Abstract
The transport of radiolabeled indoleacetic acid (IAA), and some of its conjugates, was investigated in nodal stem segments of Phaseolus vulgaris L. Donor agar blocks containing either [2-acetyl-14C]-IAA; [2-acetyl-14C]-indole-3-acetyl-L-aspartate (IAAsp); [2-acetyl-14C]-indole-3-acetyl-L-glycine (IAGly); or [2-acetyl-14C]-indole-3-acetyl-L-alanine (IAAla) were placed on either the apical or basal cut surface of stem segments each bearing an axillary bud at the midline. In some experiments, a receiver block was placed on the end opposite to the donor. After transport was terminated, the segments were divided into five equal sections plus the bud, and the radioactivity of donors, receivers and each part of the stem segment was counted.
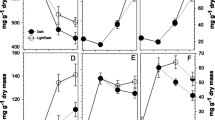
For all four substances tested, the amount of 14C transported to the axillary bud from the base was the same or greater than that from the apical end. After basipetal transport, the distribution of 14C in the segment declined sharply from apex to base. The inverse was true for acropetal transport. Transport for the three IAA conjugates did not differ substantially from each other.
The IAA transport inhibitor, N-1-naphthylphthalamic acid (NPA), inhibited basipetal 14C-IAA transport to the base of the stem segment but did not alter substantially the amount of 14C-IAA recovered from the bud. Transport of 14C-IAA from the apical end to all parts of the stem segment declined when the base of the section was treated with nonradioactive IAA. Taken together with data presented in the accompanying article [Tamas et al. (1989) Plant Growth Regul 8: 165–183], these results suggest that the transport of IAA plays a role in axillary bud growth regulation, but its effect does not depend on the accumulation of IAA in the axillary bud itself.
Similar content being viewed by others
References
Arditti J and Dunn A (1969) Experimental Plant Physiology. New York: Holt, Rinehart and Winston, Inc.
Bandurski RS and Nonhebel HM (1984) Auxins In: MBWilkins, ed. Advanced Plant Physiology, pp. 1–20. London: Pitman
Bialek K and Cohen JD (1986) Isolation and partial characterization of the major amide-linked conjugate of indole-3-acetic acid from Phaseolus vulgaris L. Plant Physiol 80: 99–104
Bialek K, Meudt WJ and Cohen JD (1983) Indole-3-acetic acid (IAA) and IAA conjugates applied to bean stem sections. Plant Physiol 73: 130–134
Cohen JD (1981) Synthesis of 14C-labelled indole-3-acetyl-aspartic acid. J. Labelled Compd Radiopharm 18: 1393–1396
Cohen JD (1982) Identification and quantitative analysis of indole-3-acetyl-L-aspartate from seeds of Glycine max L. Plant Physiol 70: 749–753
Cohen JD and Baldi BG (1983) Studies of endogenous indole-3-acetyl-L-aspartate during germination of soybeans. Proc Plant Growth Reg Soc Am 10: 117–122
Cohen JD and Bandurski RS (1982) Chemistry and physiology of the bound auxins. Ann Rev Plant Physiol 33: 403–430
Epstein E, Baldi BG and Cohen JD (1984) Chromatographic evidence for the presence of indole-3-acetyle-glutamate in seed of soybean. Proc Plant Growth Reg Soc Am 11: 184–188
Epstein E, Cohen JD and Bandurski RS (1980) Concentration and metabolic turnover of indole in germinating kernels of Zea mays L. Plant Physiol 65: 415–421
Hall SM and Hillman JR (1975) Correlative inhibition of lateral bud growth in Phaseolus vulgaris L.: Timing of bud growth following decapitation. Planta 123: 137–143
Morgan DG (1964) Influence of α-naphthylphthalamic acid on the movement of indole-3-acetic acid in plants. Nature 201: 476–477
Morris DA (1977) Transport of exogenous auxin in two-branched pea seedlings (Pisum sativum L.). Planta 136: 91–96
Morris DA and Johnson CF (1987) Regulation of auxin transport in pea (Pisum sativum L.) by phenylacetic acid: Inhibition of polar auxin transport in intact plants and stem segments. Planta 172: 408–416
Nowacki J and Bandurski RS (1980) Myo-inositol esters of indole-3-acetic acid as seed auxin precursors of Zea mays L. Plant Physiol 65: 422–427
Panigrahi BM and Audus LJ (1966) Apical dominance in Vicia faba. Ann Bot 30: 457–473
Tamas IA (1987) Hormonal regulation of apical dominance. In: JPDavies, ed. Plant Hormones and Their Role in Plant Growth and Development, pp. 393–410. Dordrecht: Martinus Nijhoff
Tamas IA, Davies PJ, Mazur BK and Campbell LB (1985) Correlative effects of fruits on plant development. In: RShibles, ed. World Soybean Research Conference III: Proceedings, pp. 858–865. Boulder: Westview Press
Tamas IA, Engles CJ, Kaplan SL, Ozbun JL and Wallace DH (1981) Role of indoleacetic acid and abscisic acid in the correlative control by fruits of axillary bud development and leaf senescence. Plant Physiol 68: 476–481
Tamas IA, Koch JL, Mazur BK and Davies PJ (1986) Auxin effects on the correlative interaction among fruits in Phaseolus vulgaris L. Proc Plant Growth Reg Soc Am 13: 208–215
Tamas IA, Ozbun JL, Wallace DH, Powell LE and Engels CJ (1979) Effect of fruits on dormancy and abscisic acid concentration in axillary buds of Phaseolus vulgaris L. Plant Physiol 64: 615–619
Tamas IA, Schlossberg-Jacobs JL, Lim R, Friedman LB and Barone CC (1989) Effect of plant growth substances on the growth of axillary buds in cultured stem segments of Phaseolus vulgaris L. Plant Growth Regul 8: 165–183
Thimann KV and Skoog F (1933) Studies on the growth hormone of plants. III. The inhibiting action of the growth substance on bud development. Proc nat Acad Sci USA 19: 714–716
Thimann KV and Skoog F (1934) On the inhibition of bud development and other functions of growth substance in Vicia faba. Proc Roy Soc Lond B 114: 317–339
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lim, R., Tamas, I.A. The transport of radiolabeled indoleacetic acid and its conjugates in nodal stem segments of Phaseolus vulgaris L.. Plant Growth Regul 8, 151–164 (1989). https://doi.org/10.1007/BF00024773
Issue Date:
DOI: https://doi.org/10.1007/BF00024773