Abstract
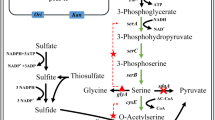
l-cysteine, a sulfur-containing amino acid with important physiological function, is usually extracted from hydrolyzed protein. In this study, we focused on a biocatalytic process with Escherichia coli whole-cell biocatalyst for the production of l-cysteine. The genes encoding l-2-amino-Δ2-thiazoline-4-carboxylic acid (l-ATC) hydrolase (atcB) and N-carbamoyl-l-cysteine (l-NCC) amidohydrolase (atcC) were synthesized and expressed in E. coli BL21. The recombinant E. coli strain was used as the whole-cell biocatalyst to convert dl-ATC, and 13.1 mM of l-cysteine was accumulated. Deletion of the l-cysteine desulfhydrase gene tnaA in E. coli BL21 resulted in a further 54.4% increase of l-cysteine production. The optimal pH and temperature of l-cysteine production were 7.0 and 37 °C, respectively. In addition, we also explored the effects of glycerol concentration on the accumulation of l-cysteine. It shows that the optimal glycerol concentration was 10%. Finally, 70.2 mM of l-cysteine was accumulated at 8 h under optimum conditions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kuśmierek K, Bald E (2008) Reduced and total glutathione and cysteine profiles of citrus fruit juices using liquid chromatography. Food Chem 106(1):340–344
Ismail NI, Hashim YZ, Jamal P et al (2014) Production of cysteine: approaches, challenges and potential Solution. Inter J Biotechfor Wellness Ind 3:95–101
Duan J, Zhang Q, Zhao H et al (2012) Cloning, expression, characterization and application of atcA, atcB and atcC from Pseudomonas sp. for the production of l-cysteine. Biotech Lett 34(6):1101–1106
Leuchtenberger W, Huthmacher K, Drauz K (2005) Biotechnological production of amino acids and derivatives: current status and prospects. Appl Microbiol Biot 69(1):1–8
Chen N, Huang J, Feng ZB et al (2009) Optimization of fermentation conditions for the biosynthesis of l-threonine by Escherichia coli. Appl Biochem Biotechnol 158(3):595–604
Park JH, Jang YS, Lee JW et al (2011) Escherichia coli W as a new platform strain for the enhanced production of l-valine by systems metabolic engineering. Biotechnol Bioeng 108(5):1140–1147
Grant GA, Hu Z, Xu XL (2001) Specific interactions at the regulatory domain-substrate binding domain interface influence the cooperativity of inhibition and effector binding in Escherichia coli D-3-phosphoglycerate dehydrogenase. J Biol Chem 276(2):1078–1083
Nakatani T, Ohtsu I, Nonaka G et al (2012) Enhancement of thioredoxin/glutaredoxin-mediated l-cysteine synthesis from S-sulfocysteine increases l-cysteine production in Escherichia coli. Microb Cell Fact 11(1):62
Park S, Imlay JA (2003) High levels of intracellular cysteine promote oxidative DNA damage by driving the fenton reaction. J Bacteriol 185(6):1942–1950
Sørensen MA, Pedersen S (1991) Cysteine, even in low concentrations, induces transient amino acid starvation in Escherichia coli. J Bacteriol 173(16):5244–5246
Sand K, Eguchi C, Yasuda N et al (1979) Metabolic pathway of l-cysteine formation from dl-2-amino-Δ2-thiazoline-4-carboxylic acid by Pseudomonas. J Gen Appl Microbiol 43(11):2373–2374
Yu Y, Liu Z, Liu C et al (2006) Cloning, expression, and identification of genes involved in the conversion of dl-2-Amino-Δ2-thiazoline-4-carboxylic acid to l-cysteine via S-Carbamyl-l-cysteine pathway in Pseudomonas sp. TS1138. Biosci Biotechnol Biochem 70(9):2262–2267
Tamura Y, Ohmachi T, Asada Y (2001) Induction of 2-amino-Δ2-thiazoline-4-carboxylic acid hydrolase and N-carbamoyl-l-cysteine amidohydrolase by S-compounds in Pseudomonas putida AJ3865. J Gen Appl Microbiol 47(4):193–200
Tamura Y, Nishino M, Ohmachi T et al (1998) N-carbamoyl-l-cysteine as an intermediate in the bioconversion from dl-2-Amino-Δ2-thiazoline-4-carboxylic acid to l-cysteine by Pseudomonas sp. ON-4a. Biosci Biotechnol Biochem 62(11):2226–2229
Huai L, Chen N, Yang W et al (2009) Metabolic control analysis of l-cysteine producing strain TS1138 of Pseudomonas sp. Biochemistry (Mosc) 74(3):288–292
Youn SH, Park HW. Shin CS (2012) Enhanced dissolution of the substrate dl-2-amino-Δ2-thiazoline-4-carboxylic acid and enzymatic production of l-cysteine at high concentrations. Eng Life Sci. 12:514–517
Gu X, Li C, Cai Y, Dong H et al (2013) Construction of lactococcus lactis thyA-null using the Red recombination system. Ann Microbiol 63(3):951–956
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97(12):6640–6645
Awano N, Wada M, Kohdoh T et al (2003) Effect of cysteine desulfhydrase gene disruption on l-cysteine overproduction in Escherichia coli. Appl Microbiol Biotechnol 62(2):239–243
Dwivedi CM, Ragin RC, Uren JR (1982) Cloning, purification, and characterization of beta-cystathionase from Escherichia coli. Biochemistry 21(13):3064–3069
Awano N, Wada M, Mori H et al (2005) Identification and functional analysis of Escherichia coli cysteine desulfhydrases. Appl Environ Microbiol 71(7):4149–4152
Read JF, Bewick S, Graves CR et al (2000) The kinetics and mechanism of the oxidation of s-methyl-l-cysteine, l-cystine and l-cysteine by potassium ferrate. Inorg Chim Acta 303(2):244–255
Sano K, Mitsugi K (1978) Enzymatic production of l-cysteine from dl-2-amino-Δ2-thiazoline-4-carboxylic acid by Pseudomonas thiazolinophilum: optimal conditions for the enzyme formation and enzymatic reaction. Agric Biol Chem 42(12):2315–2321
Youn SH, Park HW, Choe D et al (2014) Preparation of eutectic substrate mixtures for enzymatic conversion of ATC to l-cysteine at high concentration levels. Bioprocess Biosyst Eng 37(6):1193–1200
Kumar A, Pawar SS (2012) High viscosity of ionic liquids causes rate retardation of Diels-Alder reactions. Sci China Chem 55(8):1633–1637
Wang P, He JY, Yin JF (2015) Enhanced biocatalytic production of l-cysteine by Pseudomonas sp. B-3 with in situ product removal using ion-exchange resin. Bioprocess Biosyst Eng 38(3):421–428
Wada M, Takagi H (2006) Metabolic pathways and biotechnological production of l-cysteine. Appl Microbiol Biotechnol 73(1):48–54
Acknowledgements
This work was supported by Tianjin Science and Technology Support Program (NO. 16YFZCSY00770).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Ma, M., Liu, T., Wu, H., Yan, F., Chen, N., Xie, X. (2018). Enzymatic Synthesis of l-Cysteine by Escherichia coli Whole-Cell Biocatalyst. In: Liu, H., Song, C., Ram, A. (eds) Advances in Applied Biotechnology. ICAB 2016. Lecture Notes in Electrical Engineering, vol 444. Springer, Singapore. https://doi.org/10.1007/978-981-10-4801-2_48
Download citation
DOI: https://doi.org/10.1007/978-981-10-4801-2_48
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-4800-5
Online ISBN: 978-981-10-4801-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)