Abstract
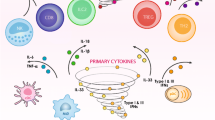
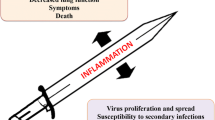
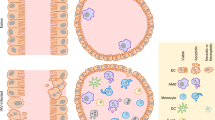
Influenza A virus is one of the most common infectious pathogen and associated with significant morbidity and mortality. Infected patients with underlying diseases show rapid progression in disease severity. The initial pathogenic process of influenza virus infection is characterized by the induction of various proinflammatory cytokines as well as host cellular trypsin-type viral envelope-processing proteases in the airway, which enhance viral multiplication. This process has been termed the “influenza virus−cytokine−trypsin” cycle. In the advanced stage of infection, the cytokine storm induces disorders of glucose and lipid metabolism in the mitochondria, resulting in ATP crisis and various functional disorders particularly in organs and cells with high ATP consumption, such as vascular endothelial cells and cardiomyocytes. This process has been termed interconnection of the “metabolic disorders−cytokine” cycle with the “influenza virus−cytokine−trypsin” cycle. The interconnection exacerbates mitochondrial ATP crisis and could lead to multiple organ failure with severe edema. Breaking these cycles and interconnection is a promising therapeutic approach against severe influenza. In this review, we discuss the pathogenesis of severe influenza viral infection based on animal experiments and the potential therapeutic options.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lipatov AS, Govorkova EA, Webby RJ, Ozaki H, Peiris M, Guan Y, et al. Influenza: emergence and control. J Virol. 2004;78:8951–9.
Kim HM, Brandt CD, Arrobio JO, Murphy B, Chanock RM, Parrott RH. Influenza A and B virus infection in infants and young children during the years 1957-1976. Am J Epidemiol. 1979;109:464–79.
Chowell G, Ayala A, Berisha V. Viboud C, Schumacher M. Risk factors for mortality among 2009 A/H1N1 influenza hospitalizations in Maricopa county, Arizona, April 2009 to March 2010. Comput Math Methods Med 2012;2012:914196.
Spraque AH, Khalil RA. Inflammatory cytokines in vascular dysfunction and vascular disease. Biochem Pharmacol. 2009;78:539–52.
Mariappan N, Elks CM, Fink B, Francis J. TNF-induced mitochondrial damage: a link between mitochondrial complex I activity and left ventricular dysfunction. Free Radic Biol Med. 2009;46:462–70.
Kido H. Influenza virus pathogenicity regulated by host cellular proteases, cytokines and metabolites, and its therapeutic options. Proc Jpn Acad Ser B Phys Biol Sci. 2015;91:351–68.
Kido H. Energy metabolic disorder is a major risk factor in severe influenza virus infection: proposals for new therapeutic options based on animal model experiments. Respir Investig. 2016;54:312–9.
Klenk H-D, Rott R, Orlich M, Blödom J. Activation of influenza A viruses by trypsin treatment. Virology. 1975;68:426–39.
Kido H, Takahashi E, Kimoto T. Role of host trypsin-type serine proteases and influenza virus–cytokine–trypsin cycle in influenza viral pathogenesis. Pathogenesis-based therapeutic options. Biochimie. 2019;166:203–13.
Wang S, Le TQ, Kurihara N, Chida J, Cisse Y, Yano M, et al. Influenza virus–cytokine–protease cycle in the pathogenesis of vascular hyperpermeability in severe influenza. J Infect Dis. 2010;202:991–1001.
Pan HY, Yamada H, Chida J, Wang S, Yano M, Yao M, et al. Up-regulation of ectopic trypsins in the myocardium by influenza A virus infection triggers acute myocarditis. Cardiovasc Res. 2011;89:595–603.
Indalao IL, Sawabuchi T, Takahashi E, Kido H. IL-1β is a key cytokine that induces trypsin upregulation in the influenza virus−cytokine−trypsin cycle. Arch Virol. 2017;162:201–11.
Mizuguchi M, Yamanouchi H, Ichiyama T, Shiomi M. Acute encephalopathy associated with influenza and other viral infection. Acta Neurol Scand Suppl. 2007;186:45–6.
Denker BM, Nigam SK. Molecular structure and assembly of the tight junction. Am J Phys. 1998;274:F1–9.
Palmer ML, Lee SY, Maniak PJ, Carlson D, Fahrenkrug SC, O’Grady SM. Protease-activated receptor regulation of Cl- secretion in Calu-3 cells requires prostaglandin release and CFTR activation. Am J Physiol Cell Physiol. 2006;290:C1189–98.
Yamane K, Indalao IL, Chida J, Yamamoto Y, Hanawa M, Kido H. Diisopropylamine dichloroacetate, a novel pyruvate dehydrogenase kinase 4 inhibitor, as a potential therapeutic agent for metabolic disorders and multiorgan failure in severe influenza. PLoS One. 2014;9:e98032.
Bersin RM, Stacpoole PW. Dichloroacetate as metabolic therapy for myocardial ischemia and failure. Am Heart J. 1997;134:841055.
Chen Y, Mizuguchi H, Yao D, Ide M, Kuroda Y, Shigematsu Y, et al. Thermolabile phenotype of carnitine palmitoyltransferase II variations as a predisposing factor for influenza-associated encephalopathy. FEBS Lett. 2005;579:2040–4.
Yao D, Mizuguchi H, Yamaguchi M, Yamada H, Chida J, Shikata K, et al. Thermal instability of compound variants of carnitine palmitoyltransferase II and impaired mitochondrial fuel utilization in influenza-associated encephalopathy. Hum Mutat. 2008;29:718–27.
Bonnefont JP, Bastin J, Laforêt P, Aubey F, Mogenet A, Romano S, et al. Long-term follow-up of bezafibrate treatment in patients with the myopathic form of carnitine palmitoyltransferase 2 deficiency. Clin Pharmacol Ther. 2010;88:101–8.
Yao M, Yao D, Yamaguchi M, Chida J, Yao D, Kido H. Bezafibrate upregulates carnitine palmitoyltransferase II expression and promotes mitochondrial energy crisis dissipation in fibroblasts of patients with influenza-associated encephalopathy. Mol Genet Metab. 2011;104:265–72.
Acknowledgments
Our studies were supported in part by Grants-in-Aid (24249059) from the Ministry of Education, Culture, Sports, Science and Technology of Japan; by Health and Labor Sciences Research Grants (Grant #12103307) from the Ministry of Health, Labor and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kido, H., Kimoto, T., Takahashi, E. (2021). Cellular and Biochemical Pathogenic Processes in Severe Influenza Virus Infection: How Does Cytokine Storm Play a Role?. In: Fujita, J. (eds) Influenza. Respiratory Disease Series: Diagnostic Tools and Disease Managements. Springer, Singapore. https://doi.org/10.1007/978-981-15-9109-9_4
Download citation
DOI: https://doi.org/10.1007/978-981-15-9109-9_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-9108-2
Online ISBN: 978-981-15-9109-9
eBook Packages: MedicineMedicine (R0)