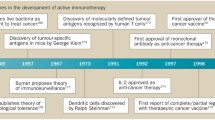
Abstract
Microbial infection is usually accompanied by inflammation and tissue wounding. These local symptoms are dependent on a host’s immune response rather than the direct cell damage by invading microbes. A microbe usually contains pattern molecules that enhance the host immune response. Clinical tests medicating cancer patients with this microbial component (namely adjuvant) were tried prior to the twentieth century, when the outline of the immune system had not yet been unveiled molecularly. These tests showed effect in some patients, though accompanied by side effects. The response to adjuvants was also useful for understanding the host–defense mechanism, later elucidated as innate immunity. Adjuvant is a general term for molecules that imitate a host immune-stimulator of microbes that activates pattern recognition receptors in the innate system to enhance interferons/cytokines and activation of the cellular immune system. This chapter outlines the history of adjuvant and the updated understanding by which an adjuvant is rationally introduced into anti-tumor immunity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AP1:
-
Activator protein-1
- BCG:
-
Bacillus Calmette-Guérin (a live attenuated strain of Mycobacterium bovis)
- BCG-CWS:
-
BCG–cell wall skeleton
- CTL:
-
Cytotoxic T lymphocyte
- DC:
-
Dendritic cell
- dsRNA:
-
Double-stranded RNA
- GMP:
-
Good Manufacturing Practice
- IFN:
-
Interferon
- IFNAR1:
-
Interferon (α and β) receptor 1
- IL:
-
Interleukin
- INAM:
-
IRF3-dependent natural killer-activating molecule
- IRF:
-
Interferon regulatory factor
- MAPK:
-
Mitogen-activated protein kinase
- MDA5:
-
Melanoma differentiation-associated protein-5
- MDP:
-
Muramyl dipeptide
- NK:
-
Natural killer
- Pam2CS:
-
S-[2,3-bis(palmitoyloxy)-(2RS)-propyl]-R-cysteinyl-S-serine
- PAMP:
-
Pathogen-associated molecular pattern
- PGN:
-
Peptidoglycan, polyI:C, polyinosinic–polycytidylic acid
- RIG-I:
-
Retinoic acid-inducible gene-I
- TAA:
-
Tumor-associated antigen
- TDM:
-
Trahalose dimycolate
- TICAM-1:
-
Toll–IL-1 receptor domain-containing adaptor molecule 1
- TLR:
-
Toll-like receptor
References
Akazawa T, Ebihara T, Okuno M, Okuda Y, Shingai M, Tsujimura K, Takahashi T, Ikawa M, Okabe M, Inoue N, Okamoto-Tanaka M, Ishizaki H, Miyoshi J, Matsumoto M, Seya T (2007) Antitumor NK activation induced by the Toll-like receptor 3-TICAM-1 (TRIF) pathway in myeloid dendritic cells. Proc Natl Acad Sci U S A 104(1):252–257
Azuma I, Ribi EE, Meyer TJ, Zbar B (1974) Biologically active components from mycobacterial cell walls. I. Isolation and composition of cell wall skeleton and component P3. J Natl Cancer Inst 52(1):95–101
Azuma M, Sawahata R, Akao Y, Ebihara T, Yamazaki S, Matsumoto M, Hashimoto M, Fukase K, Fujimoto Y, Seya T (2010) The peptide sequence of diacyl lipopeptides determines dendritic cell TLR2-mediated NK activation. PLoS One 5(9):e12550
Azuma M, Ebihara T, Oshiumi H, Matsumoto M, Seya T (2012) Cross-priming for antitumor CTL induced by soluble Ag + polyI:C depends on the TICAM-1 pathway in mouse CD11c(+)/CD8α(+) dendritic cells. Oncoimmunology 1(5):581–592
Azuma M, Takeda Y, Okada S, Nakajima H, Sugiyama H, Ebihara T, Oshiumi H, Matsumoto M, Seya T (2015) BATF3 in CD8a+DC sustains TLR3-derived IL-12 induction and CD11c+ anti-tumor T cell responses during antigen + poly(I:C) treatment. Cancer Res Immunol (submitted)
Bell JK, Askins J, Hall PR, Davies DR, Segal DM (2006) The dsRNA binding site of human Toll-like receptor 3. Proc Natl Acad Sci U S A 103(23):8792–8797
Caskey M, Lefebvre F, Filali-Mouhim A, Cameron MJ, Goulet JP, Haddad EK, Breton G, Trumpfheller C, Pollak S, Shimeliovich I, Duque-Alarcon A, Pan L, Nelkenbaum A, Salazar AM, Schlesinger SJ, Steinman RM, Sékaly RP (2011) Synthetic double-stranded RNA induces innate immune responses similar to a live viral vaccine in humans. J Exp Med 208(12):2357–2366
Coley WB (1891) Contribution to the knowledge of sarcoma. Ann Surg 14:199–200
Conforti R, Ma Y, Morel Y, Paturel C, Terme M, Viaud S, Ryffel B, Ferrantini M, Uppaluri R, Schreiber R, Combadière C, Chaput N, André F, Kroemer G, Zitvogel L (2010) Opposing effects of toll-like receptor (TLR3) signaling in tumors can be therapeutically uncoupled to optimize the anticancer efficacy of TLR3 ligands. Cancer Res 70(2):490–500
Desmet CJ, Ishii KJ (2012) Nucleic acid sensing at the interface between innate and adaptive immunity in vaccination. Nat Rev Immunol 12(7):479–491
Ebihara T, Azuma M, Oshiumi H, Kasamatsu J, Iwabuchi K, Matsumoto K, Saito H, Taniguchi T, Matsumoto M, Seya T (2010) Identification of a polyI:C-inducible membrane protein that participates in dendritic cell-mediated natural killer cell activation. J Exp Med 207(12):2675–2687
Galluzzi L, Vacchelli E, Eggermont A, Fridman WH, Galon J, Sautès-Fridman C, Tartour E, Zitvogel L, Kroemer G (2012) Trial Watch: experimental Toll-like receptor agonists for cancer therapy. Oncoimmunology 1(5):699–716
Honda K, Takaoka A, Taniguchi T (2006) Type I interferon gene induction by the interferon regulatory factor family of transcription factors. Immunity 25(3):349–360
Ishikawa E, Ishikawa T, Morita YS, Toyonaga K, Yamada H, Takeuchi O, Kinoshita T, Akira S, Yoshikai Y, Yamasaki S (2009) Direct recognition of the mycobacterial glycolipid, trehalose dimycolate, by C-type lectin Mincle. J Exp Med 206(13):2879–2888
Kang JY, Nan X, Jin MS, Youn SJ, Ryu YH, Mah S, Han SH, Lee H, Paik SG, Lee JO (2009) Recognition of lipopeptide patterns by Toll-like receptor 2-Toll-like receptor 6 heterodimer. Immunity 31:873–884
Kasamatsu J, Azuma M, Oshiumi H, Morioka Y, Okabe M, Ebihara T, Matsumoto M, Seya T (2014) INAM plays a critical role in IFN-g production by NK cells interacting with polyinosinic-polycytidylic acid-stimulated accessory cells. J Immunol 193:5199–5207
Kasamatsu J, Takahashi S, Azuma M, Matsumoto M, Morii-Sakai A, Imamura M, Teshima T, Takahashi A, Hirohashi Y, Torigoe T, Sato N, Seya T (2015) PolyI: C and mouse survivin artificially embedding human 2B peptide induce a CD4+ T cell response to autologous survivin in HLA-A*2402 transgenic mice. Immunobiology 220:74–82
Kim S, Takahashi H, Lin WW, Descargues P, Grivennikov S, Kim Y, Luo JL, Karin M (2009) Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 457(7225):102–106
Kodama K, Higashiyama M, Takami K, Oda K, Okami J, Maeda J, Akazawa T, Matsumoto M, Seya T, Wada M, Hayashi A, Toyoshima K (2009) Innate immune therapy with a BCG cell wall skeleton after radical surgery for non-small cell lung cancer: a case control study. Surg Today 39:194–200
Kono H, Rock KL (2008) How dying cells alert the immune system to danger. Nat Rev Immunol 8:279–289
Koyasu S, Moro K, Tanabe M, Takeuchi T (2010) Natural helper cells: a new player in the innate immune response against helminth infection. Adv Immunol 108:21–44
Lamm DL, Blumenstein BA, Crawford ED, Montie JE, Scardino P, Grossman HB, Stanisic TH, Smith JA Jr, Sullivan J, Sarosdy MF et al (1991) A randomized trial of intravesical doxorubicin and immunotherapy with bacille Calmette-Guérin for transitional-cell carcinoma of the bladder. N Engl J Med 325(17):1205–1209
Levine AS, Sivulich M, Wiernik PH, Levy HB (1979) Initial clinical trials in cancer patients of polyriboinosinic-polyribocytidylic acid stabilized with poly-L-lysine, in carboxymethylcellulose [poly(ICLC)], a highly effective interferon inducer. Cancer Res 39(5):1645–1650
Levy HB, Law LW, Rabson AS (1969) Inhibition of tumor growth by polyinosinic-polycytidylic acid. Proc Natl Acad Sci U S A 62(2):357–361
Liu L, Botos I, Wang Y, Leonard JN, Shiloach J, Segal DM, Davies DR (2008) Structural basis of toll-like receptor 3 signaling with double-stranded RNA. Science 320:379–381
Maruyama C (1971) Treatment of malignant tumors by an extract from tubercle bacilli (tuberculosis vaccine) [in Japanese]. Nihon Ika Daigaku Zasshi 38(5):267–276
Maruyama A, Shime H, Takeda Y, Azuma M, Matsumoto M, Seya T. (2015) Pam2 lipopeptides systemically increase myeloid-derived suppressor cells through TLR2 signaling. Biochem Biophys Res Commun 457(3):445–450
Matsumoto M, Seya T (2008) TLR3: interferon induction by double-stranded RNA including poly(I:C). Adv Drug Deliv Rev 60:805–812
Matsumoto M, Funami K, Tanabe M, Oshiumi H, Shingai M, Seto Y, Yamamoto A, Seya T (2003) Subcellular localization of Toll-like receptor 3 in human dendritic cells. J Immunol 171(6):3154–3162
Matsumoto M, Tatematsu M, Nishikawa F, Azuma M, Shime H, Seya T (2015) Defined TLR3-specific adjuvant that induces NK and cytotoxic T cell activation without significant cytokine production in vivo. Nat Commun 6:6280
Medzhitov R, Janeway CA Jr (1997) Innate immunity: the virtues of a nonclonal system of recognition. Cell 91(3):295–298
Murata M (2008) Activation of Toll-like receptor 2 by a novel preparation of cell wall skeleton from Mycobacterium bovis BCG Tokyo (SMP-105) sufficiently enhances immune responses against tumors. Cancer Sci 99(7):1435–1440
Paone A, Starace D, Galli R, Padula F, De Cesaris P, Filippini A, Ziparo E, Riccioli A (2008) Toll-like receptor 3 triggers apoptosis of human prostate cancer cells through a PKC-alpha-dependent mechanism. Carcinogenesis 29(7):1334–1342
Pardoll DM (2002) Spinning molecular immunology into successful immunotherapy. Nat Rev Immunol 2(4):227–238
Ray A, Cot M, Puzo G, Gilleron M, Nigou J (2013) Bacterial cell wall macroamphiphiles: pathogen-/microbe-associated molecular patterns detected by mammalian innate immune system. Biochimie 95(1):33–42
Rosenberg SA, Yang JC, Restifo NP (2004) Cancer immunotherapy: moving beyond current vaccines. Nat Med 10(9):909–915
Schoenen H, Bodendorfer B, Hitchens K, Manzanero S, Werninghaus K, Nimmerjahn F, Agger EM, Stenger S, Andersen P, Ruland J, Brown GD, Wells C, Lang R (2010) Cutting edge: Mincle is essential for recognition and adjuvanticity of the mycobacterial cord factor and its synthetic analog trehalose-dibehenate. J Immunol 184(6):2756–2760
Seya T, Matsumoto M, Tsuji S, Begum NA, Azuma I, Toyoshima K (2002) Structural-functional relationship of pathogen-associated molecular patterns: lessons from BCG cell wall skeleton and mycoplasma lipoprotein M161Ag. Microbes Infect 4(9):955–961
Seya T, Akazawa T, Uehori J, Matsumoto M, Azuma I, Toyoshima K (2003) Role of toll-like receptors and their adaptors in adjuvant immunotherapy for cancer. Anticancer Res 23(6a):4369–4376
Seya T, Shime H, Takaki H, Azuma M, Oshiumi H, Matsumoto M (2012) TLR3/TICAM-1 signaling in tumor cell RIP3-dependent necroptosis. Oncoimmunology 1(6):917–923
Seya T, Azuma M, Matsumoto M (2013) Targeting TLR3 with no RIG-I/MDA5 activation is effective in immunotherapy for cancer. Expert Opin Ther Targets 17(5):533–544
Shime H, Matsumoto M, Oshiumi H, Tanaka S, Nakane A, Iwakura Y, Tahara H, Inoue N, Seya T (2012) Toll-like receptor 3 signaling converts tumor-supporting myeloid cells to tumoricidal effectors. Proc Natl Acad Sci U S A 109(6):2066–2071
Shime H, Kojima A, Maruyama A, Saito Y, Oshiumi H, Matsumoto M, Seya T (2014) Myeloid-derived suppressor cells confer tumor-suppressive functions on natural killer cells via polyinosinic:polycytidylic acid treatment in mouse tumor models. J Innate Immun 6(3):293–305
Spits H, Artis D, Colonna M, Diefenbach A, Di Santo JP, Eberl G, Koyasu S, Locksley RM, McKenzie AN, Mebius RE, Powrie F, Vivier E (2013) Innate lymphoid cells—a proposal for uniform nomenclature. Nat Rev Immunol 13(2):145–149
Takeda Y, Azuma M, Hatsugai R, Akazawa T, Ebihara T, Matsumoto M, Seya T (2015) Efficient MyD88-dependent cross-priming and EG7 tumor regression induced by mycoplasma lipopeptide MALP-2s. (submitted)
Takemura R, Takaki H, Shime H, Oshiumi H, Matsumoto M, Tejima T, Seya T (2015) Necroptosis induced by the TICAM-1-RIP1/3 pathway in CT26 cells. Cancer Immunol Res (in press)
Taniguchi T, Mantei N, Schwarzstein M, Nagata S, Muramatsu M, Weissmann C (1980) Human leukocyte and fibroblast interferons are structurally related. Nature 285(5766):547–549
Tatematsu M, Nishikawa F, Seya T, Matsumoto M (2013) Toll-like receptor 3 recognizes incomplete stem structures in single-stranded viral RNA. Nat Commun 4:1833
Tokunaga T, Yamamoto T, Yamamoto S (1999) How BCG led to the discovery of immunostimulatory DNA. Jpn J Infect Dis 52(1):1–11
Tsuji S, Matsumoto M, Takeuchi O, Akira S, Azuma I, Hayashi A, Toyoshima K, Seya T (2000) Maturation of human dendritic cells by cell wall skeleton of Mycobacterium bovis bacillus Calmette-Guérin: involvement of toll-like receptors. Infect Immun 68(12):6883–6890
Uehori J, Fukase K, Akazawa T, Uematsu S, Akira S, Funami K, Shingai M, Matsumoto M, Azuma I, Toyoshima K, Kusumoto S, Seya T (2005) Dendritic cell maturation induced by muramyl dipeptide (MDP) derivatives: monoacylated MDP confers TLR2/TLR4 activation. J Immunol 174(11):7096–7103
Vacchelli E, Eggermont A, Sautès-Fridman C, Galon J, Zitvogel L, Kroemer G, Galluzzi L (2013) Trial Watch: Toll-like receptor agonists for cancer therapy. OncoImmunology 2(8):e25238
van der Bruggen P, Traversari C, Chomez P, Lurquin C, De Plaen E, Van den Eynde B, Knuth A, Boon T (1991) A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 254(5038):1643–1647
Yamazaki S, Okada K, Maruyama A, Matsumoto M, Yagita H, Seya T (2011) TLR2-dependent induction of IL-10 and Foxp3+ CD25+ CD4+ regulatory T cells prevents effective anti-tumor immunity induced by Pam2 lipopeptides in vivo. PLoS One 6(4):e18833
Yamazaki S, Maruyama A, Okada K, Matsumoto M, Morita A, Seya T (2012) Dendritic cells from oral cavity induce Foxp3(+) regulatory T cells upon antigen stimulation. PLoS One 7:e51665
Yasumoto K, Manabe H, Yanagawa E, Nagano N, Ueda H, Hirota N, Ohta M, Nomoto K, Azuma I, Yamamura Y (1979) Nonspecific adjuvant immunotherapy of lung cancer with cell wall skeleton of Mycobacterium bovis Bacillus Calmette-Guérin. Cancer Res 39:3262–3267
Acknowledgement
Thoughtful discussions by our laboratory members are gratefully acknowledged. There is no competing interest in this study. This work was supported in part by Grants-in-Aid from the Ministry of Education, Science, and Culture and the Ministry of Health, Labor, and Welfare of Japan, and by a MEXT Grant-in-Project ‘the Carcinogenic Spiral’ from the National Cancer Center Research and Development Fund (23-A-44). This work was also supported by the Takeda Science Foundation, the Yasuda Cancer Research Foundation, and the Iskra Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Japan
About this chapter
Cite this chapter
Matsumoto, M., Azuma, M., Seya, T. (2015). Adjuvant Immunotherapy for Cancer: From Basic Research to Clinical Bench. In: Seya, T., Matsumoto, M., Udaka, K., Sato, N. (eds) Inflammation and Immunity in Cancer. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55327-4_18
Download citation
DOI: https://doi.org/10.1007/978-4-431-55327-4_18
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55326-7
Online ISBN: 978-4-431-55327-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)