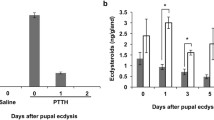
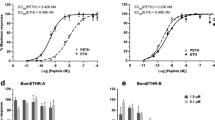
Abstract
Insects are encased in a semi-rigid exoskeleton, which provides protection, locomotion, and internal attachments sites for muscles and internal organs. However, it also limits insect’s growth. Insect must shed its old exoskeleton periodically (molting) in order to grow. After each molt, the newly formed exoskeleton is usually soft, lightly pigmented and flexible, permitting insect to extend its body size for growth. Sclerotization of newly formed cuticle must occur in a relatively short period of time after each molt and completion of body expansion in order for the new exoskeleton to be functional for protection. Insects could not survive without properly hardened cuticle. Molting and cuticle sclerotization in insects are regulated by a precise coordination of at least 6 hormones including prothoracicotropic hormone (PTTH), 20-hydroxyecdysone (20E), eclosion hormone (EH), ecdysis triggering hormone (ETH), crustacean cardioactive peptide (CCAP) and bursicon. In our previous chapter (Song and Sun 2005 edited by Liu and Kang), we already introduced signal transduction pathways of PTTH, 20E and JH. Here in this chapter, we would like to summarize the molecular mechanisms of EH, ETH, CCAP and bursicon and their coordination in regulating the final steps of the molting process i.e. ecdysis and cuticle sclerotization. In particular, we will focus on the action of bursicon. The elucidation of the key biochemical events leading to cuticle sclerotization would add an important body of knowledge towards understanding insect development and for designing more efficient pest control strategies to disrupt the cuticle sclerotization process.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
An S, Wang S, Gilbert L, et al. Global identification of bursicon-regulated genes in Drosophila melanogaster. BMC Genomics, 2008, 9: 424.
An S, Wang S, Song Q. Identification of a novel bursiconregulated transcriptional regulator, md13379, in the house fly Musca domestica. Arch. Insect Biochem. Physiol., 2009, 70: 106–121.
Arakane Y, Muthukrishnan S, Beeman RW, et al. Laccase 2 is the phenoloxidase gene required for beetle cuticle tanning. Proc. Natl. Acad. Sci. USA, 2005, 102: 11337–11342.
Baker J, Truman JW. Mutations in the Drosophila glycoprotein hormone receptor, rickets, eliminate neuropeptideinduced tanning and selectively block a stereotyped behavioral program. J. Exp. Biol., 2002, 205: 2555–2565.
Cottrell C B. The imaginal ecdysis of blowflies. The control of cuticular hardening and darkening. J. Exp. Biol., 1962a, 39: 395–411.
Cottrell C B. The imaginal ecdysis of blowflies. Detection of the blood-borne darkening factor and determination of some of its properties. J. Exp. Biol., 1962b, 39: 413–430.
Curtiss J, Halder G, Mlodzik M. Selector and signaling molecules cooperate in organ patterning. Nat. Cell Biol., 2002, 4: E48–E51.
Crozatier M, Glise B, Khemici V, Vincent A. Veinpositioning in the Drosophila wing in response to Hh; new roles of Notch signaling. Mech. Dev., 2003, 120: 529–535.
Davis M M, O’Keefe S L, Primrose D A, Hodgetts R B. A neuropeptide hormone cascade controls the precise onset of posteclosion cuticular tanning in Drosophila melanogaster. Development, 2007, 134: 4395–4404.
Dewey E M, McNabb S L, Ewer J, et al. Identification of the gene encoding bursicon, an insect neuropeptide responsible for cuticle sclerotization and wing spreading. Curr. Biol., 2004, 14: 1208–1213.
Ekengren S, Tryselius Y, Dushay M S, et al. A humoral stress response in Drosophila. Curr. Biol., 2001, 11: 714–718.
Eriksen K K, Hauser F, Schiott M, et al. Molecular cloning, genomic organization, developmental regulation, and a knock-out mutant of a novel leu-rich repeats-containing G protein-coupled receptor (DLGR-2) from Drosophila melanogaster. Genome Res., 2000, 10: 924–938.
Ewer J, Reynolds S. Neuropeptide control of molting in insects. // Pfaff DW, Arnold A P, Fahrbach S E, et al. Brain and Behavior. San Diego: Academic Press. 2002: 1–92.
Fogal W, Fraenkel G. The role of bursicon in melanization and endocuticle formation in the adult fleshfly, Sarcophaqa bullata. J. Insect Physiol., 1969, 15: 1235–1247.
Fortini M E, Artavanis-Tsakonas S. The suppressor of hairless protein participates in notch receptor signaling. Cell, 1994, 79: 273–282.
Fraenkel G, Hsiao C. Hormonal and nervous control of tanning in the fly. Science, 1962, 138: 27–29.
Fraenkel G, Hsiao C. Bursicon, a hormone which mediates tanning of the cuticle in the adult fly and other insects. J. Insect Physiol., 1965,11: 513–556.
Fraenkel G, Hsiao C, Seligman M. Properties of bursicon: an insect protein hormone that controls cuticular tanning. Science, 1966, 151: 91–93.
Giraldez A J, Cohen S M. Wingless and Notch signaling provide cell survival cues and control cell proliferation during wing development. Development, 2003, 130: 6533–6543.
Honegger H W, Market D, Pierce L A, et al. Cellular localization of bursicon using antisera against partial peptide sequences of this insect cuticle-sclerotizing neurohormone. J. Comp. Neurol., 2002, 452: 163–177.
Honegger H W, Dewey E M, Kostron B. From bioassays to Drosophila genetics: strategies for characterizing an essential insect neurohormone, bursicon. Symp. Biol. Hung., 2004, 55: 91–102.
Honegger H W, Dewey E M, Ewer J. Bursicon, the tanning hormone of insects: recent advances following the discovery of its molecular identity. J. Comp. Physiol. A., 2008, 194: 989–1005.
Howard S C, Hester A, Herman K. The Ras/PKA signaling pathway may control RNA polymerase II elongation via the Spt4p/Spt5p complex in Saccharomyces cerevisiae. Genetics, 2003, 165: 1059–1070.
Hsu S Y. New insights into the evolution of the relaxin-LGR signaling system. Trends Endocrinol. Metab., 2003, 7: 303–309.
Hsu S Y, Nakabayashi K, Nishi S, et al. Activation of orphan receptors by the hormone relaxin. Science, 2002, 295: 671–674.
Huang J, Zhang Y, Li M, et al. RNA interference-mediated silencing of the bursicon gene induces defects in wing expansion of silkworm. FEBS Lett., 2007, 581: 697–701.
Jiang C, Baehrecke E H, Thummel C S. Steroid regulated programmed cell death during Drosophila metamorphosis. Development, 1997, 124: 4673–4683.
Johannes B, Preiss A. Wing vein formation in Drosophila melanogaster: Hairless is involved in the cross-talk between Notch and EGF signaling pathways. Mech. Dev., 2002, 115: 3–14.
Johnson S A, Milner M J. The final stages of wing development in Drosophila melanogaster. Tissue Cell, 1987, 19: 505–513.
Kaltenhauser U, Kellermann J, Andersson K, et al. Purifcation and partial characterization of bursicon, a cuticle sclerotizing neuropeptide in insects, from Tenebrio molitor. Insect Biochem. Mol. Biol., 1995, 25: 525–533.
Kiger J A, Natzle J E, Kimbrell D A, Paddy M R, Kleinhesselink K, Green M M. Tissue remodeling during maturation of the Drosophila wing. Dev. Biol., 2007, 301: 178–191.
Kimura K, Kodama A, Hayasaka Y, Ohta T. Activation of the cAMP/PKA signaling pathway is required for post-ecdysial cell death in wing epidermal cells of Drosophila melanogaster. Development, 2004, 131: 1597–1606.
Klein T, Seugnet L, Haenlin M, Martinez Arias A. Two different activities of Suppressor of Hairless during wing development in Drosophila. Development, 2000, 127: 3553–3566.
Kostron B, Marquardt K, Kaltenhauser U, Honegger H. Bursicon, the cuticle sclerotizing hormonecomparison of its molecular mass in different insects. J. Insect Physiol., 1995, 41: 1045–1053.
Kumagai J, Hsu S Y, Matsumi H, et al. INSL3/Leydig insulin-like peptide activates the LGR8 receptor important in testis descent. J. Biol. Chem., 2002, 277: 31283–31286.
Luo C W, Dewey E M, Sudo S, et al. Bursicon, the insect cuticle-hardening hormone, is a heterodimeric cystine knot protein that activates G protein-coupled receptor LGR2. Proc. Natl. Acad. Sci.,2005, 102: 2820–2825.
Mills R R, Lake C R. Hormonal control of tanning in the American cockroach. IV. Preliminary purification of the hormone. J. Insect Physiol., 1966, 12: 1395–1401.
Mills R R, Nielsen D J. Changes in the diuretic and antidiuretic properties of the haemolymph during the six-day vitellogenic cycle in the American cockroach. Gen. Comp. Endocrinol., 1967, 9: 380–382.
Mendive F M, Van Loy T, Claeysen S, et al. Drosophila molting neurohormone bursicon is a heterodimer and the natural agonist of the orphan receptor DLGR2. FEBS Lett., 2005, 579: 2171–2176.
Natzle J E, Kiger J A, Green M M. Bursicon signaling mutations separate the epithelialmesenchymal transition from programmed cell death during Drosophila melanogaster wing maturation. Genetics, 2008, 180: 885–893.
Nishi S, Hsu S Y, Zell K, Hsueh A J. Characterization of two fly LGR (leucine-rich repeatcontaining, G protein-coupled receptor) proteins homologous to vertebrate glycoprotein hormone receptors: constitutive activation of wild-type fly LGR1 but not LGR2 in transfected mammalian cells. Endocrinol., 2000, 141: 4081–4090.
Reynolds S E. Hormonal regulation of cuticle extensibility in newly emerged adult blowflies. J. Insect Physiol., 1976, 22: 529–534.
Reynolds S E. Control of cuticle extensibility in the wings of adult Manduca at the time of eclosion: effects of eclosion hormone and bursicon. J. Exp. Biol., 1977, 70: 27–39.
Reynolds S E. Integration of behavior and physiology in ecdysis. Adv. J. Insect Physiol., 1980, 15: 475–595.
Reynolds S E. Bursicon. // Downer R G H, Laufer H. Endocrinology of Insects. Liss: Alan R. 1983: 235–348.
Schweisguth F. Suppressor of Hairless is required for signal reception during lateral inhibition in Drosophila pupal notum. Development, 1995, 121: 1875–1888.
Schweisguth F, Posakony JW. Antagonistic activities of Suppressor of Hairless and Hairless control alternative cell fates in the Drosophila adult epidermis. Development, 1994, 120: 1433–1441.
Seligman L M, Doy E A, Crossley A C. Hormonal control of morphogenetic cell death of the wing hypodermis in Lucilia cuprina. Tissue Cell, 1975, 7: 281–296.
Seligman M, Doy F A. Studies on cyclic AMP mediation of hormonally induced cytolysis of the alary hypodermal cells and of hormonally controlled dopa synthesis in Lucilia cuprina. Israel J. Ent., 1972, 7: 129–142.
Seligman M, Doy F A. Hormonal regulation of disaggregation of cellular fragments in the haemolymph of Lusilia cuprina. J. Insect Physiol., 1973, 19: 125–135.
Seligman I M. Bursicon. // Miller, T.A. Neurohormonal Techniques in Insects. Berlin: Springer, 1980: 137–153.
Taghert P H, Truman JW. Identification of the bursiconcontaining neurones in abdominal ganglia of the tobacco hornworm, Manduca sexta. J. Exp. Biol.,1982, 98: 385–401.
Truman J W. The eclosion hormone system of insects. Prog. Brain,1992, 92: 361–374.
Van Loy T, Van Hiel M B, Vandersmissen H P, et al. Evolutionary conservation of bursicon in the animal kingdom. Gen. Comp. Endocrinol.,2007, 153: 59–63.
Von Knorre D, Gersch M, Kusch T. Zur Frage der Beeinflussung des ‘tanning’ phanomens durch zyklisches-3′, 5′ AMP. Zool. Jb. (Physiol.), 1972, 76: 434–440.
Wang S, An S, Song Q. Transcriptional expression of bursicon and novel bursicon-regulated genes in the house fly Musca domestica. Arch. Insect Biochem. Physiol., 2008, 68: 100–112.
Wang S J, An S H, Stanley D, Song Q. Cloning and characterization of a bursicon-regulated gene Su (H) in the house fly Musca domestica. Insect Science, 2009, 16: 207–217.
Wesley C S. Notch and Wingless regulate expression of cuticle patterning genes. Mol. Cell Biochem., 1999, 19: 5743–5758.
Zanassi P, Paolillo M, Feliciello A, et al. cAMP-dependent protein kinase induces cAMPresponse element-binding protein phosphorylation via an intracellular calcium release/ERK-dependent pathway in striatal neurons. J. Biol. Chem., 2001, 276: 11487–11495.
Zitnan D, Adams M E. Neuroendocrine Regulation of Insect Ecdysis. // Gilbert, L.I., Iatrou K, S.S., Gill, S.S. Comprehensive Insect Biochemistry, Physiology, Pharmacology, and Molecular Biology, Volume 3. London: Elsevier Press, 2005: 1–60.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Higher Education Press, Beijing and Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Song, Q., An, S. (2011). Bursicon, a Neuropeptide Hormone That Controls Cuticle Tanning and Beyond. In: Liu, T., Kang, L. (eds) Recent Advances in Entomological Research. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-17815-3_8
Download citation
DOI: https://doi.org/10.1007/978-3-642-17815-3_8
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-17814-6
Online ISBN: 978-3-642-17815-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)