & AENNETT, T. (1988) Diversity of fast myosin heavy chain expression during development of gastrocnemius, bicep brachii, and posterior latissimus dorsi muscles in normal and dystrophic chickens. Dev. Biol. 130, 220–31.
AANDMAN, E., MATSUDA, R. & STROHMAN, R. C. (1982) Developmental appearance of myosin heavy and light chain isoforms in vivo and in vitro in chicken skeletal muscle. Dev. Biol. 93, 508–18.
AENFIELD, P. A., LOWEY, S., LEBLANC, D. D. & WALLER, G. S. (1983) Myosin isozymes in avian skeletal muscles. II. Fractionation of myosin isozymes from adult and embryonic chicken pectoralis muscle by immunoaffinity chromatography. J. Muscle Res. Cell Motil. 4, 717–38.
AUCKINGHAM, M. E. (1989) The control of muscle gene expression: a review of molecular studies on the production and processing of primary transcripts. Br. Med. Bull. 45, 608–29.
CROW, M. T. (1987) The determinants of muscle fiber type during embryonic development. Am. Zool. 27, 1043–56.
CROW, M. T. & STOCKDALE, F. E. (1986) The developmental program of fast myosin heavy chain expression in avian skeletal muscles. Dev. Biol. 118, 333–42.
CROW, M. T., OLSON, P. S. & STOCKDALE, F. E. (1983) Myosin light-chain expression during avian muscle development. J. Cell Biol. 96, 736–44.
DHOOT, G. K. (1989) Evidence for the presence of a distinct embryonic isoform of myosin heavy chain in chicken skeletal muscle. Differentiation 40, 176–83.
GAUTHIER, G. F. (1987) Vertebrate muscle fiber types and neuronal regulation of myosin expression. Am. Zool. 27, 1033–42.
GAUTHIER, G. F. (1990) Differential distribution of myosin isoforms among the myofibrils of individual developing muscle fibers. J. Cell Biol. 110, 693–701.
GAUTHIER, G.F. & LOWEY, S. (1977) Polymorphism of myosin among skeletal muscle fiber types. J. Cell Biol. 74, 760–79.
GAUTHIER, G. F. & LOWEY, S. (1979) Distribution of myosin isoenzymes among skeletal muscle fiber types. J. Cell Biol. 81, 10–25.
GAUTHIER, G. F., LOWEY, S., AENFIELD, P.A. & HOBBS, A. W. (1982) Distribution and properties of myosin isozymes in developing avian and mammalian skeletal muscles. J. Cell Biol. 92, 471–484.
GAUTHIER, G. F., AURKE, R. E., LOWEY, S. & HOBBS, A. W. (1983) Myosin isozymes in normal and cross-reinnervated cat skeletal muscle fibers. J. Cell Biol. 97, 756–71.
GAUTHIER, G. F., ONO, R. D. & HOBBS, A. W. (1984) Curare-induced transformation of myosin pattern in developing skeletal muscle fibers. Dev. Biol. 105, 144–54.
GORDON, T., PURVES, R. D. & VRBOVÁ, G. (1977) Differentiation of electrical and contractile properties of slow and fast muscle fibers. J. Physiol. (Lond.) 269, 535–47.
HAMBURGER, V. & HAMILTON, H. L. (1951) A series of normal stages in the development of the chick embryo. J. Morphol. 88, 49–92.
HARRIS, A. J., FITZSIMONS, R. B. & MCEWAN, J. C. (1989) Neural control of the sequence of expression of myosin heavy chain isoforms in foetal mammalian muscles. Development 107, 751–69.
HOFMANN, S., DUSTERHOFT, S. & PETTE, D. (1988) Six myosin heavy chain isoforms are expressed during chick breast muscle development. FEBS Lett. 238, 245–8.
HUGHES, S. M. & ALAU, H. M. (1992) Muscle fiber pattern is independent of cell lineage in postnatal rodent development. Cell 68, 659–71.
KWONG, W. H. & GAUTHIER, G. F. (1987) Neuromuscular junctions in adult and developing fast and slow muscles. Anat. Rec. 219, 409–19.
LOWEY, S., WALLER, G. S. & AANDMAN, E. (1991) Neonatal and adult myosin heavy chains form homodimers during avian skeletal muscle development. J. Cell Biol. 113, 303–10.
LOWEY, S., AENFIELD, P. A., LEBLANC, D. D. & WALLER, G. S. (1983) Myosin isozymes in avian skeletal muscles. I. Sequential expression of myosin isozymes in developing chicken pectoralis muscles. J. Muscle Res. Cell Motil. 4, 695–716.
LOWEY, S., SARTORE, S., GAUTHIER, G. F., WALLER, S. & HOBBS, A. W. (1986) Myosin isozyme transitions in embryonic chicken pectoralis muscle. In Molecular Biology of Muscle Development (edited by EMERSON, C., FISCHMAN, D. A., NADAL-GINARD, B. & SIDDIQUI, M. A. Q.) pp. 225–36. New York: Alan R. Liss.
MILLER, J. B. & STOCKDALE, F. E. (1986) Developmental regulation of the multiple myogenic cell lineages of the avian embryo. J. Cell Biol. 103, 2197–208.
PETTE, D. & VRBOVÁ, G. (1985) Neural control of phenotypic expression in mammalian muscle fibers. Muscle Nerve 8, 676–89.
PETTE, D. & STARON, R. S. (1990) Cellular and molecular diversities of mammalian skeletal muscle fibers. Rev. Physiol. Biochem. Pharmacol. 116, 1–76.
REISER, P. J., STOKES, B. T. & RALL, J. A. (1982) Isometric contractile properties and velocity of shortening during avian myogenesis. Am. J. Physiol. 243, C177-C183.
RUSHBROOK, J. I., WEISS, C., YAO, T-T. & LIN, J. (1988) Complexity of myosin species in the avian posterior latissimus dorsi muscle. J. Muscle Res. Cell Motil. 9, 552–62.
RUSHBROOK, J. I., WEISS, C. & YAO, T-T. (1991) Developmental myosin heavy chain progression in avian type IIB muscle fibres. J. Muscle Res. Cell Motil. 12, 281–91.
SCHAFER, D. A., MILLER, J. B. & STOCKDALE, F. E. (1987) Cell diversification within the myogenic lineage: in vitro generation of two types of myoblasts from a single myogenic progenitor cell. Cell 48, 659–70.
SHEAR, C. R., AANDMAN, E. & ROSSER, B. W. C. (1988) Myosin heavy chain expression during development and following denervation of fast fibers in the red strip of the chicken pectoralis. Dev. Biol. 127, 326–37.
VanHORN, R. & CROW, M. T. (1989) Fast myosin heavy chain expression during the early and late embryonic stages of chicken skeletal muscle development. Dev. Biol. 134, 279–88.
WHALEN, R. G., SELL, S. M., AUTLER-BROWNE, G.S., SCHWARTZ, K., AOUVERET, P. & PINSET-HARSTROM, I. (1981) Three myosin heavy-chain isozymes appear sequentially in rat muscle development. Nature (Lond.) 292, 805–9.
WINKELMANN, D. A. & LOWEY, S. (1986) Probing myosin head structure with monoclonal antibodies. J. Mol. Biol. 188, 595–612.
WINKELMANN, D. A., LOWEY, S. & PRESS, J. L. (1983) Monoclonal antibodies localize changes on myosin heavy chain isozymes during avian myogenesis. Cell 34, 295–306.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gauthier, G.F., Orfanos, G. Developmental transitions in the myosin patterns of two fast muscles. J Muscle Res Cell Motil 14, 99–109 (1993). https://doi.org/10.1007/BF00132184
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00132184