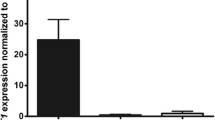
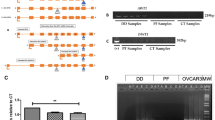
Abstract
Comparative gene expression analyses of Dupuytren’s disease tissues and unaffected palmar fascia have consistently identified large numbers of gene transcripts that encode extra-cellular matrix molecules. The relevance of a modified extra-cellular matrix to Dupuytren’s disease progression is unclear. In this study, we have tested two hypotheses: (1) that a modified extra-cellular matrix facilitates Dupuytren’s disease cell migration and (2) that Dupuytren’s disease cells modify their extra-cellular matrix to induce adjacent cells to take on disease cell characteristics. Our data suggest that transforming growth factor β1, periostin and a disintegrin and metalloprotease 12, molecules associated with the extra-cellular matrix and upregulated in Dupuytren’s disease, either inhibit or have no discernable effect on three-dimensional migration of primary Dupuytren’s disease cells relative to cells from phenotypically unaffected palmar fascia. In contrast, a collagen substrate conditioned by primary Dupuytren’s disease cells, but not patient-matched control cells, induces rapid β-catenin accumulation in cells from phenotypically unaffected palmar fascia. As upregulated cytoplasmic β-catenin is one of several characteristics that distinguish DD cells from cells of the adjacent palmar fascia, these data support the hypothesis that a modified extra-cellular matrix promotes Dupuytren’s disease progression by inducing fibroblasts resident in the palmar fascia to take on a disease cell phenotype.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Amini Nik S, Ebrahim RP, Van Dam K, Cassiman JJ, Tejpar S (2007) TGF-beta modulates beta-Catenin stability and signaling in mesenchymal proliferations. Exp Cell Res 313(13):2887–2895
Badalamente MA, Sampson SP, Hurst LC, Dowd A, Miyasaka K (1996) The role of transforming growth factor beta in Dupuytren’s disease. J Hand Surg Am 21(2):210–215
Berndt A, Kosmehl H, Mandel U, Gabler U, Luo X, Celeda D, Zardi L, Katenkamp D (1995) TGF beta and bFGF synthesis and localization in Dupuytren’s disease (nodular palmar fibromatosis) relative to cellular activity, myofibroblast phenotype and oncofetal variants of fibronectin. Histochem J 27(12):1014–1020
Bisson MA, McGrouther DA, Mudera V, Grobbelaar AO (2003) The different characteristics of Dupuytren’s disease fibroblasts derived from either nodule or cord: expression of alpha-smooth muscle actin and the response to stimulation by TGF-beta1. J Hand Surg Br 28(4):351–356
Bunyan AR, Mathur BS (2000) Medium thickness plantar skin graft for the management of digital and palmar flexion contractures. Burns 26(6):575–580
Caraci F, Gili E, Calafiore M, Failla M, La Rosa C, Crimi N, Sortino MA, Nicoletti F, Copani A, Vancheri C (2008) TGF-beta1 targets the GSK-3beta/beta-catenin pathway via ERK activation in the transition of human lung fibroblasts into myofibroblasts. Pharmacol Res 57(4):274–282
Cheon SS, Wei Q, Gurung A, Youn A, Bright T, Poon R, Whetstone H, Guha A, Alman BA (2006) Beta-catenin regulates wound size and mediates the effect of TGF-beta in cutaneous healing. FASEB J 20(6):692–701
Citron N, Hearnden A (2003) Skin tension in the aetiology of Dupuytren’s disease; a prospective trial. J Hand Surg Br 28(6):528–530
Evans RB, Dell PC, Fiolkowski P (2002) A clinical report of the effect of mechanical stress on functional results after fasciectomy for Dupuytren’s contracture. J Hand Ther 15(4):331–339
Goffin JM, Pittet P, Csucs G, Lussi JW, Meister JJ, Hinz B (2006) Focal adhesion size controls tension-dependent recruitment of alpha-smooth muscle actin to stress fibers. J Cell Biol 172(2):259–268
Gupta R, Allen F, Tan V, Bozentka DJ, Bora FW, Osterman AL (1998) The effect of shear stress on fibroblasts derived from Dupuytren’s tissue and normal palmar fascia. J Hand Surg Am 23(5):945–950
Hinz B (2009) Tissue stiffness, latent TGF-beta1 activation, and mechanical signal transduction: implications for the pathogenesis and treatment of fibrosis. Curr Rheumatol Rep 11(2):120–126
Hinz B, Dugina V, Ballestrem C, Wehrle-Haller B, Chaponnier C (2003) Alpha-smooth muscle actin is crucial for focal adhesion maturation in myofibroblasts. Mol Biol Cell 14(6):2508–2519
Howard JC, Varallo VM, Ross DC, Roth JH, Faber KJ, Alman B, Gan BS (2003) Elevated levels of beta-catenin and fibronectin in three-dimensional collagen cultures of Dupuytren’s disease cells are regulated by tension in vitro. BMC Musculoskelet Disord 4:16
Howard JC, Varallo VM, Ross DC, Faber KJ, Roth JH, Seney S, Gan BS (2004) Wound healing-associated proteins Hsp47 and fibronectin are elevated in Dupuytren’s contracture. J Surg Res 117(2):232–238
Hueston JT (1992) Regression of Dupuytren’s contracture. J Hand Surg Br 17(4):453–457
Hurst LC, Badalamente MA, Makowski J (1986) The pathobiology of Dupuytren’s contracture: effects of prostaglandins on myofibroblasts. J Hand Surg Am 11(1):18–23
Iwasaki H, Muller H, Stutte HJ, Brennscheidt U (1984) Palmar fibromatosis (Dupuytren’s contracture). Ultrastructural and enzyme histochemical studies of 43 cases. Virchows Arch A Pathol Anat Histopathol 405(1):41–53
Luck JV (1959) Dupuytren’s contracture; a new concept of the pathogenesis correlated with surgical management. J Bone Joint Surg Am 41(4):635–664
Qian A, Meals RA, Rajfer J, Gonzalez-Cadavid NF (2004) Comparison of gene expression profiles between Peyronie’s disease and Dupuytren’s contracture. Urology 64(2):399–404
Rajesh KR, Rex C, Mehdi H, Martin C, Fahmy NR (2000) Severe Dupuytren’s contracture of the proximal interphalangeal joint: treatment by two-stage technique. J Hand Surg Br 25(5):442–444
Rayan GM, Tomasek JJ (1994) Generation of contractile force by cultured Dupuytren’s disease and normal palmar fibroblasts. Tissue Cell 26(5):747–756
Rayan GM, Parizi M, Tomasek JJ (1996) Pharmacologic regulation of Dupuytren’s fibroblast contraction in vitro. J Hand Surg Am 21(6):1065–1070
Rehman S, Salway F, Stanley JK, Ollier WE, Day P, Bayat A (2008) Molecular phenotypic descriptors of Dupuytren’s disease defined using informatics analysis of the transcriptome. J Hand Surg Am 33(3):359–372
Shih B, Wijeratne D, Armstrong DJ, Lindau T, Day P, Bayat A (2009) Identification of biomarkers in Dupuytren’s disease by comparative analysis of fibroblasts versus tissue biopsies in disease-specific phenotypes. J Hand Surg Am 34(1):124–136
Thampatty BP, Wang JH (2007) A new approach to study fibroblast migration. Cell Motil Cytoskeleton 64(1):1–5
Tomasek J, Rayan GM (1995) Correlation of alpha-smooth muscle actin expression and contraction in Dupuytren’s disease fibroblasts. J Hand Surg Am 20(3):450–455
Tomasek JJ, Schultz RJ, Episalla CW, Newman SA (1986) The cytoskeleton and extracellular matrix of the Dupuytren’s disease “myofibroblast”: an immunofluorescence study of a nonmuscle cell type. J Hand Surg Am 11(3):365–371
Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA (2002) Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 3(5):349–363
Vi L, Feng L, Zhu RD, Wu Y, Satish L, Gan BS, O’Gorman DB (2009a) Periostin differentially induces proliferation, contraction and apoptosis of primary Dupuytren’s disease and adjacent palmar fascia cells. Exp Cell Res 315(20):3574–3586
Vi L, Gan BS, O’Gorman DB (2009b) The potential roles of cell migration and extra-cellular matrix interactions in Dupuytren’s disease progression and recurrence. Med Hypotheses 74(3):510–512
Vi L, Njarlangattil A, Wu Y, Gan BS, O’Gorman DB (2009c) Type-1 collagen differentially alters beta-catenin accumulation in primary Dupuytren’s Disease cord and adjacent palmar fascia cells. BMC Musculoskelet Disord 10(1):72
Wipff PJ, Hinz B (2009) Myofibroblasts work best under stress. J Bodyw Mov Ther 13(2):121–127
Wipff PJ, Rifkin DB, Meister JJ, Hinz B (2007) Myofibroblast contraction activates latent TGF-beta1 from the extracellular matrix. J Cell Biol 179(6):1311–1323
Acknowledgments
We would like to acknowledge Ms Karen Nygard at the Biotron of the University of Western Ontario for her expert assistance with stereomicroscopy. We also acknowledge the Canadian Institutes of Health Research and The Lawson Health Research Institute Internal Research Fund for financial support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Vi, L., Wu, Y., Gan, B.S., O’Gorman, D.B. (2012). Primary Dupuytren’s Disease Cell Interactions with the Extra-cellular Environment: A Link to Disease Progression?. In: Eaton, C., Seegenschmiedt, M., Bayat, A., Gabbiani, G., Werker, P., Wach, W. (eds) Dupuytren’s Disease and Related Hyperproliferative Disorders. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-22697-7_19
Download citation
DOI: https://doi.org/10.1007/978-3-642-22697-7_19
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-22696-0
Online ISBN: 978-3-642-22697-7
eBook Packages: MedicineMedicine (R0)