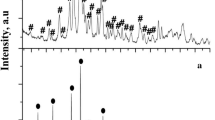
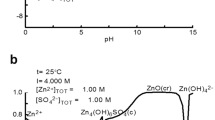
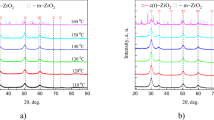
Abstract
The unique low temperature synthesis of zinc pyrovanadate from oxides was proposed. Zn3V2O7(OH)2·2(H2O) was synthesized by ultrasonic (US) method using ZnO and V2O5 as raw materials. It was established using SEM and TEM methods that Zn3V2O7(OH)2·2(H2O) has the structure of nanosheets. The DTA method and XRD analysis showed the formation of the Zn3V2O8 phase after the removal of crystallization water from Zn3V2O7(OH)2·2(H2O). Ultrasonic treatment of oxides as initial reagents allows obtaining Zn3V2O8 with the specific surface area of 14 m2/g. For comparison, Zn3V2O8 was synthesized by solid-state (SS) synthesis from oxides. The properties of zinc pyrovanadate obtained by US synthesis and conventional SS synthesis were compared. The advantages of US method over conventional SS synthesis were noted.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Putluru SSR, Schill L, Godiksen A, Poreddy R, Mossin S, Jensen AD, Fehrmann R (2016) Promoted V2O5/TiO2 catalysts for selective catalytic reduction of NO with NH3 at low temperatures. Appl Catal B 183:282–290. https://doi.org/10.1016/j.apcatb.2015.10.044
Zazhigalov VA, Diyuk EA, Sidorchuk VV (2014) Development of VPO catalysts supported on mesoporous modified material based on an aerosol gel. Kinet Catal 55(3):399–408. https://doi.org/10.1134/S002315840904017X
Pessoa JC, Etcheverry S (2015) Vanadium compounds in medicine. Coordination Chem Rev 301–302:24–48. https://doi.org/10.1016/j.ccr.2014.12.002
Semiz S (2022) Vanadium as potential therapeutic agent for COVID-19: A focus on its antiviral, antiinflamatory, and antihyperglycemic effects. J Trace Elem Med Biol 69:126887. https://doi.org/10.1016/j.jtemb.2021.126887
Qian T, Fan B, Wang H, Zhu S (2019) Structure and luminescence properties of Zn3V2O8 yellow phosphor for white light emitting diodes. Chem Phys Lett 715:34–39. https://doi.org/10.1016/j.cplett.2018.11.022
Gan L, Deng D, Zhang Y, Li G, Wang X, Jiang L, Wang C (2014) Zn3V2O8 hexagon nanosheets: a high-performance anode material for lithium-ion batteries. J Mater Chem A 2:2461–2466. https://doi.org/10.1039/C3TA14242F
Xia C, Guo J, Lei Y, Liang H, Zhao C, Alshareef H (2017) Rechargeable aqueous Zinc-Ion battery based on porous framework Zinc pyrovanadate intercalation cathode. Adv Mater 1705580. https://doi.org/10.1002/adma.201705580
Khallouk K, Solhy A, Kherbeche A, Dubreucq E, Kouisni L, Barakat A (2020) Effective catalytic delignification and fractionation of lignocellulosic biomass in water over Zn3V2O8 mixed oxide. ACS Omega 5(1):304–316. https://doi.org/10.1021/acsomega.9b02159
Khallouk K, Solhy A, Idrissi N, Flaud V, Kherbeche A, Barakat A (2020) Microwave-assisted selective oxidation of sugars to carboxylic acids derivatives in water over zinc-vanadium mixed oxide. Chem Eng J 385:123914. https://doi.org/10.1016/j.cej.2019.123914
Wang D, Tang J, Zou Z, Ye J (2005) Photophysical and photocatalytic properties of a new series of visible-light-driven photocatalysts M3V2O8 (M = Mg, Ni, Zn). Chem Mater 17:5177–5182. https://doi.org/10.1021/cm051016x
Shi R, Wang Y, Zhoub F, Zhu Y (2011) Zn3V2O7(OH)2(H2O)2 and Zn3V2O8 nanostructures: controlled fabrication and photocatalytic performance. J Mater Chem 21:6313–6320. https://doi.org/10.1039/C0JM04451B
Mondal C, Ganguly M, Sinha AK, Pal J, Sahoo R, Pal T (2013) Robust cubooctahedron Zn3V2O8 in gram quantity: a material for photocatalytic dye degradation in water. Cryst Eng Comm 15:6745–6751. https://doi.org/10.1039/C3CE40852C
Liu F, Guan Y, Sun R, Liang X, Sun P, Liu F, Lu G (2015) Mixed potential type acetone sensor using stabilized zirconia and M3V2O8 (M: Zn, Co and Ni) sensing electrode. Sens Actuat B 221:673–680. https://doi.org/10.1016/j.snb.2015.07.023
Hua K, Cui M, Luo Z, Fang D, Bao R, Qi Q, Yi J, Sun B, Chen C (2019) Fabrication of Zinc pyrovanadate (Zn3(OH)2V2O7·2H2O) nanosheet spheres as an ethanol gas sensor. J Alloy Compd 801:581–588. https://doi.org/10.1016/j.jallcom.2019.06.015
Zhang SY, Xiao X, Lu M, Li ZQ (2013) Zn3V2O7(OH)22H2O and Zn3(VO4)2 3D microspheres as anode materials for lithium-ion batteries. J Mater Sci 48:3679–3685. https://doi.org/10.1007/s10853-013-7164-5
Zhang S, Lei N, Ma W, Zhang Z, Sun Z, Wang Y (2014) Fabrication of ultralong Zn3V2O7(OH)22H2O nanobelts and its application in lithium-ion batteries. Mat Lett 129:91–94. https://doi.org/10.1016/j.matlet.2014.05.047
Gopal R, Calvo C (1971) Crystal structure of a-Zn3(VO4)2. Can J Chem 49:3056–3059. https://doi.org/10.1139/v71-510
Clark GM, Pick AN (1975) DTA study of the reactions of V2O5 with metal (II) oxides. J Therm Anal 7:289–300. https://doi.org/10.1007/BF01911939
Nord AG, Stefanidis T (1985) Crystal chemistry of a—(Zn, M)2V2O7 solid solutions correlation between preference for five-coordination and extension of solid solubility. Mat Res Bull 20:845–851. https://doi.org/10.1016/0025-5408(85)90064-9
Kurzawa M, Rychlowska-Himmel I, Bosacka M, Blonska-Tabero A (2001) Reinvestigation of phase equilibria in the V2O5–ZnO system. J Therm Anal Calorim 64:1113–1119. https://doi.org/10.1023/A:1011524424682
Yang G, Li S, Wu M, Wang C (2016) Zinc pyrovanadate nanosheet of atomic thickness: excellent li-storage properties and investigation of electrochemical mechanism. J Mater Chem A 4:10974–10985. https://doi.org/10.1039/C6TA02782B
Mazloom F, Masjedi-Arani M, Salavati-Niasari M (2017) Rapid and solvent-free solid-state synthesis and characterization of Zn3V2O8 nanostructures and their phenol red aqueous solution photodegradation. Solid State Sci 70:101–109. https://doi.org/10.1016/j.solidstatesciences.2017.06.013
Low WH, Khiew PS, Lim SS, Siong CW, Chia CH, Ezeigwe ER (2019) Facile synthesis of graphene-Zn3V2O8 nanocomposite as a high performance electrode material for symmetric supercapacitor. J Alloy Compd 784:847–858. https://doi.org/10.1016/j.jallcom.2019.01.137
Bayat A, Mahjoub AR, Amini MM (2018) Optical and magnetic properties of zinc vanadates: synthetic design of colloidal Zn3V2O7(OH)2(H2O)2, ZnV2O4 and Zn3V2O8 nanostructures. J Mater Sci: Mater Electron 29:2915–2926. https://doi.org/10.1007/s10854-017-8222-6
Diyuk OA, Zazhigalov VA, Shcherban ND, Permyakov VV, Diyuk NV, Shcherbakov SM, Sachuk1 OV, Dulian P (2021) Kinetics of ZnMoO4·0.8H2O and α-ZnMoO4 formation at ultrasonic treatment of ZnO and MoO3 mixture. In: Fesenko O, Yatsenko L (eds) Nanocomposites, nanostructures, and their applications. Springer Proceedings in Physics book series, vol 263, 87–101. https://doi.org/10.1007/978-3-030-74741-1_6
Luo J, Ning X, Zhan L, Zhou X (2021) Facile construction of a fascinating Z-scheme AgI/Zn3V2O8 photocatalyst for the photocatalytic degradation of tetracycline under visible light irradiation. Sep Purif Technol 255:117691. https://doi.org/10.1016/j.seppur.2020.117691
Hoyos DA, Echavarria A, Saldarriaga C (2001) Synthesis and structure of a porous zinc vanadate, Zn3(VO4)2·3H2O. J Mater Sci 36:5515–5518. https://doi.org/10.1023/A:1012418706071
Acknowledgements
This work was financially supported by NASU Program “New functional substances and materials of chemical production” (project 13-21).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Diyuk, O.A. et al. (2023). Ultrasonic Synthesis and Characterization of Zinc Pyrovanadate Nanostructures. In: Fesenko, O., Yatsenko, L. (eds) Nanomaterials and Nanocomposites, Nanostructure Surfaces, and Their Applications . Springer Proceedings in Physics, vol 279. Springer, Cham. https://doi.org/10.1007/978-3-031-18096-5_16
Download citation
DOI: https://doi.org/10.1007/978-3-031-18096-5_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-18095-8
Online ISBN: 978-3-031-18096-5
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)