Summary
Theophylline (1,3 dimethylxanthine) is one of the most widely used drugs in the therapy of bronchial asthma and chronic obstructive pulmonary diseases. It is now being increasingly recognized that theophylline is able to down-regulate, both in-vitro and in-vivo, a variety of inflammatory and immune cell functions, such as the late-phase reaction after challenge with the occupational agent toluene diisocyanate and the bronchial responsiveness. The suppressive activity of theophylline is due to its ability to decrease the release of inflammatory mediators and might have important implications with regards to its therapeutic activity in asthma. In this report we summarize the effects of theophylline on TNF-α release, an inflammatory mediator which plays a key role in the pathogenesis of airways inflammation associated with bronchial asthma, by human peripheral blood monocytes (BM) and alveolar macrophages (AM) as well as on the activation and the proliferation of lymphocytes.
The data reported in this paper show that theophylline suppresses TNF-α release and TNF-α gene expression by BM and AM in a dose- dependent fashion at a concentration comparable to the in-vivo therapeutic levels of theophylline in the blood with a maximal inhibition at 2 h and that removal of theophylline after pre-incubation completely abrogates the inhibitory activity on TNF-α release, demonstrating that the continuous presence of the drug is required for suppression of TNF-α release.
In addition, theophylline is also able to affect the function of lymphocytes reducing their activation and proliferation by decreasing the production of IL-2, IL-2 receptor, transferrin and class IMHC antigens.
These data indicate that theophylline is capable of modulating important inflammatory and immune processes and that the therapeutic activity of this drug might be partly related to its down-regulating effects.
Zusammenfassung
Können Cytokine beim Asthma durch Theophyllin moduliert werden? Asthma ist eine chronisch-entzündliche Erkrankung, welche durch die Anhäufung von entzündlichen und immunologischen Effektorzellen (wie Eosinophile, Lymphozyten, Mastzellen und Makrophagen) in den Atemwegen und in der Bronchialschleimhaut charakterisiert ist.
Diese Zellen spielen eine wichtige Rolle bei der Pathogenese der Erkrankung, in dem sie sich gegenseitig beeinflussen, aber auch auf die strukturellen Zellverbände durch Expression von Adhäsionsmolekülen und die Freisetzung von Entzündungscytokinen wirken.
Wir haben angenommen, daß unter diesen Cytokinen der Tumornekrosisfaktor Alpha (TNF Alpha), ein 17-dK Cytokin, das von Lipopolysaccharid (LPS)-stimulierten mononukleären Phagozyten produziert wird, an wichtiger Stelle in die Pathogenese der Erkrankung involviert ist, weil es mit pleomorphen biologischen Aktivitäten ausgestattet ist.
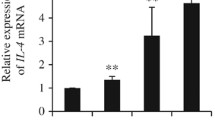
Wir konnten sehen, daß Alveolarmakrophagen (AM) von schweren asthmatischen Patienten spontan große Mengen von TNF Alpha freisetzten (18 ± 3 ng/ml). Weiters sahen wir, daß Endothelin-1 die TNF- Alpha-Freisetzung bei normalen AM steigerte (4 ± 1 ng/ml), bei den AM von schweren Asthmatikern aber die TNF Alpha-Freisetzung aus den AM deutlich verminderte (14 ± 3 ng/ml). Diese Befunde legen nahe, daß beim Bronchialasthma die AM spontan dazu aktiviert werden, TNF Alpha freizusetzen, und daß ihre Fähigkeit vermindert wird, auf heterogene Reize zu reagieren.
Wir haben dann den Effekt von Theophyllin (1,3 Dimethylxanthine) auf die Freisetzung von TNF Alpha aus peripheren Blutmonozyten (BM) und AM untersucht. Diesbezügliche frühere Studien hatten folgendes ergeben:
-
1.
Theophyllin vermindert die Funktion von Entzündungs- und Immunzellen durch Steigerung der intrazellulären Anhäufung von cAMP;
-
2.
Die Freisetzung von TNF Alpha aus mononuklearen Phagozyten wird durch den cAMP Spiegel reguliert.
Theophyllin reduziert dosis-abhängig den bioaktiven TNF-Alpha- Ausstoß aus menschlichen BM. Eine signifikante Hemmung wurde bei 100 µM beobachtet (40,8 ± 5,9% bei Kontrollen; p < 0,01) und bei 50 µM (59,2 ± 4,8% bei Kontrollen; p < 0,05), während die Aktivität von Theophyllin bei 10 µM (71,2 ± 8,9% bei Kontrollen) nicht statistisch signifikant war. Die Aktivität erreichte ihr Maximum nach 2 Stunden und fiel nach 4 Stunden (59,0 ± 5,2% der Kontrollen) und nach 24 Stunden (89,1 ±3,1% der Kontrollen) wieder ab. Eine Northern Blot- Analyse von RNA, die aus menschlichen BM extrahiert worden waren, zeigte, daß Theophyllin imstande war, die TNF Alpha-Gen-Expression zu vermindern. Zumal TNF Alpha an wichtiger Stelle in der Pathogenese der bronchialen Hyperreaktivität und des Asthma involviert ist, lassen diese Befunde vermuten, daß die therapeutische Aktivität von Theophyllin zumindest teilweise über seinen Effekt auf den TNF Alpha Ausstoß von mononukleären Phagozyten erklärbar ist.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Piafsky KM, Ogilvie R (1975) Dosage of theophylline in bronchial asthma. N Engl J Med 292: 1218–1222
Aubier M, De Troyer A, Sampson M, Macklem PT, Roussos C (1981) Aminophylline improves diaphragmatic contractility. N Engl J Med 305: 249–252
Pauwels RA (1987) The effects of theophylline on airway inflammation. Chest 92: 32s–37s
Mapp CE, Boschetto P, Dal Vecchio L, Crescioli S, De Marzo N, Paleari D, Fabbri LM (1987) Protective effect of antiasthma drugs on late asthmatic reactions and increased airway responsiveness induced by toluene diisocyanate in sensitized subjects. Am Rev Respir Dis 136: 1403–1407
Crescioli S, Spinazzi A, Plebani M, Pozzani M, Mapp CE, Boschetto P, Fabbri LM (1991) Theophylline inhibits early and late reactions induced by allergens in asthmatic subjects. Ann Allergy 66: 245–251
Metzger WJ, Zavala D, Richerson HB, Moseley P, Iwamota P, Monick M, Sjoerdsama K, Hunninghake G (1987) Local allergen challenge and bronchoalveolar lavage of allergic asthmatic lungs. Am Rev Respir Dis 135: 433–440
Joseph M, Tonnel AB, Torpier G, Capron A, Amoux B, Benveniste J (1983) The involvement of IgE in the secretory processes of alveolar macrophages from asthmatics patients. J Clin Invest 71: 221–226
Poston R, Chanez P, Lacoste JY, Litchfield P, Lee TK, Bousquet J (1992) Immunohistochemical characterization of the cellular infiltration of asthmatic bronchi. Am Rev Respir Dis 145: 918–921
Chanez P, Bousquet J, Couret I et al (1991) Increased numbers of hypodense alveolar macrophages in patients with bronchial asthma. Am Rev Respir Dis 144: 923–930
Amoux B, Joseph M, Simoes MH, Tonnel AB, Duroux P, Capron A, Benveniste J (1987) Antigenic release of PAF-acether and β-glucuronidase from alveolar macrophages of asthmatics. Bull Eur Physiopathol Respir 23: 119–124
Gosset P, Tsicopoulos A, Wallaert B, Vannimenus C, Joseph M, Tonnel AB, Capron A (1991) Increased secretion of tumor necrosis factor alpha and interleukin-6 by alveolar macrophages consecutive to the development of the late asthmatic reaction. J Allergy Clin Immunol 88: 561–571
Calhoun WJ, Reed HE, Moest DR, Stevens CA (1992) Enhanced superoxide production by alveolar macrophages and air-space cells, airway inflammation, and alveolar macrophage density changes after segmental antigen bronchoprovocation in allergic subjects. Am Rev Respir Dis 145: 317–325
Chanez P, Vignola AM, Lacoste P, Michel FB, Godard P, Bousquet J (1993) Increased expression of ICAM-1 and LFA-1 on alveolar macrophages from asthmatics. Allergy (in press) 48: 576–580
Gosset P, Tsicopoulos A, Benoit W, Joseph M, Capron A, Tonnel AB (1992) Tumor necrosis alpha and interleukin-6 production by human mononuclear phagocytes from allergic asthmatics after Ig-E dependent stimulation. Am Rev Respir Dis 146: 768–784
Chanez P, Vignola AM, Springal DR, Campbell AM, Farce M, Polak J, Michel FB, Godard FB, Bousquet J (1993) Endothelin and airway cells in asthma. Am Rev Respir Dis 147: A517
Yanagawa H, Sone S, Sugihara K, Tanaka K, Ogura T (1991) Interleukin-4 down-regulates Interleukin-6 production by human alveolar at protein and mRNA levels. Microbiol Immunol 35: 879–893
Carswell EA, Old LJ, Green S, Fiore N, Williamson B (1975) An endotoxin- induced serum factor that causes necrosis of tumors. Proc Natl Acad Sci USA 72: 3666–3670
Beutler B, Cerami A (1987) Cachectin: more than a tumor necrosis factor. N Engl JMed 316: 379–385
Lynch JP III, Toews GB (1989) Tumor necrosis factor-a. A multifaceted mediator of inflammation. Chest 96: 457–459
Taffet SM, Singhel KJ, Overholtzer JF, Shurtleff SA (1989) Regulation of tumor necrosis factor expression in a macrophage-like cell line by lipopoly- saccharide and cyclic AMP Cell Immunol 120: 291–300
Matsushima K, Morishita K, Yoshimura T, Lavu S, Kobayashi Y, Lew W, Appella E, Kung HF, Leonard EJ, Oppenheim JJ (1988) Molecular cloning of a human monocyte-derived neutrophil chemotactic factor (MDNCF) and the induction of MDNCF mRNA by interleukin 1 and tumor necrosis factor. J Exp Med 167: 1883–1893
Salyer JL, Bohnsack JF, Knape WA, Shigeoka AO, Ashwood ER, Hill HR (1990) Mechanisms of tumor necrosis alpha alteration of PMN adhesion and migration. Am J Pathol 136: 831–841
Moser R, Scleiffenbaum B, Groschurth P, Fehr J (1989) Interleukin 1 and tumor necrosis factor stimulate human vascular endothelial cells to promote transendothelial neutrophil passage. J Clin Invest 83: 444–455
Ohno I, Tanno Y, Yamauchi K, Takishima T (1990) Gene expression and production of tumor necrosis factor by a rat basophilic leukemia cell line (RBL-2H3) with IgE receptor triggering. Immunology 70: 88–93
Rail TW (1980) The Xanthines. In: Oilman AG, Goodman LS, Rail TW, Murad F (eds) The pharmacological basis of therapeutics. Mac Millan, New York, pp 592–607
O’Neill SH, Sitar DS, Klass DJ, Taraska VA, Kepron W, Mitenko PA (1986) The pulmonary disposition of theophylline and its influence on human alveolar macrophage bactericidal function. Am Rev Respir Dis 134: 1225–1228
Shapira Z, Shoat B, Boner G, Levi J, Joshua H, Servadio C (1982) Theophylline: a possible immunoregulator of T-cells. Transpl Proc 14: 113–118
Pardi R, Zocchi MR, Ferrero E, Ciboddo GE, Invemardi L, Rugarli C (1984) In vivo effects of a single infusion of theophylline on human peripheral blood lymphocytes. Clin Exp Immunol 57: 722–728
Scordamaglia A, Ciprandi G, Ruffoni S, Caria M, Paolieri F, Venuti D, Canonica GW (1988) Theophylline and the immune response: in vitro and in vivo effects. Clin Immunol Immunpathol 46: 238–246
Rao KMK, Currie MS, McCachren SP, Cohen HJ (1991) Pentoxifylline and other methylxanthines inhibit interleukin-2 receptor expression in human lymphocytes. Cell Immunol 135: 314–325
Coskey LA, Roth MD (1991) Differential regulation of natural killer cell activity by theophylline and enprophylline. Am Rev Respir Dis 143: A229
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1995 Springer-Verlag/Wien
About this chapter
Cite this chapter
Vignola, A.M. et al. (1995). Can cytokines in asthma be modulated by theophylline?. In: Kummer, F. (eds) Asthma. Springer, Vienna. https://doi.org/10.1007/978-3-7091-7539-2_11
Download citation
DOI: https://doi.org/10.1007/978-3-7091-7539-2_11
Publisher Name: Springer, Vienna
Print ISBN: 978-3-211-82670-6
Online ISBN: 978-3-7091-7539-2
eBook Packages: Springer Book Archive