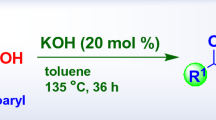
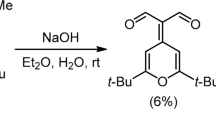
Abstract
Acid-catalyzed rearrangement of tertiary α-acetylenic (terminal) alcohols, leading to the formation of α,β-unsaturated ketones rather than the corresponding α,β- unsaturated aldehydes. Cf. Meyer–Schuster rearrangement.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Rupe, H.; Kambli, E. Helv. Chim. Acta 1926, 9, 672.
Swaminathan, S.; Narayanan, K. V. Chem. Rev. 1971, 71, 429–438. (Review).
Hasbrouck, R. W.; Anderson Kiessling, A. D. J. Org. Chem. 1973, 38, 2103–2106.
Baran, J.; Klein, H.; Schade, C.; Will, E.; Koschinsky, R.; Bäuml, E.; Mayr, H. Tetrahedron 1988, 44, 2181–2184.
Barre, V.; Massias, F.; Uguen, D. Tetrahedron Lett. 1989, 30, 7389–7392.
An, J.; Bagnell, L.; Cablewski, T.; Strauss, C. R.; Trainor, R. W. J. Org. Chem. 1997, 62, 2505–2511.
Yadav, J. S.; Prahlad, V.; Muralidhar, B. Synth. Commun. 1997, 27, 3415–3418.
Takeda, K.; Nakane, D.; Takeda, M. Org. Lett. 2000, 2, 1903–1905.
Weinmann, H.; Harre, M.; Neh, H.; Nickisch, K.; Skötsch, C.; Tilstam, U. Org. Proc. Res. Dev. 2002, 6, 216–219.
Mullins, R. J.; Collins, N. R. Meyer–Schuster Rearrangement. In Name Reactions for Homologations-Part II; Li, J. J., Ed.; Wiley: Hoboken, NJ, 2009, pp 305–318. (Review).
Chang, Y.-J.; Wang, Z.-Z.; Luo, L.-G.; Dai, L.-Y. Chem. Papers 2012, 66, 33–38.
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Li, J.J. (2014). Rupe rearrangement. In: Name Reactions. Springer, Cham. https://doi.org/10.1007/978-3-319-03979-4_239
Download citation
DOI: https://doi.org/10.1007/978-3-319-03979-4_239
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-03978-7
Online ISBN: 978-3-319-03979-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)