Abstract
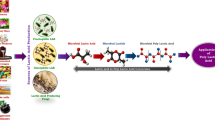
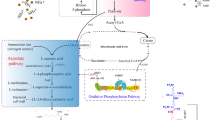
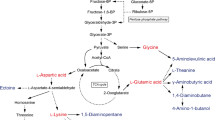
Lactic acid production and its polymerization to poly-lactide (PLA) using renewable resources have recently gained advancement in the field of biomedical science. It is greeted as a promising alternative to tackle the alarmingly environmental, economical and technological issues raised from excessive use of petroleum-based plastics. PLA due to its good processability and biocompatibility always has fascinated researchers in the clinical sector, yet its high degree of hydrophobicity and absence of reactive groups cause steric hindrance and impeded biofunctionalization of PLA surface for cell attachment. PLA production from renewable resources showed a significant reduction in greenhouse gas emissions and fossil energy use as compared to conventional petrochemical-based polymers, thus reducing the threat of global warming. Although lactic acid production from lactic acid bacteria (LAB) is an illustrious domain, production of the green chemical using fungal biomachineries is yet a domain to be explored. Artificial intelligence (AI), a high-tech next-generation technology, is being adapted in all research fields starting from big data analysis to personalized medicines. AI-based mycology modelling for lactic acid production unlocks new prospects for the researcher. The present article is an attempt to explain the potentials of lactic acid production using fungal machineries for sustainable development.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wu H, He AY, Kong XP, Jiang M, Chen XP, Zhu DW, Liu GP, Jin WQ (2015) Acetone–butanol–ethanol production using pH control strategy and immobilized cells in an integrated fermentation–pervaporation process. Process Biochem 50(4):614–622. https://doi.org/10.1016/j.procbio.2014.12.006
Talaia G, Gournas C, Saliba E, Barata-Antunes C, Casal M, André B, Diallinas G, Paiva S (2017) The α-arrestin Bul1p mediates lactate transporter endocytosis in response to alkalinization and distinct physiological signals. J Mol Biol 429(23):3678–3695. https://doi.org/10.1016/j.jmb.2017.09.014
Balla E, Daniilidis V, Karlioti G, Kalamas T, Stefanidou M, Bikiaris ND, Vlachopoulous A, Koumentakou I, Bikiaris DN (2021) Poly (lactic acid): a versatile biobased polymer for the future with multifunctional properties—from monomer synthesis, polymerization techniques and molecular weight increase to PLA applications. Polymers 13(11):1822. https://doi.org/10.3390/polym13111822
Martinez FAC, Balciunas EM, Salgado JM, Gonzalez JMD, Converti A, de Souza Oliveira RP (2013) Lactic acid properties, applications and production: a review. Trends Food Sci Technol 30(1):70–83. https://doi.org/10.1016/j.tifs.2012.11.007
Narayanan N, Roychoudhury PK, Srivastava A (2004) L (+) lactic acid fermentation and its product polymerization. Electron J Biotechnol 7(2):167–178. https://doi.org/10.4067/S0717-34582004000200008
Chen L, Zeng A, Dong H, Li Q, Niu C (2012) A novel process for recovery and refining of L-lactic acid from fermentation broth. Bioresour Technol 112:280–284. https://doi.org/10.1016/j.biortech.2012.02.100
Igoe RS (2011) Dictionary of food ingredients. Springer Science & Business Media
Mohanty JN, Das PK, Nanda S, Nayak P, Pradhan P (2015) Comparative analysis of crude and pure lactic acid produced by lactobacillus fermentum and its inhibitory effects on spoilage bacteria. The pharma. Innovations 3(11, Part A):38
Vaidya AN, Pandey RA, Mudliar S, Kumar MS, Chakrabarti T, Devotta S (2005) Production and recovery of lactic acid for polylactide—an overview. Crit Rev Environ Sci Technol 35(5):429–467. https://doi.org/10.1080/10643380590966181
Komesu A, de Oliveira JAR, da Silva Martins LH, Maciel MRW, Maciel Filho R (2017) Lactic acid production to purification: a review. Bioresources 12(2):4364–4383
Wang L, & Yang ST (2007) Solid state fermentation and its applications. Bioprocessing for value-added products from renewable resources, 465-489. DOI:https://doi.org/10.1016/B978-044452114-9/50019-0
Panesar PS, Kaur S (2015) Bioutilisation of agro-industrial waste for lactic acid production. Int J Food Sci Technol 50(10):2143–2151. https://doi.org/10.1111/ijfs.12886
Gleason FH, Price JS (1969) Lactic acid fermentation in lower fungi. Mycologia 61(5):945–956. https://doi.org/10.1080/00275514.1969.12018817
Zhang ZY, Jin B, Kelly JM (2007) Production of lactic acid from renewable materials by Rhizopus fungi. Biochem Eng J 35(3):251–263. https://doi.org/10.1016/j.bej.2007.01.028
Krishna BS, Nikhilesh GSS, Tarun B, Saibaba N, Gopinadh R (2018) Industrial production of lactic acid and its applications. Int J Biotechnol Res 1:42–54
Gao C, Ma C, Xu P (2011) Biotechnological routes based on lactic acid production from biomass. Biotechnol Adv 29(6):930–939. https://doi.org/10.1016/j.biotechadv.2011.07.022
Wang Z, Wang Y, Yang ST, Wang R, Ren H (2010) A novel honeycomb matrix for cell immobilization to enhance lactic acid production by Rhizopus oryzae. Bioresour Technol 101(14):5557–5564. https://doi.org/10.1016/j.biortech.2010.02.064
Okano K, Tanaka T, Ogino C, Fukuda H, Kondo A (2010) Biotechnological production of enantiomeric pure lactic acid from renewable resources: recent achievements, perspectives, and limits. Appl Microbiol Biotechnol 85(3):413–423. https://doi.org/10.1007/s00253-009-2280-5
Das M, Kundu D, Rastogi A, Singh J, Banerjee R (2018) Biotechnological exploitation of poly-lactide produced from cost effective lactic acid. Principles and applications of fermentation technology, vol 18. Wiley, Hoboken, NJ, pp 401–416. https://doi.org/10.1002/9781119460381.ch18
Mayo B, Aleksandrzak-Piekarczyk T, Fernández M, Kowalczyk M, Álvarez-Martín P, & Bardowski J (2010) Updates in the metabolism of lactic acid bacteria. Biotechnology of lactic acid bacteria: Novel applications, 3-33. https://doi.org/10.1002/9780813820866.ch1
Juturu V, Wu JC (2016) Microbial production of lactic acid: the latest development. Crit Rev Biotechnol 36(6):967–977. https://doi.org/10.3109/07388551.2015.1066305
Abdel-Rahman MA, Tashiro Y, Sonomoto K (2013) Recent advances in lactic acid production by microbial fermentation processes. Biotechnol Adv 31(6):877–902. https://doi.org/10.1016/j.biotechadv.2013.04.002
Abedi E, Hashemi SMB (2020) Lactic acid production–producing microorganisms and substrates sources-state of art. Heliyon 6(10):e04974. https://doi.org/10.1016/j.heliyon.2020.e04974
Ge XY, Qian H, Zhang WG (2009) Improvement of l-lactic acid production from Jerusalem artichoke tubers by mixed culture of Aspergillus niger and Lactobacillus sp. Bioresour Technol 100(5):1872–1874. https://doi.org/10.1016/j.biortech.2008.09.049
John RP, Anisha GS, Nampoothiri KM, Pandey A (2009) Direct lactic acid fermentation: focus on simultaneous saccharification and lactic acid production. Biotechnol Adv 27(2):145–152. https://doi.org/10.1016/j.biotechadv.2008.10.004
Thitiprasert S, Sooksai S, Thongchul N (2011) In vivo regulation of alcohol dehydrogenase and lactate dehydrogenase in Rhizopus oryzae to improve L-lactic acid fermentation. Appl Biochem Biotechnol 164(8):1305–1322. https://doi.org/10.1007/s12010-011-9214-2
van Maris AJ, Konings WN, van Dijken JP, Pronk JT (2004) Microbial export of lactic and 3-hydroxypropanoic acid: implications for industrial fermentation processes. Metab Eng 6(4):245–255. https://doi.org/10.1016/j.ymben.2004.05.001
Casal M, Queirós O, Talaia G, Ribas D, & Paiva S (2016) Carboxylic acids plasma membrane transporters in Saccharomyces cerevisiae. Yeast membrane transport, 229-251.DOI: https://doi.org/10.1007/978-3-319-25304-6_9
Hettema EH, Van Roermund CW, Distel B, van den Berg M, Vilela C, Rodrigues-Pousada C, Wanders RJ, Tabak HF (1996) The ABC transporter proteins Pat1 and Pat2 are required for import of long-chain fatty acids into peroxisomes of Saccharomyces cerevisiae. EMBO J 15(15):3813–3822. https://doi.org/10.1002/j.1460-2075.1996.tb00755.x
Okorokov LA, Kulakovskaya TV, Lichko LP, Polorotova EV (1985) H+/ion antiport as the principal mechanism of transport systems in the vacuolar membrane of the yeast Saccharomyces carlsbergensis. FEBS Lett 192(2):303–306. https://doi.org/10.1016/0014-5793(85)80130-7
Grobler J, Bauer F, Subden RE, Van Vuuren HJ (1995) The mae1 gene of Schizosaccharomyces pombe encodes a permease for malate and other C4 dicarboxylic acids. Yeast 11(15):1485–1491. https://doi.org/10.1002/yea.320111503
Casal M, Paiva S, Andrade RP, Gancedo C, Leão C (1999) The lactate-proton symport of Saccharomyces cerevisiae is encoded by JEN1. J Bacteriol 181(8):2620–2623. https://doi.org/10.1128/JB.181.8.2620-2623.1999
Wakamatsu M, Tomitaka M, Tani T, Taguchi H, Kida K, Akamatsu T (2013) Improvement of ethanol production from D-lactic acid by constitutive expression of lactate transporter Jen1p in Saccharomyces cerevisiae. Biosci Biotechnol Biochem 77(5):1114–1116. https://doi.org/10.1271/bbb.120985
Soares-Silva I, Sá-Pessoa J, Myrianthopoulos V, Mikros E, Casal M, Diallinas G (2011) A substrate translocation trajectory in a cytoplasm-facing topological model of the monocarboxylate/H+ symporter Jen1p. Mol Microbiol 81(3):805–817. https://doi.org/10.1111/j.1365-2958.2011.07729.x
Soares-Silva I, Paiva S, Diallinas G, Casal M (2007) The conserved sequence NXX [S/T] HX [S/T] QDXXXT of the lactate/pyruvate: H+ symporter subfamily defines the function of the substrate translocation pathway. Mol Membr Biol 24(5–6):464–474. https://doi.org/10.1080/09687680701342669
Alves R, Sousa-Silva M, Vieira D, Soares P, Chebaro Y, Lorenz MC, Casai M, Soares-Silva, Paiva S (2020) Carboxylic acid transporters in Candida pathogenesis. MBio 11(3):e00156–e00120. https://doi.org/10.1128/mBio.00156-20
Andrade RP, Casal M (2001) Expression of the lactate permease gene JEN1 from the yeast Saccharomyces cerevisiae. Fungal Genet Biol 32(2):105–111. https://doi.org/10.1006/fgbi.2001.1254
Sanz P, Alms GR, Haystead TA, Carlson M (2000) Regulatory interactions between the Reg1-Glc7 protein phosphatase and the Snf1 protein kinase. Mol Cell Biol 20(4):1321–1328. https://doi.org/10.1128/MCB.20.4.1321-1328.2000
Haurie V, Perrot M, Mini T, Jenö P, Sagliocco F, Boucherie H (2001) The transcriptional activator Cat8p provides a major contribution to the reprogramming of carbon metabolism during the diauxic shift in Saccharomyces cerevisiae. J Biol Chem 276(1):76–85. https://doi.org/10.1074/jbc.M008752200
Olesen JT, Guarente L (1990) The HAP2 subunit of yeast CCAAT transcriptional activator contains adjacent domains for subunit association and DNA recognition: model for the HAP2/3/4 complex. Genes Dev 4(10):1714–1729. https://doi.org/10.1101/gad.4.10.1714
Mota S, Vieira N, Barbosa S, Delaveau T, Torchet C, Le Saux A, Garcia M, Pereira A, Lemoine S, Couipier F, Darzacq X, Benard L, Casal M, Devaux F, Paiva S (2014) Role of the DHH1 gene in the regulation of monocarboxylic acids transporters expression in Saccharomyces cerevisiae. PLoS One 9(11):e111589. https://doi.org/10.1371/journal.pone.0111589
Becuwe M, Vieira N, Lara D, Gomes-Rezende J, Soares-Cunha C, Casal M, Haguenauer-Tsapis R, Vincent O, Paiva S, Léon S (2012) A molecular switch on an arrestin-like protein relays glucose signalling to transporter endocytosis. J Cell Biol 196(2):247–259. https://doi.org/10.1083/jcb.201109113
Paiva S, Vieira N, Nondier I, Haguenauer-Tsapis R, Casal M, Urban-Grimal D (2009) Glucose-induced ubiquitylation and endocytosis of the yeast Jen1 transporter: role of lysine 63-linked ubiquitin chains. J Biol Chem 284(29):19228–19236. https://doi.org/10.1074/jbc.M109.008318
de Kok S, Nijkamp JF, Oud B, Roque FC, de Ridder D, Daran JM, Pronk JT, van Maris AJ (2012) Laboratory evolution of new lactate transporter genes in a jen1 Δ mutant of Saccharomyces cerevisiae and their identification as ADY2 alleles by whole-genome resequencing and transcriptome analysis. FEMS Yeast Res 12(3):359–374. https://doi.org/10.1111/j.1567-1364.2011.00787.x
Paiva S, Devaux F, Barbosa S, Jacq C, Casal M (2004) Ady2p is essential for the acetate permease activity in the yeast Saccharomyces cerevisiae. Yeast 21(3):201–210. https://doi.org/10.1002/yea.1056
Augstein A, Barth K, Gentsch M, Kohlwein SD, Barth G (2003) Characterization, localization and functional analysis of Gpr1p, a protein affecting sensitivity to acetic acid in the yeast Yarrowia lipolytica. Microbiology 149(3):589–600. https://doi.org/10.1099/mic.0.25917-0
Ribas D, Soares-Silva I, Vieira D, Sousa-Silva M, Sa-Pessoa J, Azevedo-Silva J, Viegas SC, Arraiano CM, Diallinas G, Paiva S, Soares P, Casal M (2019) The acetate uptake transporter family motif “NPAPLGL (M/S)” is essential for substrate uptake. Fungal Genet Biol 122:1–10. https://doi.org/10.1016/j.fgb.2018.10.001
Pacheco A, Talaia G, Sá-Pessoa J, Bessa D, Gonçalves MJ, Moreira R, Paiva S, Casal M, Queirós O (2012) Lactic acid production in Saccharomyces cerevisiae is modulated by expression of the monocarboxylate transporters Jen1 and Ady2. FEMS Yeast Res 12(3):375–381. https://doi.org/10.1111/j.1567-1364.2012.00790.x
Turner TL, Lane S, Jayakody LN, Zhang GC, Kim H, Cho W, Jin YS (2019) Deletion of JEN1 and ADY2 reduces lactic acid yield from an engineered Saccharomyces cerevisiae, in xylose medium, expressing a heterologous lactate dehydrogenase. FEMS Yeast Res 19(6):foz050. https://doi.org/10.1093/femsyr/foz050
Singh SK, Ahmed SU, Pandey A (2006) Metabolic engineering approaches for lactic acid production. Process Biochem 41(5):991–1000. https://doi.org/10.1016/j.procbio.2005.12.004
Upadhyaya BP, DeVeaux LC, Christopher LP (2014) Metabolic engineering as a tool for enhanced lactic acid production. Trends Biotechnol 32(12):637–644. https://doi.org/10.1016/j.tibtech.2014.10.005
Wakai S, Arazoe T, Ogino C, Kondo A (2017) Future insights in fungal metabolic engineering. Bioresour Technol 245:1314–1326. https://doi.org/10.1016/j.biortech.2017.04.095
Narendranath NV, Thomas KC, Ingledew WM (2001) Effects of acetic acid and lactic acid on the growth of Saccharomyces cerevisiae in a minimal medium. J Ind Microbiol Biotechnol 26(3):171–177. https://doi.org/10.1038/sj.jim.7000090
Peetermans A, Foulquié-Moreno MR, Thevelein JM (2021) Mechanisms underlying lactic acid tolerance and its influence on lactic acid production in Saccharomyces cerevisiae. Microbial. Cell 8(6):111. https://doi.org/10.15698/mic2021.06.751
Sugiyama M, Akase SP, Nakanishi R, Kaneko Y, Harashima S (2016) Overexpression of ESBP6 improves lactic acid resistance and production in Saccharomyces cerevisiae. J Biosci Bioeng 122(4):415–420. https://doi.org/10.1016/j.jbiosc.2016.03.010
Zhong W, Yang M, Hao X, Sharshar MM, Wang Q, Xing J (2020) Improvement of D-lactic acid production at low pH through expressing acid-resistant gene IoGAS1 in engineered Saccharomyces cerevisiae. J Chem Technol Biotechnol 96(3):732–742. https://doi.org/10.1002/jctb.6587
Baek SH, Kwon EY, Kim YH, Hahn JS (2015) Metabolic engineering and adaptive evolution for efficient production of D-lactic acid in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 100(6):2737–2748. https://doi.org/10.1007/s00253-015-7174-0
Kuanyshev N, Rao CV, Dien B, Jin YS (2020) Domesticating a food spoilage yeast into an organic acid-tolerant metabolic engineering host: lactic acid production by engineered Zygosaccharomyces bailii. Biotechnol Bioeng 118(1):372–382. https://doi.org/10.1002/bit.27576
Flikweert MT, de Swaaf M, van Dijken JP, Pronk JT (1999) Growth requirements of pyruvate-decarboxylase-negative Saccharomyces cerevisiae. FEMS Microbiol Lett 174(1):73–79. https://doi.org/10.1111/j.1574-6968.1999.tb13551.x
Song JY, Park JS, Kang CD, Cho HY, Yang D, Lee S, Cho KM (2016) Introduction of a bacterial acetyl-CoA synthesis pathway improves lactic acid production in Saccharomyces cerevisiae. Metab Eng 35:38–45. https://doi.org/10.1016/j.ymben.2015.09.006
Zhong W, Yang M, Mu T, Wu F, Hao X, Chen R, Sharshar MM, Thygesen A, Wang Q, Xing J (2019) Systematically redesigning and optimizing the expression of D-lactate dehydrogenase efficiently produces high-optical-purity D-lactic acid in Saccharomyces cerevisiae. Biochem Eng J 144:217–226. https://doi.org/10.1016/j.bej.2018.09.013
Yamada R, Ogura K, Kimoto Y, Ogino H (2019) Toward the construction of a technology platform for chemicals production from methanol: D-lactic acid production from methanol by an engineered yeast Pichia pastoris. World J Microbiol Biotechnol 35(2):37. https://doi.org/10.1007/s11274-019-2610-4
Kong X, Zhang B, Hua Y, Zhu Y, Li W, Wang D, Hong J (2019) Efficient L-lactic acid production from corncob residue using metabolically engineered thermo-tolerant yeast. Bioresour Technol 273:220–230. https://doi.org/10.1016/j.biortech.2018.11.018
Gomes D, Rodrigues AC, Domingues L, Gama M (2015) Cellulase recycling in biorefineries is it possible? Appl Microbiol Biotechnol 99(10):4131–4143. https://doi.org/10.1007/s00253-015-6535-z
Mori T, Kako H, Sumiya T, Kawagishi H, Hirai H (2016) Direct lactic acid production from beech wood by transgenic white-rot fungus Phanerochaete sordida YK-624. J Biotechnol 239:83–89. https://doi.org/10.1016/j.jbiotec.2016.10.014
Yamada R, Kumata Y, Mitsui R, Matsumoto T, Ogino H (2021) Improvement of lactic acid tolerance by cocktail δ-integration strategy and identification of the transcription factor PDR3 responsible for lactic acid tolerance in yeast Saccharomyces cerevisiae. World J Microbiol Biotechnol 37(2):1–10. https://doi.org/10.1007/s11274-020-02977-1
Yamada R, Wakita K, Mitsui R, Ogino H (2017) Enhanced d-lactic acid production by recombinant Saccharomyces cerevisiae following optimization of the global metabolic pathway. Biotechnol Bioeng 114(9):2075–2084. https://doi.org/10.1002/bit.26330
Kim JW, Jang JH, Yeo HJ, Seol J, Kim SR, Jung YH (2019) Lactic acid production from a whole slurry of acid-pretreated spent coffee grounds by engineered Saccharomyces cerevisiae. Appl Biochem Biotechnol 189(1):206–216. https://doi.org/10.1007/s12010-019-03000-6
Hábová V, Melzoch K, Rychtera M, Sekavová B (2004) Electrodialysis as a useful technique for lactic acid separation from a model solution and a fermentation broth. Desalination 162:361–372. https://doi.org/10.1016/S0011-9164(04)00070-0
Joglekar HG, Rahman I, Babu S, Kulkarni BD, Joshi A (2006) Comparative assessment of downstream processing options for lactic acid. Sep Purif Technol 52(1):1–17. https://doi.org/10.1016/j.seppur.2006.03.015
Hulse GK, Stalenberg V, McCallum D, Smit W, O'neil G, Morris N, Tait RJ (2005) Histological changes over time around the site of sustained release naltrexone-poly (DL-lactide) implants in humans. J Control Release 108(1):43–55. https://doi.org/10.1016/j.jconrel.2005.08.001
Lunelli BH, Andrade RR, Atala DI, Maciel MRW, Maugeri Filho F, Maciel Filho R (2010) Production of lactic acid from sucrose: strain selection, fermentation, and kinetic modeling. Appl Biochem Biotechnol 161(1):227–237. https://doi.org/10.1007/s12010-009-8828-0
Komesu A, Wolf Maciel MR, Rocha de Oliveira JA, da Silva Martins LH, Maciel Filho R (2017) Purification of lactic acid produced by fermentation: focus on non-traditional distillation processes. Sep Purif Rev 46(3):241–254. https://doi.org/10.1080/15422119.2016.1260034
Krzyżaniak A, Leeman M, Vossebeld F, Visser TJ, Schuur B, de Haan AB (2013) Novel extractants for the recovery of fermentation derived lactic acid. Sep Purif Technol 111:82–89. https://doi.org/10.1016/j.seppur.2013.03.031
Nakano S, Ugwu CU, Tokiwa Y (2012) Efficient production of d-(−)-lactic acid from broken rice by lactobacillus delbrueckii using ca(OH)2 as a neutralizing agent. Bioresour Technol 104:791–794. https://doi.org/10.1016/j.biortech.2011.10.017
Idler C, Venus J, Kamm B (2015) Microorganisms for the production of lactic acid and organic lactates. Microorg Biorefineries 26:225–273. https://doi.org/10.1007/978-3-662-45209-7_9
Bailly M (2002) Production of organic acids by bipolar electrodialysis: realizations and perspectives. Desalination 144(1–3):157–162. https://doi.org/10.1016/S0011-9164(02)00305-3
Choi JH, Kim SH, Moon SH (2002) Recovery of lactic acid from sodium lactate by ion substitution using ion-exchange membrane. Sep Purif Technol 28(1):69–79. https://doi.org/10.1016/S1383-5866(02)00014-X
Lee EG, Moon SH, Chang YK, Yoo IK, Chang HN (1998) Lactic acid recovery using two-stage electrodialysis and its modelling. J Membr Sci 145(1):53–66. https://doi.org/10.1016/S0376-7388(98)00065-9
Kim YH, Moon SH (2001) Lactic acid recovery from fermentation broth using one-stage electrodialysis. J Chem Technol Biotechnol 76(2):169–178. https://doi.org/10.1002/jctb.368
Martins PF, Medeiros HHR, Sbaite P, Maciel MW (2013) Enrichment of oxyterpenes from orange oil by short path evaporation. Sep Purif Technol 116:385–390. https://doi.org/10.1016/j.seppur.2013.06.011
Komesu A, Martins PF, Lunelli BH, Oliveira J, Maciel Filho R, Maciel MRW (2014) Evaluation of lactic acid purification from fermentation broth by hybrid short path evaporation using factorial experimental design. Sep Purif Technol 136:233–240. https://doi.org/10.1016/j.seppur.2014.09.010
Luongo V, Policastro G, Ghimire A, Pirozzi F, Fabbricino M (2019) Repeated-batch fermentation of cheese whey for semi-continuous lactic acid production using mixed cultures at uncontrolled pH. Sustainability 11(12):3330. https://doi.org/10.3390/su11123330
Kamble SP, Barve PP, Joshi JB, Rahman I, Kulkarni BD (2012) Purification of lactic acid via esterification of lactic acid using a packed column, followed by hydrolysis of methyl lactate using three continuously stirred tank reactors (CSTRs) in series: a continuous pilot plant study. Ind Eng Chem Res 51(4):1506–1514. https://doi.org/10.1021/ie200642j
Sanz MT, Murga R, Beltrán S, Cabezas JL, Coca J (2004) Kinetic study for the reactive system of lactic acid esterification with methanol: methyl lactate hydrolysis reaction. Ind Eng Chem Res 43(9):2049–2053. https://doi.org/10.1021/ie034031p
U.S. Patent 6,384,276,2002 Process for the preparation of lactic acid by evaporative crystallisation
Filachione EM, Fisher CH (1946) Purification of lactic acid. Ind Eng Chem 38(2):228–232
Arora MB, Hestekin JA, Snyder SW, St. Martin EJ, Lin YJ, Donnelly MI, Millard CS (2007) The separative bioreactor: a continuous separation process for the simultaneous production and direct capture of organic acids. Sep Sci Technol 42(11):2519–2538. https://doi.org/10.1080/01496390701477238
Bai DM, Jia MZ, Zhao XM, Ban R, Shen F, Li XG, Xu SM (2003) L (+)-lactic acid production by pellet-form Rhizopus oryzae R1021 in a stirred tank fermentor. Chem Eng Sci 58(3–6):785–791. https://doi.org/10.1016/S0009-2509(02)00608-5
Marták J, Schlosser Š, Sabolová E, Krištofı́ková L, Rosenberg M (2003) Fermentation of lactic acid with Rhizopus arrhizus in a stirred tank reactor with a periodical bleed and feed operation. Process Biochem 38(11):1573–1583. https://doi.org/10.1016/S0032-9592(03)00059-1
Park EY, Kosakai Y, Okabe M (1998) Efficient production of l-(+)-lactic acid using mycelial cotton-like flocs of Rhizopus oryzae in an air-lift bioreactor. Biotechnol Prog 14(5):699–704. https://doi.org/10.1021/bp9800642
Thongchul N, Yang ST (2003) Controlling filamentous fungal morphology by immobilization on a rotating fibrous matrix to enhance oxygen transfer and L (+)-lactic acid production by Rhizopus oryzae. ACS Symp Ser 862(3):36–51. https://doi.org/10.1021/bk-2003-0862.ch003
Du J, Cao N, Gong CS, Tsao GT (1998) Production of L-lactic acid by Rhizopus oryzae in a bubble column fermenter. Appl Biochem Biotechnol 70:323–329. https://doi.org/10.1007/BF02920148
Zhou Y, Domínguez JM, Cao N, Du J, & Tsao GT (1999) Optimization of L-lactic acid production from glucose by Rhizopus oryzae ATCC 52311. In Twentieth symposium on biotechnology for fuels and chemicals (pp. 401–407). Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-4612-1604-9_37
Miura S, Arimura T, Hoshino M, Kojima M, Dwiarti L, Okabe M (2003) Optimization and scale-up of l-lactic acid fermentation by mutant strain Rhizopus sp. MK-96-1196 in airlift bioreactors. J Biosci Bioeng 96(1):65–69. https://doi.org/10.1016/S1389-1723(03)90098-3
Yin P, Nishina N, Kosakai Y, Yahiro K, Pakr Y, Okabe M (1997) Enhanced production of L (+)-lactic acid from corn starch in a culture of Rhizopus oryzae using an air-lift bioreactor. J Ferment Bioeng 84(3):249–253. https://doi.org/10.1016/S0922-338X(97)82063-6
Silva EM, Yang ST (1995) Kinetics and stability of a fibrous-bed bioreactor for continuous production of lactic acid from unsupplemented acid whey. J Biotechnol 41(1):59–70. https://doi.org/10.1016/0168-1656(95)00059-Y
Eş I, Khaneghah AM, Barba FJ, Saraiva JA, Sant'Ana AS, Hashemi SMB (2018) Recent advancements in lactic acid production-a review. Food Res Int 107:763–770. https://doi.org/10.1016/j.foodres.2018.01.001
Shi Z, Wei P, Zhu X, Cai J, Huang L, Xu Z (2012) Efficient production of l-lactic acid from hydrolysate of Jerusalem artichoke with immobilized cells of Lactococcus lactis in fibrous bed bioreactors. Enzym Microb Technol 51(5):263–268. https://doi.org/10.1016/j.enzmictec.2012.07.007
Chotisubha-anandha N, Thitiprasert S, Tolieng V, Thongchul N (2011) Improved oxygen transfer and increased L-lactic acid production by morphology control of Rhizopus oryzae in a static bed bioreactor. Bioprocess Biosyst Eng 34(2):163–172. https://doi.org/10.1007/s00449-010-0457-z
Pimtong V, Ounaeb S, Thitiprasert S, Tolieng V, Sooksai S, Boonsombat R, Tanasupawat S, Assabumrungrat S, Thongchul N (2017) Enhanced effectiveness of Rhizopus oryzae by immobilization in a static bed fermentor for l-lactic acid production. Process Biochem 52:44–52. https://doi.org/10.1016/j.procbio.2016.09.020
Kosseva MR, Panesar PS, Kaur G, Kennedy JF (2009) Use of immobilised biocatalysts in the processing of cheese whey. Int J Biol Macromol 45(5):437–447. https://doi.org/10.1016/j.ijbiomac.2009.09.005
Faschian R, De S, Pörtner R (2016) Multi-fixed-bed bioreactor system applied for bioprocess development of immobilized lactic acid bacteria. Open Biotechnol J 10:1. https://doi.org/10.2174/1874070701610010001
Fan R, Ebrahimi M, Czermak P (2017) Anaerobic membrane bioreactor for continuous lactic acid fermentation. Membranes 7(2):26. https://doi.org/10.3390/membranes7020026
Smith Jr, CV (1969) The use of ultrafiltration membrane for activated sludge separation. In Proc. 24th Annual Purdue Industrial Waste Conf. (pp. 1300-1310)
Carrère H, Blaszkow F, de Balmann HR (2001) Modelling the clarification of lactic acid fermentation broths by cross-flow microfiltration. J Membr Sci 186(2):219–230. https://doi.org/10.1016/S0376-7388(00)00677-3
Cysewski GR, Wilke CR (1977) Rapid ethanol fermentations using vacuum and cell recycle. Biotechnol Bioeng 19(8):1125–1143. https://doi.org/10.1002/bit.260190804
Puziss M, Hedén CG (1965) Toxin production by clostridium tetani in biphasic liquid cultures. Biotechnol Bioeng 7(3):355–366. https://doi.org/10.1002/bit.260070305
Kwon S, Yoo IK, Lee WG, Chang HN, Chang YK (2001) High-rate continuous production of lactic acid by lactobacillus rhamnosus in a two-stage membrane cell-recycle bioreactor. Biotechnol Bioeng 73(1):25–34. https://doi.org/10.1002/1097-0290(20010405)73:1%3C25::AID-BIT1033%3E3.0.CO;2-N
Kulozik U, Hammelehle B, Pfeifer J, Kessler HG (1992) High reaction rate continuous bioconversion process in a tubular reactor with narrow residence time distributions for the production of lactic acid. J Biotechnol 22(1–2):107–116. https://doi.org/10.1016/0168-1656(92)90135-V
Jeantet R, Maubois JL, Boyaval P (1996) Semicontinuous production of lactic acid in a bioreactor coupled with nanofiltration membranes. Enzym Microb Technol 19(8):614–619. https://doi.org/10.1016/S0141-0229(96)00073-7
Wenten IG, Khoiruddin K, Aryanti PT, Victoria AV, Tanukusuma G (2018) Membrane-based zero-sludge palm oil mill plant. Rev Chem Eng 36(2):237–263. https://doi.org/10.1515/revce-2017-0117
Zhang Y, Chen X, Qi B, Luo J, Shen F, Su Y, Khan R, Wan Y (2014) Improving lactic acid productivity from wheat straw hydrolysates by membrane integrated repeated batch fermentation under non-sterilized conditions. Bioresour Technol 163:160–166. https://doi.org/10.1016/j.biortech.2014.04.038
Moueddeb H, Sanchez J, Bardot C, Fick M (1996) Membrane bioreactor for lactic acid production. J Membr Sci 114(1):59–71. https://doi.org/10.1016/0376-7388(95)00307-X
Tay A, Yang ST (2002) Production of L (+)-lactic acid from glucose and starch by immobilized cells of Rhizopus oryzae in a rotating fibrous bed bioreactor. Biotechnol Bioeng 80(1):1–12. https://doi.org/10.1002/bit.10340
Liu T, Miura S, Yaguchi M, Arimura T, Park EY, Okabe M (2006) Scale-up of L-lactic acid production by mutant strain Rhizopus sp. Mk-96-1196 from 0.003 m3 to 5 m3 in airlift bioreactors. J Biosci Bioeng 101(1):9–12. https://doi.org/10.1263/jbb.101.9
Abubakar A, Suberu HA, Bello IM, Abdulkadir R, Daudu OA, Lateef AA (2013) Effect of pH on mycelial growth and sporulation of Aspergillus parasiticus. J Plant Sci 1(4):64–67. https://doi.org/10.11648/j.jps.20130104.13
Niaz M, Iftikhar T, Qureshi FF, Niaz M (2014) Extracellular lipase production by Aspergillus nidulans (MBL-S-6) under submerged fermentation. Int J Agric Biol 16(3):536–542
Jin T, Zhang H (2008) Biodegradable polylactic acid polymer with nisin for use in antimicrobial food packaging. J Food Sci 73(3):M127–M134. https://doi.org/10.1111/j.1750-3841.2008.00681.x
Shahri SZ, Vahabzadeh F, Mogharei A (2020) Lactic acid production by loofah-immobilized Rhizopus oryzae through one-step fermentation process using starch substrate. Bioprocess Biosyst Eng 43(2):333–345. https://doi.org/10.1007/s00449-019-02231-5
Teke GM, Pott RW (2020) Design and evaluation of a continuous semipartition bioreactor for in situ liquid-liquid extractive fermentation. Biotechnol Bioeng 118(1):58–71. https://doi.org/10.1002/bit.27550
Singhvi M, Gokhale D (2013) Biomass to biodegradable polymer (PLA). RSC Adv 3(33):13558–13568. https://doi.org/10.1039/C3RA41592A
Farah S, Anderson DG, Langer R (2016) Physical and mechanical properties of PLA, and their functions in widespread applications—a comprehensive review. Adv Drug Deliv Rev 107:367–392. https://doi.org/10.1016/j.addr.2016.06.012
Gupta B, Revagade N, Hilborn J (2007) Poly (lactic acid) fiber: an overview. Prog Polym Sci 32(4):455–482. https://doi.org/10.1016/j.progpolymsci.2007.01.005
Jamshidian M, Tehrany EA, Imran M, Jacquot M, Desobry S (2010) Poly-lactic acid: production, applications, nanocomposites, and release studies. Compr Rev Food Sci Food Saf 9(5):552–571. https://doi.org/10.1111/j.1541-4337.2010.00126.x
Garlotta D (2001) A literature review of poly (lactic acid). J Polym Environ 9(2):63–84. https://doi.org/10.1023/A:1020200822435
Ghosh K, Jones BH (2021) Roadmap to biodegradable plastics—current state and research needs. ACS Sustain Chem Eng 9(18):6170–6187. https://doi.org/10.1021/acssuschemeng.1c00801
Emergen Research. Polylactic Acid Market By Product Type (Racemic PLLA (Poly-L-lactic Acid), Regular PLLA (Poly-L-lactic Acid), PDLA (Poly-D-lactic Acid), PDLLA (Poly-DL-lactic Acid)), Distribution Channel, BY Application (Packaging, Textile, Transport, Agriculture, Electronics, Medical), Forecasts to 2027. 2020, September [cited 2021; https://www.emergenresearch.com/industry-report/polylactic-acid-market
Mehta R, Kumar V, Bhunia H, Upadhyay SN (2005) Synthesis of poly (lactic acid): a review. J Macromol Sci Polym Rev 45(4):325–349. https://doi.org/10.1080/15321790500304148
Ahmed J, Varshney SK (2011) Polylactides—chemistry, properties and green packaging technology: a review. Int J Food Prop 14(1):37–58. https://doi.org/10.1080/10942910903125284
Lasprilla AJ, Martinez GA, Lunelli BH, Jardini AL, Maciel Filho R (2012) Poly-lactic acid synthesis for application in biomedical devices—a review. Biotechnol Adv 30(1):321–328. https://doi.org/10.1016/j.biotechadv.2011.06.019
Van Hilst F, Hoefnagels R, Junginger M, Shen L, Wicke B (2017) Sustainable biomass for energy and materials: a greenhouse gas emission perspective. Copernicus Institute of Sustainable Development Working Paper, Utrecht
Masutani K, & Kimura Y (2014) PLA synthesis. From the monomer to the polymer https://doi.org/10.1039/9781782624806-00001
Ehsani M, Khodabakhshi K, Asgari M (2014) Lactide synthesis optimization: investigation of the temperature, catalyst and pressure effects. E-Polymers 14(5):353–361. https://doi.org/10.1515/epoly-2014-0055
Moon SI, Lee CW, Taniguchi I, Miyamoto M, Kimura Y (2001) Melt/solid polycondensation of L-lactic acid: an alternative route to poly (L-lactic acid) with high molecular weight. Polymer 42(11):5059–5062. https://doi.org/10.1016/S0032-3861(00)00889-2
Mathew AP, Oksman K, Sain M (2005) Mechanical properties of biodegradable composites from poly lactic acid (PLA) and microcrystalline cellulose (MCC). J Appl Polym Sci 97(5):2014–2025. https://doi.org/10.1002/app.21779
Nagahata R, Sano D, Suzuki H, Takeuchi K (2007) Microwave-assisted single-step synthesis of poly (lactic acid) by direct polycondensation of lactic acid. Macromol Rapid Commun 28(4):437–442. https://doi.org/10.1002/marc.200600715
Cao HL, Wang P, Yuan WB (2009) Microwave-assisted synthesis of poly (L-lactic acid) via direct melt polycondensation using solid super-acids. Macromol Chem Phys 210(23):2058–2062. https://doi.org/10.1002/macp.200900231
Zahari SSNS, Mansor MH, Azman HH, Rosli D (2020) Uncatalysed Polycondensation of Lactic Acid to Polylactic Acid under Microwave Irradiation: Effect of Microwave Power. J Phys: Conference Series 1551(1):012001). IOP Publishing. https://doi.org/10.1088/1742-6596/1551/1/012001
Li G, Zhao M, Xu F, Yang B, Li X, Meng X, Teng L, Sun F, Li Y (2020) Synthesis and biological application of polylactic acid. Molecules 25(21):5023. https://doi.org/10.3390/molecules25215023
Kim KW, Woo SI (2002) Synthesis of high-molecular-weight poly (L-lactic acid) by direct polycondensation. Macromol Chem Phys 203(15):2245–2250. https://doi.org/10.1002/1521-3935(200211)203:15<2245::AID-MACP2245>3.0.co;2-3
Hadiantoro S, Widjajanti K, Putri DIK, & Nikmah L (2021) Poly lactic acid (pla) development using lactic acid product from rice straw fermentation with azeotropic polycondensation process. In IOP Conference Series: Materials Science and Engineering (Vol. 1053, No. 1, p. 012043). IOP Publishing. https://doi.org/10.1088/1757-899x/1053/1/012043
Avérous L (2008) Polylactic acid: synthesis, properties and applications. In Monomers, polymers and composites from renewable resources (pp. 433–450). Elsevier. https://doi.org/10.1016/b978-0-08-045316-3.00021-1
Kricheldorf HR, Dunsing R (1986) Polylactones, 8. Mechanism of the cationic polymerization of L, L-dilactide. Die Makromolekulare Chemie. Macromol Chem Phys 187(7):1611–1625. https://doi.org/10.1002/macp.1986.021870706
Baśko M, Kubisa P (2010) Cationic polymerization of l, l-lactide. J Polym Sci A Polym Chem 48(12):2650–2658. https://doi.org/10.1002/pola.24048
Penczek S (1980) Cationic ring-opening polymerization of heterocyclic monomers. I Mechanisms https://doi.org/10.1007/bfb0023043
Sutar AK, Maharana T, Dutta S, Chen CT, Lin CC (2010) Ring-opening polymerization by lithium catalysts: an overview. Chem Soc Rev 39(5):1724–1746. https://doi.org/10.1039/b912806a
Uyama H, & Kobayashi S (2019) Synthesis of polyesters II: hydrolase as catalyst for ring-opening polymerization. Enzymatic polymerization towards green polymer chemistry: green chemistry and sustainable technology; Kobayashi, S., Uyama, H., Kadokawa, J., eds, 165-197. https://doi.org/10.1007/978-981-13-3813-7_6
Zhao H, Nathaniel GA, Merenini PC (2017) Enzymatic ring-opening polymerization (ROP) of lactides and lactone in ionic liquids and organic solvents: digging the controlling factors. RSC Adv 7(77):48639–48648. https://doi.org/10.1039/c7ra09038b
Whulanza Y, Rahman SF, Suyono EA, Yohda M, Gozan M (2018) Use of Candida rugosa lipase as a biocatalyst for L-lactide ring-opening polymerization and polylactic acid production. Biocatal Agric Biotechnol 16:683–691. https://doi.org/10.1016/j.bcab.2018.09.015
Bukhari A, Atta M, Idris A (2017) Synthesis of poly (L-) lactide under simultaneous cooling and microwave heating by using immobilised Candida Antarctica lipase B. Chem Eng Trans 56:1447–1452. https://doi.org/10.3303/CET1756242
Auras R, Harte B, Selke S (2004) An overview of polylactides as packaging materials. Macromol Biosci 4(9):835–864. https://doi.org/10.1002/mabi.200400043
Eerikäinen T, Linko P, Linko S, Siimes T, Zhu YH (1993) Fuzzy logic and neural network applications in food science and technology. Trends Food Sci Technol 4(8):237–242. https://doi.org/10.1016/0924-2244(93)90137-Y
Mohammadi V, & Minaei S (2019) Artificial intelligence in the production process. In Engineering tools in the beverage industry (pp. 27–63). Woodhead Publishing. https://doi.org/10.1016/B978-0-12-815258-4.00002-0
Singh A, Majumder A, Goyal A (2008) Artificial intelligence based optimization of exocellular glucansucrase production from Leuconostoc dextranicum NRRL B-1146. Bioresour Technol 99(17):8201–8206. https://doi.org/10.1016/j.biortech.2008.03.038
Ng A (2000) CS229 Lecture notes CS229 Lecture notes, 1(1), 1–3. https://backspaces.net/temp/ML/CS229.pdf
Karray F, Karray FO, De Silva CW (2004) Soft computing and intelligent systems design: theory, tools, and applications. Pearson Education
Chaturvedi DK (2008) Artificial neural network and supervised learning. Soft Computing: Techniques and its Applications in Electrical Engineering, 23-50. https://doi.org/10.1007/978-3-540-77481-5_3
Panagou EZ, Kodogiannis V, Nychas GE (2007) Modelling fungal growth using radial basis function neural networks: the case of the ascomycetous fungus Monascus ruber van Tieghem. Int J Food Microbiol 117(3):276–286. https://doi.org/10.1016/j.ijfoodmicro.2007.03.010
Ahmad A, Banat F, Taher H (2021) Comparative study of lactic acid production from date pulp waste by batch and cyclic–mode dark fermentation. Waste Manag 120:585–593. https://doi.org/10.1016/j.wasman.2020.10.029
Lunelli BH, Melo DN, de Morais ER, Victorino IR, de Toledo ECV, Maciel MRW, & Maciel Filho R (2011) Real-time optimization for lactic acid production from sucrose fermentation by Lactobacillus plantarum. In Computer aided chemical engineering (vol. 29, pp. 1396-1400). Elsevier. https://doi.org/10.1016/B978-0-444-54298-4.50058-1
Carroll DL (1996) Chemical laser modeling with genetic algorithms. AIAA J 34(2):338–346. https://doi.org/10.2514/3.13069
Dulf EH, Vodnar DC, Dulf FV (2018) Modeling tool using neural networks for l (+)-lactic acid production by pellet-form Rhizopus oryzae NRRL 395 on biodiesel crude glycerol. Chem Cent J 12(1):1–9. https://doi.org/10.1186/s13065-018-0491-5
Saleh B, Maher I, Abdelrhman Y, Heshmat M, Abdelaal O (2021) Adaptive neuro-fuzzy inference system for modelling the effect of slurry impacts on PLA material processed by FDM. Polymers 13(1):118. https://doi.org/10.3390/polym13010118
Jakkula V (2006) Tutorial on support vector machine (svm). School of EECS, Washington State University, 37. https://course.ccs.neu.edu/cs5100f11/resources/jakkula.pdf
Segler MH, Preuss M, Waller MP (2018) Planning chemical syntheses with deep neural networks and symbolic AI. Nature 555(7698):604–610. https://doi.org/10.1038/nature25978
Singhvi MS, Zinjarde SS, Gokhale DV (2019) Polylactic acid: synthesis and biomedical applications. J Appl Microbiol 127(6):1612–1626. https://doi.org/10.1111/jam.14290
Yanoso-Scholl L, Jacobson JA, Bradica G, Lerner AL, O'Keefe RJ, Schwarz EM, Zuscik MJ, Awad HA (2010) Evaluation of dense polylactic acid/beta-tricalcium phosphate scaffolds for bone tissue engineering. J Biomed Mater Res A 95(3):717–726. https://doi.org/10.1002/jbm.a.32868
Haaparanta AM, Järvinen E, Cengiz IF, Ellä V, Kokkonen HT, Kiviranta I, Kellomäki M (2014) Preparation and characterization of collagen/PLA, chitosan/PLA, and collagen/chitosan/PLA hybrid scaffolds for cartilage tissue engineering. J Mater Sci Mater Med 25(4):1129–1136. https://doi.org/10.1007/s10856-013-5129-5
Gupta S, Sharma A, Kumar JV, Sharma V, Gupta PK, Verma RS (2020) Meniscal tissue engineering via 3D printed PLA monolith with carbohydrate based self-healing interpenetrating network hydrogel. Int J Biol Macromol 162:1358–1371. https://doi.org/10.1016/j.ijbiomac.2020.07.238
Mohandesnezhad S, Pilehvar-Soltanahmadi Y, Alizadeh E, Goodarzi A, Davaran S, Khatamian M, Zarghami N, Samiei M, Aghazadeh M, Akbarzadeh A (2020) In vitro evaluation of Zeolite-nHA blended PCL/PLA nanofibers for dental tissue engineering. Mater Chem Phys 252:123152. https://doi.org/10.1016/j.matchemphys.2020.123152
Dong Y, Feng SS (2006) Nanoparticles of poly (D, L-lactide)/methoxy poly (ethylene glycol)-poly (D, L-lactide) blends for controlled release of paclitaxel. J Biomed Mater Res Part A 78(1):12–19. https://doi.org/10.1002/jbm.a.30684
Siafaka PI, Barmbalexis P, Bikiaris DN (2016) Novel electrospun nanofibrous matrices prepared from poly (lactic acid)/poly (butylene adipate) blends for controlled release formulations of an anti-rheumatoid agent. Eur J Pharm Sci 88:12–25. https://doi.org/10.1016/j.ejps.2016.03.021
Yu Y, Chen CK, Law WC, Weinheimer E, Sengupta S, Prasad PN, Cheng C (2014) Polylactide-graft-doxorubicin nanoparticles with precisely controlled drug loading for pH-triggered drug delivery. Biomacromolecules 15(2):524–532. https://doi.org/10.1021/bm401471p
Mhlanga N, Sinha Ray S, Lemmer Y, Wesley-Smith J (2015) Polylactide-based magnetic spheres as efficient carriers for anticancer drug delivery. ACS Appl Mater Interfaces 7(40):22692–22701. https://doi.org/10.1021/acsami.5b07567
Li W, Fan X, Wang X, Shang X, Wang Q, Lin J, Hu Z, Li Z (2018) Stereocomplexed micelle formation through enantiomeric PLA-based Y-shaped copolymer for targeted drug delivery. Mater Sci Eng C 91:688–695. https://doi.org/10.1016/j.msec.2018.06.006
Chen J, Tian B, Yin X, Zhang Y, Hu D, Hu Z, Liu M, Pan Y, Zhao J, Li H, Hou C, Wang J, Zhang Y (2007) Preparation, characterization and transfection efficiency of cationic PEGylated PLA nanoparticles as gene delivery systems. J Biotechnol 130(2):107–113. https://doi.org/10.1016/j.jbiotec.2007.02.007
Camović M, Biščević A, Brčić I, Borčak K, Bušatlić S, Ćenanović N, Dedović A, Mulalić A, Osmanlić M, Sirbubalo M, Tucak A, & Vranić E (2019) Coated 3d printed PLA microneedles as transdermal drug delivery systems. In International conference on medical and biological engineering (pp. 735–742). Springer, Cham. https://doi.org/10.1007/978-3-030-17971-7_109
Vaňková E, Kašparová P, Khun J, Machková A, Julák J, Sláma M, Hodek J, Ulrychová L, Weber J, Obrová K, Kosulin K, Lion T, Scholtz V (2020) Polylactic acid as a suitable material for 3D printing of protective masks in times of COVID-19 pandemic. PeerJ 8:e10259. https://doi.org/10.7717/peerj.10259
Buluş E, Buluş GS, Yakuphanoglu F (2020) Production of polylactic acid-activated charcoal nanofiber membranes for COVID-19 pandemic by electrospinning technique and determination of filtration efficiency. J Mater Electronic Devices 4(1):21–26
Alghounaim M, Almazeedi S, Al Youha S, Papenburg J, Alowaish O, AbdulHussain G, Rawan Al-Shemali R, Albuloushi A, Alzabin S, Al-Wogayan K, Al-Mutawa Y, Al-Sabah S (2020) Low-cost polyester-tipped three-dimensionally printed nasopharyngeal swab for the detection of severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2). J Clin Microbiol 58(11):e01668–20. https://doi.org/10.1128/jcm.01668-20
Holm VK, Mortensen G, Vishart M, Petersen MA (2006) Impact of poly-lactic acid packaging material on semi-hard cheese. Int Dairy J 16(8):931–939. https://doi.org/10.1016/j.idairyj.2005.09.001
Lukic I, Vulic J, Ivanovic J (2020) Antioxidant activity of PLA/PCL films loaded with thymol and/or carvacrol using scCO2 for active food packaging. Food Packag Shelf Life 26:100578. https://doi.org/10.1016/j.fpsl.2020.100578
Moustafa H, El Kissi N, Abou-Kandil AI, Abdel-Aziz MS, Dufresne A (2017) PLA/PBAT bionanocomposites with antimicrobial natural rosin for green packaging. ACS Appl Mater Interfaces 9(23):20132–20141. https://doi.org/10.1021/acsami.7b05557
Knoch S, Pelletier F, Larose M, Chouinard G, Dumont MJ, Tavares JR (2020) Surface modification of PLA nets intended for agricultural applications. Colloids Surf A Physicochem Eng Asp 598:124787. https://doi.org/10.1016/j.colsurfa.2020.124787
Khan H, Kaur S, Baldwin TC, Radecka I, Jiang G, Bretz I, Duale K, Adamus G, & Kowalczuk M (2020) Effective control against broadleaf weed species provided by biodegradable PBAT/PLA mulch film embedded with the herbicide 2-methyl-4-chlorophenoxyacetic acid (MCPA). https://doi.org/10.1021/acssuschemeng.0c00991
Souza PMS, Sommaggio LRD, Marin-Morales MA, Morales AR (2020) PBAT biodegradable mulch films: study of ecotoxicological impacts using Allium cepa, Lactuca sativa and HepG2/C3A cell culture. Chemosphere 256:126985. https://doi.org/10.1016/j.chemosphere.2020.126985
Jabbar A, Tausif M, Tahir HR, Basit A, Bhatti MRA, Abbas G (2019) Polylactic acid/lyocell fibre as an eco-friendly alternative to polyethylene terephthalate/cotton fibre blended yarns and knitted fabrics. J Text Inst 111(1):129–138. https://doi.org/10.1080/00405000.2019.1624070
Huang XX, Tao XM, Zhang ZH, Chen P (2016) Properties and performances of fabrics made from bio-based and degradable polylactide acid/poly (hydroxybutyrate-co-hydroxyvalerate)(PLA/PHBV) filament yarns. Text Res J 87(20):2464–2474. https://doi.org/10.1177/0040517516671128
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Das, M. et al. (2023). Lactic Acid Production from Fungal Machineries and Mechanism of PLA Synthesis: Application of AI-Based Technology for Improved Productivity. In: Satyanarayana, T., Deshmukh, S.K. (eds) Fungi and Fungal Products in Human Welfare and Biotechnology. Springer, Singapore. https://doi.org/10.1007/978-981-19-8853-0_8
Download citation
DOI: https://doi.org/10.1007/978-981-19-8853-0_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-8852-3
Online ISBN: 978-981-19-8853-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)