Abstract
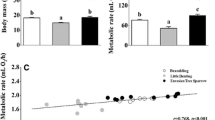
Information on body mass, basal metabolic rate (BMR) and body composition were obtained from adult female kittiwakes (Rissa tridactyla) breeding on Svalbard (79°N). While body mass, BMR and body composition was stable during the incubation period, both body mass and BMR decreased significantly during the chick-rearing period. Both whole-body and massspecific BMR decreased during this period indicating that the reduction in BMR must be due to an altered metabolic intensity. Body composition analyses revealed that while most organs masses changed in parallel to total body mass, the liver showed a disproportional decrease. In a subsample of individuals, a significant relationship was also found between liver mass and BMR. The results suggest that BMR is not a species-specific character, but rather a flexible physiological parameter, with liver mass being an important determinator.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Bech C, ∅stnes JE (1999) Influence of body composition on the metabolic rate of nestling European shags (Phalacrocorax aristotelis). J Comp Physiol B 169: 263–270
Bech C, Langseth I, Gabrielsen GW (1999) Repeatability of basal metabolism in breeding female kittiwakes Rissa Tridactyla. Proc R Soc Lond B 266: 2161–2167
Bennett PM, Harvey PH (1987) Active and resting metabolism in birds: allometry, phylogeny and ecology. J Zool, Lond 213: 327–363
Burness GP, Ydenberg RC, Hochachka PW (1998) Interindividual variability in body composition and resting oxygen consumption rate in breeding Tree Swallows, Tachycineta bicolor. Physiol Zool 71: 247–256
Burrin DG, Britton RA, Ferrell CL (1988) Visceral organ size and hepatocyte metabolic activity in fed and fasted rats. J Nutr 118: 1547–1552
Chappell MA, Bech C, Buttemer WA (1999) The relationship of central and peripheral organ masses to aerobic performance variation in house sparrows. J Exp Biol 202: 2269–2279
Daan S, Masman D, Groenewold A (1990) Avian basal metabolic rates: their association with body composition and energy expenditure in nature. Am J Physiol 259: R333-R340
Freed LA (1981) Loss of mass in breeding wrens: stress or adaptation? Ecology 62:1179–1186
Gabrielsen GW, Mehlum F, Karlsen HE (1988) Thermoregulation in four species of arctic seabirds. J Comp Physiol B 157. 703–708
Go let GH, Irons DB (1999) Raising young reduces body condition and fat stores in black-legged kittiwakes. Oecologia 120: 530–538
Goodmann MN, Ruderman NB (1980) Starvation in the rat.I. Effect of age and obesity on organ weights, RNA, DNA and protein. Am J Physiol 239: E269–276
Hammond KA, Konarzewski M, Torres RM, Diamond J (1994) Metabolic ceilings under a combination of peak energy demands. Physiol Zool 67: 1479–1506
IUPS Thermal Commission (1987) Glossary of terms for thermal physiology. Pflügers Archiv 410: 567–587
Krebs HA (1950) Body size and tissue respiration. Biochim Biophys Acta 4: 249–269
McNab BK (1988) Food habits and the basal rate of metabolism in birds. Oecologia 77: 343–349
Moreno J (1989) Strategies of mass changes in breeding birds. Biol J Linn Soc 37: 297–310
Niimi AJ (1978) Lag adjustment between estimated and actual physiological responses conducted in flow-through systems. J Fish Res Board Can 35: 1265–1269
Norberg RA (1981) Temporary weight decrease in breeding birds may result in more fledged young. Am Nat 118: 838–850
Piersma T, Lindström A(1997) Rapid reversible changes in organ size as a component of adaptive behaviour. Trends Ecol Evol 12: 134–138
Piersma T, Bruinzeel L, Drent R, Kersten M, VanderMeer J, Wiersma P (1996) Variability in basal metabolic rate of a long-distance migrant shorebird (Red Knot, Calidris canutus) reflects shifts in organ sizes. Physiol Zool 69: 191–217
Reynolds PS, Lee RM (1996) Phylogenetic analysis of avian energetics: Passerines and nonpasserines do not differ. Am Nat 147: 735–759
Scott I, Evans PR (1992) The metabolic output of avian (Sturnus vulgaris, Calidris alpina) adipose tissue, liver and skeletal muscle: implications for BMR/body mass relationships. Comp Biochem Physiol 103A: 329–332
Trevelyan R, Harvey PH, Pagel MD (1990) Metabolic rates and life histories in birds. Funct Ecol 4: 135–141
Weber TP, Piersma T (1996) Basal metabolic rate and the mass of tissues differing in metabolic scope: migration-related covariation between individual Knots Calidris canutus. J Avian Biol 27: 215–224
Withers PC (1977) Measurements of VO2, VCO2 and evaporative water loss with a flowthrough mask. J Appl Physiol 42: 120–123
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Langseth, I., Moe, B., Fyhn, M., Gabrielsen, G.W., Bech, C. (2000). Flexibility of Basal Metabolic Rate in Arctic breeding Kittiwakes (Rissa tridactyla). In: Heldmaier, G., Klingenspor, M. (eds) Life in the Cold. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-04162-8_48
Download citation
DOI: https://doi.org/10.1007/978-3-662-04162-8_48
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-08682-3
Online ISBN: 978-3-662-04162-8
eBook Packages: Springer Book Archive