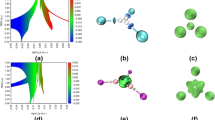
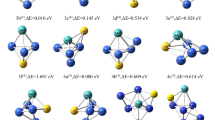
Abstract
Usefulness of Molecular Orbital analysis for prediction and interpretation of a variety of high-pressure phenomena will be illustrated for the case of elusive gold monofluoride, Au(I)F.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Evans, C.J., and Gerry, M.C.L., 2000, Confirmation of the existence of gold(I) fluoride, AuF: Microwave spectrum and structure. J. Am. Chem. Soc. 122: 1560.
Gažo, J., Bersuker, I.B., Garaj, J., Kabešová, M., Kohout, J., Langfelderová, H., Melník, M., Serátor, M., and Valach, F., 1976, Plasticity of the coordination sphere of copper(II) complexes, its manifestation and causes. Coord. Chem. Rev. 19: 253.
Gillespie, R. and Nyholm, R. S. 1957, Inorganic stereochemistry. Quart. Rev. 11: 339.
Grochala, W., 2007, Atypical compounds of gases, which have been called ‘noble’.Chem. Soc. Rev. 36: 1632.
Grochala, W., 2009, On chemical bonding between helium and oxygen. Pol. J. Chem. 83: 87.
Grochala, W., Feng, J., Hoffmann, R., and Ashcroft, N.W., 2007, The chemical imagination at work in very tight places. Angew. Chem. Int. Ed. Engl. 46: 3620.
Hoffmann, R., 1971, Interaction of orbitals through space and through bonds. Acc. Chem. Res. 4: 1.
Hoffmann, R., 1988, Solids and Surfaces: A Chemist’s View of Bonding in Extended Structures, VCH, New York.
Hoffmann, R., 1998, Qualitative thinking in the age of modern computational chemistry – or what Lionel Salem knows. J. Mol. Str. (Theochem) 424: 1.
Kurzydłowski, D., and Grochala, W., 2008a, Elusive AuF in the solid state as accessed via high-pressure comproportionation. Chem. Commun. 1073.
Kurzydłowski, D., and Grochala, W., 2008b, Xenon as a mediator of chemical reactions? Case of scarce gold monofluoride, AuF, and its adduct with xenon.Z. Anorg. Allg. Chem. 634: 1082.
Kutzelnigg, W., 1996, Friedrich Hund and chemistry. Angew. Chem. 35: 573.
Landrum, G.A., and Dronskowski, R., 2000, The orbital origins of magnetism: from atoms to molecules to ferromagnetic alloys. Angew. Chem. Int. Ed. Engl. 39: 1560.
Landrum, G.A., and Hoffmann, R., 1998, Secondary bonding between chalcogens or pnicogens and halogens. Angew. Chem. Int. Ed. Engl. 37: 1887.
Lennard-Jones, J.E., 1929, The electronic structure of some diatomic molecules. Trans. Faraday Soc. 25: 668.
Mohr, F., 2004, The chemistry of gold-fluoro compounds: A continuing challenge for gold chemists.Gold Bull. 37: 164.
Mulliken, R.S., 1934, A new electroaffinity scale; together with data on valence states and on valence ionization potentials and electron affinities. J. Chem. Phys. 2: 782.
Mulliken, R.S., 1967, Spectroscopy, molecular orbitals, and chemical bonding. Science. 3785: 157.
Pearson, R.G., 1985, Absolute electronegativity and absolute hardness of Lewis acids and bases. J. Am. Chem. Soc. 107: 6081.
Piela, L., 2007, Ideas of quantum chemistry, Elsevier, Amsterdam.
Pyykkü, P., 1995, Predicted chemical bonds between rare gases and Au+ . J. Am. Chem. Soc. 117: 2067.
Pyykkü, P., 2004, Theoretical chemistry of gold. Angew. Chem. Int. Ed. Engl. 43, 4412.
Saenger, K.L., and Sun, C.P., 1992, Yellow emission bands produced during gold etching in O2-CF4 rf glow-discharge plasmas: Evidence for gas-phase AuF. Phys. Rev. A 46: 670.
Schrüder, D., Hrušak, J., Tornieporth–Oetting, I.C., Klapoetke T.M., and Schwarz, H., 1994, Neutral gold(I) fluoride does indeed exist. Angew. Chem., Int. Ed. Engl. 33: 212.
Schwerdtfeger, P., McFeaters, J.S., Stephens, R.L., Liddell, M.J., Dolg, M., Hess, B.A. 1994, Can AuF be synthesized? A theoretical study using relativistic configuration interaction and plasma modeling techniques. Chem. Phys. Lett. 218: 362.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer Science+Business Media B.V.
About this paper
Cite this paper
Grochala, W., KurzydŁowski, D. (2010). Molecular Orbital Approach to Interpret High Pressure Phenomena – Case of Elusive Gold Monofluoride. In: Boldyreva, E., Dera, P. (eds) High-Pressure Crystallography. NATO Science for Peace and Security Series B: Physics and Biophysics. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-9258-8_29
Download citation
DOI: https://doi.org/10.1007/978-90-481-9258-8_29
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-481-9257-1
Online ISBN: 978-90-481-9258-8
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)