Abstract
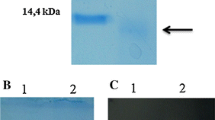
The coding region of copper/zinc-superoxide dismutase (Cu/Zn-SOD) cDNA from sweet potato, Ipomoea batatas (L.) Lam. cv. Tainong 57, was introduced into an expression vector, pET-20b(+). The Cu/Zn-SOD purified by His-tagged technique showed two active forms (dimer and monomer). The amount of proteins of dimer and monomer appeared to be equal, but the activity of dimeric form was seven times higher than that of monomeric form. The enzyme was dissociated into monomer by imidazole buffer above 1.0 M, acidic pH (below 3.0), or SDS (above 1%). The enzyme is quite stable. The enzyme activity is not affected at 85 °C for 20 min, in alkali pH 11.2, or in 0.1 M EDTA and also quite resistant to proteolytic attack. Dimer is more stable than monomer. The thermal inactivation rate constant k dcalculated for the monomer at 85 °C was 0.029 min-1 and the half-life for inactivation was about 28 min. In contrast, there is no significant change of dimer activity after 40 min at 85 °C. The enzyme dimer and monomer retained 83% and 58% of original activity, respectively, after 3 h incubation with trypsin at 37 °C, while those retained 100% and 31% of original activity with chymotrypsin under the same condition. These results suggest subunit interaction might change the enzyme conformation and greatly improve the catalytic activity and stability of the enzyme. It is also possible that the intersubunit contacts stabilize a particular optimal conformation of the protein or the dimeric structure enhances catalytic activity by increasing the electrostatic steering of substrate into the active site.
Similar content being viewed by others
References
Asada K, Kanematsu S, and Hayakawa T: Phylogenic distribution of three types of superoxide dismutase in organisms and in cell organelles. In: Banmister JV, Hill HAO (eds) Chemical and Biochemical Aspects of Superoxide And Superoxide Dismutase, pp. 136–153. Elsevier, Amsterdam (1980).
Bannister JV, Parker MW: The presence of a copper/zinc superoxide dismutase in the bacterium Photobacterium leiognathi: a likely case of gene transfer from eukaryotes to prokaryotes. Proc Natl Acad Sci USA 82: 149–152 (1985).
Beauchamp C, Fridovich I: Improved assays and an assay applicable to acrylamide gel. Anal Biochem 44: 276–287 (1971).
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochemistry 72: 218–254 (1976).
Brock CJ, Walker JE: Superoxide dismutase from Bacillus stearothermophilus. Complete amino acid sequence of a manganese enzyme. Biochemistry 19: 2473–2882 (1980).
Bruschi M, Hatchikian EC, Bonical J, Bovier-Lapierre G, Couchoud P: The N-terminal sequence of superoxide from the strict anaerobe Desulfovibrio desulfuricans. FEBS Lett 76: 121–124 (1977).
Fridovich I: Superoxide dismutases. Adv Enzymol 58: 61–79 (1986).
Goscin SA, Fridivich I: The purification and properties of superoxide dismutase from Saccharomyces cerevisiae. Biochim Biophys Acta 289: 276–283 (1972).
Harris JI, Auffret AD, Northrop FD, Walker JE: Structural comparisons of superoxide dismutases. Eur J Biochemistry 106: 297–303 (1980).
Hassan HM: Determination of microbial damage caused by oxygen free radicals, and the protective role of superoxide dismutase. Meth Enzymol 105: 405–412 (1984).
Inouye K, Osaki A, Tonomura B: Dissociation of dimer of bovine erythrocyte Cu,Zn-superoxide dismutase and activity of the monomer subunit: effects of urea, temperature, and enzyme concentration. J Biochemistry 115: 507–515 (1994).
Kanematsu S, Asada K: Cu,Zn-superoxide dismutase in rice: occurrence of an active monomeric enzyme and two types of isozyme in leaf and non-photosynthetic tissues. Plant Cell Physiol 30: 381–391 (1989).
Lin CT, Yeh KW, Kao MC, Shaw JF: Cloning and characterization of a cDNA encoding the copper/zinc-superoxide dismutase from sweet potato tuberous root. Plant Mol Biol 23: 911–913 (1993).
Malinowski DP, Fridovich I: Bovine erythrocyte superoxide dismutase: diazo coupling, subunit interaction, and electrophoretic variants. Biochemistry 18: 137–144 (1979).
Malinowski DP, Fridovich I: Subunit association and side-chain reactivities of bovine erythrocyte superoxide dismutase in denaturing solvents. Biochemistry 18: 5055–5060 (1979).
Nakano M: Superoxide dismutase composition for periodontal use. US Patent 4, 842, 846: 1–8 (1989).
Stallings WC, Pattridge KA, Strong RK, Ludwig ML: Manganese and iron superoxide dismutases are structural homologs. JBC 259: 10695–10699 (1984).
Steinman HM: The amino acid sequence of mamgano superoxide dismutase from Escherichia coli B. JBC 253: 8708–8720 (1978).
Tainer JA, Getzoff ED, Richardson JS, Richardson DC: Structure and mechanism of copper,zinc superoxide dismutase. Nature 306: 284–287 (1983).
Wilder MS, Mass A: Composition for preventing or alleviating skin irritation by formulations containing superoxide dismutase. US Patent 4, 957, 740: 1–12 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lin, CT., Lin, MT., Chen, YT. et al. Subunit interaction enhances enzyme activity and stability of sweet potato cytosolic Cu/Zn-superoxide dismutase purified by a His-tagged recombinant protein method. Plant Mol Biol 28, 303–311 (1995). https://doi.org/10.1007/BF00020249
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020249