Abstract
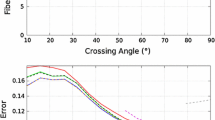
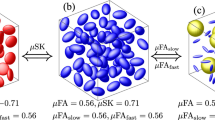
Diffusional Kurtosis Imaging (DKI) is able to capture non-Gaussian diffusion and has become a popular complement to the more traditional Diffusion Tensor Imaging (DTI). In this paper, we demonstrate how strongly the presence of fiber crossings and the exact crossing angle affect measures from diffusional kurtosis, limiting their interpretability as indicators of tissue microstructure. We alleviate this limitation by modeling fiber crossings with a mixture of cylindrically symmetric kurtosis models. Based on results on simulated and on real-world data, we conclude that explicitly including crossing geometry in kurtosis models leads to parameters that are more specific to other aspects of tissue microstructure, such as scale and homogeneity.
Chapter PDF
Similar content being viewed by others
Keywords
- Fractional Anisotropy
- Diffusion Tensor Image
- Bayesian Information Criterion
- Superior Longitudinal Fasciculus
- Fiber Crossing
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Behrens, T.E.J., Johansen-Berg, H., Jbabdi, S., Rushworth, M.F.S., Woolrich, M.W.: Probabilistic diffusion tractography with multiple fibre orientations: What can we gain? NeuroImage 34, 144–155 (2007)
Dell’Acqua, F., Simmons, A., Williams, S.C.R., Catani, M.: Can spherical deconvolution provide more information than fiber orientations? hindrance modulated orientational anisotropy, a true-tract specific index to characterize white matter diffusion. Human Brain Mapping 34(10), 2464–2483 (2013)
Ennis, D.B., Kindlmann, G., Rodriguez, I., Helm, P.A., McVeigh, E.R.: Visualization of tensor fields using superquadric glyphs. Magnetic Resonance in Medicine 53(1), 169–176 (2005)
Jensen, J.H., Helpern, J.A.: MRI quantification of non-gaussian water diffusion by kurtosis analysis. NMR in Biomedicine 23(7), 698–710 (2010)
Kreher, B., Schneider, J., Mader, I., Martin, E., Hennig, J., Il’yasov, K.: Multitensor approach for analysis and tracking of complex fiber configurations. Magnetic Resonance in Medicine 54, 1216–1225 (2005)
Malcolm, J.G., Shenton, M.E., Rathi, Y.: Filtered multitensor tractography. IEEE Trans. on Medical Imaging 29(9), 1664–1675 (2010)
Panagiotaki, E., Schneider, T., Siow, B., Hall, M.G., Lythgoe, M.F., Alexander, D.C.: Compartment models of the diffusion MR signal in brain white matter: A taxonomy and comparison. NeuroImage 59, 2241–2254 (2012)
Raffelt, D., Tournier, J.D., Rose, S., Ridgway, G.R., Henderson, R., Crozier, S., Salvado, O., Connelly, A.: Apparent fibre density: A novel measure for the analysis of diffusion-weighted magnetic resonance images. NeuroImage 59(4), 3976–3994 (2012)
Reisert, M., Kiselev, V.G., Dihtal, B., Kellner, E., Novikov, D.S.: MesoFT: unifying diffusion modelling and fiber tracking. In: Golland, P., Hata, N., Barillot, C., Hornegger, J., Howe, R. (eds.) MICCAI 2014, Part III. LNCS, vol. 8675, pp. 201–208. Springer, Heidelberg (2014)
Schultz, T., Groeschel, S.: Auto-calibrating spherical deconvolution based on ODF sparsity. In: Mori, K., Sakuma, I., Sato, Y., Barillot, C., Navab, N. (eds.) MICCAI 2013, Part I. LNCS, vol. 8149, pp. 663–670. Springer, Heidelberg (2013)
Schultz, T., Westin, C.F., Kindlmann, G.: Multi-diffusion-tensor fitting via spherical deconvolution: A unifying framework. In: Jiang, T., Navab, N., Pluim, J.P.W., Viergever, M.A. (eds.) MICCAI 2010, Part I. LNCS, vol. 6361, pp. 673–680. Springer, Heidelberg (2010)
Tabesh, A., Jensen, J.H., Ardekani, B.A., Helpern, J.A.: Estimation of tensors and tensor-derived measures in diffusional kurtosis imaging. Magnetic Resonance in Medicine 65, 823–836 (2011)
Taquet, M., Scherrer, B., Boumal, N., Macq, B., Warfield, S.K.: Estimation of a multi-fascicle model from single b-value data with a population-informed prior. In: Mori, K., Sakuma, I., Sato, Y., Barillot, C., Navab, N. (eds.) MICCAI 2013, Part I. LNCS, vol. 8149, pp. 695–702. Springer, Heidelberg (2013)
Tournier, J.D., Calamante, F., Connelly, A.: Robust determination of the fibre orientation distribution in diffusion MRI: Non-negativity constrained super-resolved spherical deconvolution. NeuroImage 35, 1459–1472 (2007)
Tuch, D.S., Reese, T.G., Wiegell, M.R., Makris, N., Belliveau, J.W., Wedeen, V.J.: High angular resolution diffusion imaging reveals intravoxel white matter fiber heterogeneity. Magnetic Resonance in Medicine 48, 577–582 (2002)
Zhang, H., Schneider, T., Wheeler-Kingshott, C.A., Alexander, D.C.: NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. NeuroImage 61(4), 1000–1016 (2012)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this paper
Cite this paper
Ankele, M., Schultz, T. (2015). Quantifying Microstructure in Fiber Crossings with Diffusional Kurtosis. In: Navab, N., Hornegger, J., Wells, W., Frangi, A. (eds) Medical Image Computing and Computer-Assisted Intervention -- MICCAI 2015. MICCAI 2015. Lecture Notes in Computer Science(), vol 9349. Springer, Cham. https://doi.org/10.1007/978-3-319-24553-9_19
Download citation
DOI: https://doi.org/10.1007/978-3-319-24553-9_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24552-2
Online ISBN: 978-3-319-24553-9
eBook Packages: Computer ScienceComputer Science (R0)