Summary
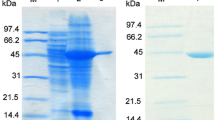
Escherichia coli transformed by a composite plasmid-containing α-amino-ε-caprolactam racemase structural gene, downstream to the tac promoter, showed a high productivity, which made the enzyme purification and crystallization easier. Complete nucleotide sequence was determined by collaboration of shotgun DNA-sequencing and automatic protein sequencing of lysylendopeptidase digest, which revealed that the racemase consists of 435 amino acids.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Ahmed,S.A., Esaki,N., and Soda, K. (1982) FEBS Letters, J50, 370–373.
Fukumura, T. (1977) Agric. Biol. Chem. 41, 1327–1330.
Yanai, A., Nakamura, N., and Oshihara, W. (1985) In: Abstracts of the Ann. Meeting of the American Society for Microbiol, pp 249.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1987 Birkhäuser Verlag Basel
About this chapter
Cite this chapter
Naoko, N., Oshihara, W., Yanai, A. (1987). α-Amino-ε-Caprolactam Racemase for L-Lysine Production. In: Korpela, T.K., Christen, P. (eds) Biochemistry of Vitamin B6 . Birkhäuser Congress Reports. Birkhäuser, Basel. https://doi.org/10.1007/978-3-0348-9308-4_81
Download citation
DOI: https://doi.org/10.1007/978-3-0348-9308-4_81
Publisher Name: Birkhäuser, Basel
Print ISBN: 978-3-0348-9989-5
Online ISBN: 978-3-0348-9308-4
eBook Packages: Springer Book Archive