Abstract
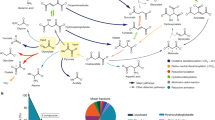
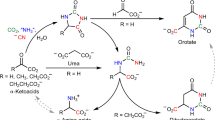
The biosynthesis of hydroxamic acids containing δ-N-hydroxyornithine has now been investigated and appears to proceed by reactions analogous to those in hadacidin biosynthesis. Thus, we have found that δ-N-hydroxyornithine is incorporated into ferrichrome and ferrichrome A by Ustilago sphaerogena to an extent of 35–50% in six hour periods. Ornithine is incorporated to approximately one-half the extent as its δ-hydroxylamino derivative under identical conditions. δ-N-Acetyl-δ-N-hydroxyornithine, the hydroxamic acid subunit of ferrichrome, was found to be a precursor of ferrichrome but not ferrichrome A, in which the acyl groups of the hydroxamate functions are β-methylglutaconate instead of acetate.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Beecham, A. F., J. Fridrichsons, and A. Mcl. Mathieson: The structure and absolute configuration of gliotoxin and the absolute configuration of sporidesmin. Tetrahedron Letters No 27, 3131 (1966a).
Beecham, A. F., and A. Mcl. Mathieson: The circular dichroism of gliotoxin. Tetrahedron Letters No 27, 3139 (1966b).
Bell, N. R., J. R. Johnson, B. S. Wilde, and R. B. Woodward: The structure of gliotoxin. J. Am. Soc. 80, 1001 (1958).
Behal, V.: Investigation of the orgin of 0-methyl groups in griseofulvin using some ‘4C-labelled substrates. Folia Microbiol. 11, 184 (1966).
Klimov, A. N., V. A. Bazanov, and T. P. Efimova: Some data on the mechanism of griseofulvin biosynthesis. Antibiotiki 10, 914 (1965).
Parrish, F. W., B. J. Wiley, E. G. Simmons, and L. Long: Production of aflatoxins and kojic acid by species of Aspergillus and Penicillium. Appl. Microbiol. 14, 139 (1966).
Martin, J. R., T. J. Perun, and R. L. Girolami: Studies on the biosynthesis of the erythromycins. I. Isolation and structure of an intermediate glycoside, 3-a-L-mycarosylerythronolide B. Biochemistry 5, 2852 (1966).
Martin, J. R., and W. Rosenbrook: Studies on the biosynthesis of the erythromycins. II. Isolation and structure of biosynthetic intermediate, 6-deoxyerythronolide B. Biochemistry 6, 435 (1967).
Bohlmann, F.: Natürlich vorkommende Acetylen-Verbindungen. Fortschr. chem. Forsch. 6, 65 (1966).
Bohlmann, F., C. Arndt, H. Bornowski u. K. M. Kleine: Polyacetylenverbindungen. Xxxi. Über Polyine aus der Familie der Umbelliferen. Chem. Ber. 94, 958 (1961).
Bohlmann, F., C. Arndt, and C. Zdero: Polyacetylenverbindungen. Cii. Über neue Enolätherpolyine aus Anaphalis und Gnaphalium-Arten. Chem. Ber. 99, 1648 (1966).
Bohlmann, F., and G. Florentz: Polyacetylenverbindungen. Xcvi. Über die Biogenese der spiroketalen Ather-Polyine. Chem. Ber. 99, 990 (1966).
Bohlmann, F., and R. Jente: Polyacetylenverbindungen. Xcvii. Zur Biogenese der Phenylpolyine. Chem. Ber. 99, 995 (1966).
Bohlmann, F., W. V. Kap-Herr, R. Jente and G. Grau: Polya CetylenVerbindungen. CV. Über die Biogenese natürlicher Acetylenverbindungen. Chem. Ber. 99, 2091 (1966).
Bohlmann, F., and H. G. Kapteyn: Polyacetylenverbindungen. Ciii. Über die Polyine aus Chrysanthemum serotinum L. Chem. Ber. 99, 1830 (1966).
Bohlmann, F., and J. Laser: Polyacetylenverbindungen. Civ. Zur Biogenese eines Thioäthers aus Anthemis tinctoria L. Chem. Ber. 99, 1834 (1966).
Bohlmann, F., M. Wotschokowsky, U. Hinz u. W. Lucas: Polyacetylenverbindungen. Xcv. Über die Biogenese einiger Thiophenverbindungen. Chem. Ber. 99, 984 (1966).
Bohlmann, F., and C. Zdero: Polyacetylenverbindungen. IC. Über ein Thiophenlacton aus Chamaemelum nobile L. Chem. Ber. 99, 1226 (1966).
Bu’Locx, J. D.: Biogenesis of natural polyacetylenes. Compt. Phytochem. 1966, 79.
Bu’Locx, J. D.: Biosynthesis of polyacetylenes in fungi, p. 141. In: J. F. Snell, Biosynthesis of antibiotics. New York: Academic Press 1967.
Bu’Locx, J. D., and G. N. Smith: The origin of naturally-occurring acetylenes. J. Chem. Soc. 1967, 332.
Hodge, P., E. R. H. JoNEs, and G. Lowe: Natural acetylenes Part Xxii. transDehydromatricaria ester as a biosynthetic precursor of some fungal polyacetylenes. J. Chem. Soc. 1966, 1216.
Jones, E. R. H., S. Safe, and V. Thaller: Natural acetylenes Part Xxiii. A C18 Polyacetylenic keto-aldehyde related to falcarinone from an Umbellifer (Pastinaca sativa L.). J. Chem. Soc. 1966, 1220.
Walker, M. S., and J. B. Walker: Enzymatic studies on the biosynthesis of streptomycin: Transamidination of inosamine and streptamine derivatives. J. Biol. Chem. 241, 1262 (1966).
Nomi, R., O. Nimi, and T. Miyazaki: Biosynthesis of streptomycin. Part 1. Properties of streptomycin-producing supernatant obtained from mycelium suspension of Streptomyces griseus. Agr. Biol. Chem. 30, 296 (1966).
Grisebach, H., U. U. DÖBereiner: Zur Biosynthese der Apiose. 1. Einbau markierter Vorstufen in Apiose bei Apium petroselinum. Z. Naturforsch. 21, 429 (1966).
Hanessian, S., D. C. Dejongh, and J. A. Mccloskey: Further evidence on the structure of cordycepin. Biochim. et Biophys. Acta 117, 480 (1966).
Kaczka, E. A., N. R. Trenner, B. Arison, R. W. Walker, and K. Folkers: Identification of cordycepin, a metabolite of Coryceps militaris, as 3’-deoxy-adenosine. Biochem. Biophys. Research Commun. 14, 456 (1964b).
Kemp, M. B., and J. R. Quayle: Incorporation of Cl units into allulose phosphate by extracts of Pseudomonas methanica. Biochem. J. 99, 41 (1966).
Matsushima, K., and F. J. Simpson: The purification and properties of n-allosephosphate isomerase of Aerobacter aerogenes. Can. J. Microbiol. 12, 313 (1966).
Suhadolnik, R. J., and J. G. Cora: Further evidence for the biosynthesis of cordycepin and proof of the structure of 3-deoxyribose. Biochim. et Biophys. Acta 91, 661 (1964a).
Castro, A. J., M. L. Furcolow, G. R. Gale, G. E. Means, and G. Tertzakian: Antimicrobial properties of pyrrole derivatives. J. Med. Chem. 10, 29 (1967).
Feofilova, E. P.: Effect of pyrrole derivatives and precursors on growth and pigment production by Streptomyces longispororuber. Mikrobiologiya 35, 64 (1966) (Chem. Abstr. 64, 16311 (1966).
Harashima, K., N. Tsuchida, and J. Nagatsu: Prodigiosin-25 C. A new prodigiosinlike pigment. Agr. Biol. Chem. 30, 309 (1966).
Morrison, D. A.: Prodigiosin synthesis in mutants of Serratia marcescens. J. Bacteriol. 91, 1599 (1966).
Nakajima, M.: Mechanism of prodigiosin biosynthesis. Bull. Osaka Med. School 11, 39 (1965a) [Chem. Abstr. 64, 20243 (1966)].
Nakajima, M.: Spectral property of prodigiosin. Bull. Osaka Med. School 11, 56 (1965b) [Chem. Abstr. 64, 20068 (1966)].
Shaposhnikov, V. N., M. N. Bekhtereva, N. V. Tarasova, N. M. Vinokurova, and Yu. M. Khokhlova: Organic acids and their role in formation of prodigiosin-like pigments in Streptomyces longissimus rameus and Streptomyces aureoverticillatus. Dokl. Akad. Nauk S.S.S.R. 166, 219 (1966) [Chem. Abstr. 64, 11583 (1966)].
Wasserman, H. H., J. E. Mckeon, L. A. Smith, and P. Forgione: Studies On prodigiosin and the bipyrrole precursor. Tetrahedron, Suppl. 8, part II, 647 (1966).
Wasserman, H. H., and G. C. Rodgers: Prodigiosin analogs from Streptomyces (Streptomycetaceae) and Serratia (Enterobacteriaceae) bacteria. Abstr. Papers. Iupac Symposium, the Chemistry of Natural Products, 2B-45, p. 101. Stockholm 1966.
Whitnack, G. C., and G. Soli: Characterization of bacterial pigments by single-sweep polarography. J. Electroanal. Chem. 12, 60 (1966).
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1967 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Emery, T. et al. (1967). Addenda. In: Gottlieb, D., Shaw, P.D. (eds) Biosynthesis. Antibiotics. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-38441-1_33
Download citation
DOI: https://doi.org/10.1007/978-3-662-38441-1_33
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-37650-8
Online ISBN: 978-3-662-38441-1
eBook Packages: Springer Book Archive