Abstract
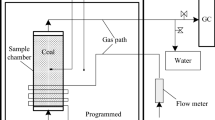
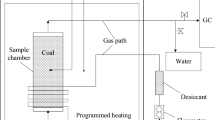
To investigate the effect on coal spontaneous combustion due to high geo-temperature, an experimental apparatus was developed to measure parameters for two kinds of coal samples under different temperatures: one was pretreated at constant temperature of 40 °C (G coal sample), and the other was a rising temperature from room temperature (20 °C, Sample C). Based on the relationship between CO concentration and the temperature variation, a calculation model of the apparent activation energy of coal was established. The results indicated that the oxygen consumption rate, generation rates of CO and CO2, and heat release intensity of Sample G were higher than C coal sample. This trend was more and more obvious as the temperature is increased. Furthermore, the apparent activation energy of G coal sample was smaller than sample C, especially at low temperatures, which demonstrated that the oxidation ability and reactivity of sample G was enhanced, and less energy was required to break the barrier of oxidation reaction. Under the same condition, the speed of oxidation reaction was faster for the coal in the high geo-temperature environment, which was more susceptible to spontaneous combustion.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Stracher, G.B.: Coal fires burning around the world: a global catastrophe. Int. J. Coal Geol. 59(1–2), 1–6 (2004)
Xie, J., Xue, S., Cheng, W., Wang, G.: Early detection of spontaneous combustion of coal in underground coal mines with development of an ethylene enriching system. Int. J. Coal Geol. 85(1), 123–127 (2011)
Xiao, Y., Li, Q., Deng, J., Shu, C., Wang, W.: Experimental study on the corresponding relationship between the index gases and critical temperature for coal spontaneous combustion. J. Therm. Anal. Calorim. 127(1), 1009–1017 (2017)
Xie, H.P., Zhou, H.W., Xue, D.J., Wang, H.W., Zhang, R., Gao, F.: Research and consideration on deep coal mining and critical mining depth. J. China Coal Soc. 37(37), 535–542 (2012)
Chen, W., Liang, S., Liu, J.: Proposed split-type vapor compression refrigerator for heat hazard control in deep mines. Appl. Therm. Eng. 105, 425–435 (2016)
He, M.: Application of HEMS cooling technology in deep mine heat hazard control. Min. Sci. Technol. 19(3), 269–275 (2009)
Qin, B., Sun, Q., Wang, D., Zhang, L., Xu, Q.: Analysis and key control technologies to prevent spontaneous coal combustion occurring at a fully mechanized caving face with large obliquity in deep mines. Min. Sci. Technol. 19(4), 446–451 (2009)
He, M., Cao, X., Xie, Q., Yang, J., Qi, P., Yang, Q., et al.: Principles and technology for stepwise utilization of resources for mitigating deep mine heat hazards. Min. Sci. Technol. 20(1), 20–27 (2010)
Guo, P., Qin, F.: Preventive measures against heat hazard and its utilization in Zhangshuanglou Coal Mine. J. China Coal Soc. 38(S2), 393–398 (2013)
Guo, P., He, M., Zheng, L., Zhang, N.: A geothermal recycling system for cooling and heating in deep mines. Appl. Therm. Eng. 116, 833–839 (2017)
Sasmito, A.P., Kurnia, J.C., Birgersson, E., Mujumdar, A.S.: Computational evaluation of thermal management strategies in an underground mine. Appl. Therm. Eng. 90, 1144–1150 (2015)
Millar, D., Trapani, K., Romero, A.: Deep mine cooling, a case for Northern Ontario: Part I. Int. J. Min. Sci. Technol. 26(4), 721–727 (2016)
Trapani, K., Romero, A., Millar, D.: Deep mine cooling, a case for Northern Ontario: Part II. Int. J. Min. Sci. Technol. 26(6), 1033–1042 (2016)
Arisoy, A., Beamish, B.: Reaction kinetics of coal oxidation at low temperatures. Fuel 159, 412–417 (2015)
Li, B., Chen, G., Zhang, H., Sheng, C.: Development of non-isothermal TGA–DSC for kinetics analysis of low temperature coal oxidation prior to ignition. Fuel 118(8), 385–391 (2014)
Qi, G., Wang, D., Zheng, K., Xu, J., Qi, X., Zhong, X.: Kinetics characteristics of coal low-temperature oxidation in oxygen-depleted air. J. Loss Prev. Process Ind. 35, 224–231 (2015)
Wang, D., Xin, H., Qi, X., Dou, G., Qi, G., Ma, L.: Reaction pathway of coal oxidation at low temperatures: a model of cyclic chain reactions and kinetic characteristics. Combust. Flame 163, 447–460 (2016)
Zhang, Y., Wu, J., Chang, L., Wang, J., Xue, S., Li, Z.: Kinetic and thermodynamic studies on the mechanism of low-temperature oxidation of coal: A case study of Shendong coal (China). Int. J. Coal Geol. 120, 41–49 (2013)
Ozbas, K.E., Kök, M.V., Hicyilmaz, C.: Comparative kinetic analysis of raw and cleaned coals. J. Therm. Anal. Calorim. 69(2), 541–549 (2002)
Deng, J., Li, Q., Xiao, Y., Wen, H.: The effect of oxygen concentration on the non-isothermal combustion of coal. Thermochim. Acta 653, 106–115 (2017)
Yu, L.Y., Li, P.S.: Thermogravimetric analysis of coal and sludge co-combustion with microwave radiation dehydration. J. Energy Inst. 87(3), 220–226 (2014)
Chen, G., Ma, X., Lin, M., Lin, Y., Yu, Z.: Study on thermochemical kinetic characteristics and interaction during low temperature oxidation of blended coals. J. Energy Inst. 88(3), 221–228 (2015)
Xu, J.: Determination theory of coal spontaneous combustion zone. China Coal Industry Publishing House, Beijing (PR China) (2001)
Deng, J., Zhao, J., Zhang, Y., Huang, A., Liu, X., Zhai, X., et al.: Thermal analysis of spontaneous combustion behavior of partially oxidized coal. Process Saf. Environ. Prot. 104, 218–224 (2016)
Deng, J., Xiao, Y., Li, Q., Lu, J., Wen, H.: Experimental studies of spontaneous combustion and anaerobic cooling of coal. Fuel 157, 261–269 (2015)
Wang, C., Yang, Y., Tsai, Y., Deng, J., Shu, C.: Spontaneous combustion in six types of coal by using the simultaneous thermal analysis-Fourier transform infrared spectroscopy technique. J. Therm. Anal. Calorim. 126(3), 1591–1602 (2016)
Baris, K., Kizgut, S., Didari, V.: Low-temperature oxidation of some Turkish coals. Fuel 93, 423–432 (2012)
Wang, H., Dlugogorski, B. Z., Kennedy, E. M.: Coal oxidation at low temperatures: oxygen consumption, oxidation products, reaction mechanism and kinetic modelling. Prog. Energ. Combust. Sci. 29(6), 487–513 (2003)
Tang, Y., Xue, S.: Laboratory study on the spontaneous combustion propensity of lignite undergone heating treatment at low temperature in inert and low-oxygen environments. Energy Fuels 29(8), 4683–4689 (2015)
Su, H., Zhou, F., Li, J., Qi, H.: Effects of oxygen supply on low-temperature oxidation of coal: a case study of Jurassic coal in Yima. China. Fuel 202, 446–454 (2017)
Zhang, Y., Wang, J., Wu, J., Xue, S., Li, Z., Chang, L.: Modes and kinetics of CO2 and CO production from low-temperature oxidation of coal. Int. J. Coal Geol. 140, 1–8 (2015)
Zhou, C., Zhang, Y., Wang, J., Xue, S., Wu, J., Chang, L.: Study on the relationship between microscopic functional group and coal mass changes during low-temperature oxidation of coal. Int. J. Coal Geol. 171, 212–222 (2017)
Song, Z., Kuenzer, C.: Coal fires in China over the last decade: A comprehensive review. Int. J. Coal Geol. 133, 72–99 (2014)
Tang, Y.: Sources of underground CO: Crushing and ambient temperature oxidation of coal. J. Loss Prev. Process Ind. 38, 50–57 (2015)
Acknowledgements
We thank for the financial support of National Natural Science Foundation Funded Project (Grant No.: 51504186, 51574193), Industrial Technology Research of Shaanxi Province Government (Grant No.: 2016GY–191).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Science Press and Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Deng, J., Lei, C., Xiao, Y., Ma, L., Wang, K., Shu, C. (2019). The Effect of High Geo-Temperature Environment on Coal Spontaneous Combustion: An Experimental Study. In: Chang, X. (eds) Proceedings of the 11th International Mine Ventilation Congress. Springer, Singapore. https://doi.org/10.1007/978-981-13-1420-9_46
Download citation
DOI: https://doi.org/10.1007/978-981-13-1420-9_46
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-1419-3
Online ISBN: 978-981-13-1420-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)