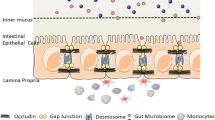
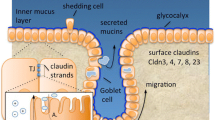
Abstract
The gastrointestinal system digests and absorbs nutrients while integrating exogenous and endogenous signals that contribute to create immune tolerance. Epithelial cells forming the physical barrier regulate ion, solutes, and water absorption in the gut. Tight junctions (TJs) are specialized cell-cell junctions that bring adjacent gastrointestinal epithelial cells together and seal the paracellular space. Additionally, TJs are important for maintaining cellular polarity in epithelial cells and in the gastrointestinal epithelium itself. Dysfunctional gastrointestinal TJ enhances leakage of luminal contents into the interstitium, a process that stimulates the development of inflammatory disorders. Therefore, the pathophysiology of the TJs has gained attention in the medical field. Several studies aimed to investigate the viability of those structures are now used in the diagnosis for patients with chronic gastrointestinal inflammation. This chapter focuses on analyzing the role of the TJs in the gut, the techniques employed to study these structures, as well as various mechanisms responsible for compromising the TJs and the intestinal epithelial barrier.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AJ:
-
Adherens junctions
- AJC:
-
Apical junctional complex
- CK1:
-
Casein kinase 1
- cPKC:
-
Ca2+-dependent protein kinase C
- ECL1:
-
First extracellular loop
- ECL2:
-
Second extracellular loop
- GI:
-
Gastrointestinal
- GuK:
-
Guanylate kinase homology
- IBD:
-
Inflammatory bowel disease
- IEC:
-
Intestinal epithelial cells
- Ig:
-
Immunoglobulin
- IHC:
-
Immunohistochemistry
- IL:
-
Interleukin
- IP:
-
Intestinal permeability
- JAMs:
-
Junctional adhesion molecules
- KO:
-
Knockout
- MUPP1:
-
Multi-PDZ domain protein 1
- PDZ:
-
PSD95, DlgA, ZO1 homology
- PKA:
-
Protein kinase A
- QIIME:
-
Quantitative Insights Into Microbial Ecology
- SH3:
-
Non-catalytic SRC homology 3
- TJs:
-
Tight junctions
- ZO:
-
Zonula occludens
References
Kiela, P. R., & Ghishan, F. K. (2016). Physiology of Intestinal Absorption and Secretion. Best Practice & Research. Clinical Gastroenterology, 30(2), 145-159. https://doi.org/10.1016/j.bpg.2016.02.007
Cheng, L. K., O’Grady, G., Du, P., Egbuji, J. U., Windsor, J. A., & Pullan, A. J. (2010). Gastrointestinal system. Wiley Interdisciplinary Reviews. Systems Biology and Medicine, 2(1), 65-79. https://doi.org/10.1002/wsbm.19
Kararli, T. T. (1995). Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharmaceutics & Drug Disposition, 16(5), 351-380. https://doi.org/10.1002/bdd.2510160502
Cereijido, M., Contreras, R. G., & Shoshani, L. (2004). Cell adhesion, polarity, and epithelia in the dawn of metazoans. Physiological Reviews, 84(4), 1229-1262. https://doi.org/10.1152/physrev.00001.2004
Farquhar, M. G., & Palade, G. E. (1963). Junctional complexes in various epithelia. The Journal of Cell Biology, 17, 375-412. https://doi.org/10.1083/jcb.17.2.375
Gumber, S., Nusrat, A., & Villinger, F. (2014). Immunohistological characterization of intercellular junction proteins in rhesus macaque intestine. Experimental and Toxicologic Pathology: Official Journal of the Gesellschaft Fur Toxikologische Pathologie, 66(9-10), 437-444. https://doi.org/10.1016/j.etp.2014.07.004
Madara, J. L., Nash, S., Moore, R., & Atisook, K. (1990). Structure and function of the intestinal epithelial barrier in health and disease. Monographs in Pathology, 31, 306-324.
Furuse, M., Hirase, T., Itoh, M., Nagafuchi, A., Yonemura, S., Tsukita, S., & Tsukita, S. (1993). Occludin: A novel integral membrane protein localizing at tight junctions. The Journal of Cell Biology, 123(6 Pt 2), 1777-1788. https://doi.org/10.1083/jcb.123.6.1777
Tsukita, S., & Furuse, M. (2000). Pores in the wall: Claudins constitute tight junction strands containing aqueous pores. The Journal of Cell Biology, 149(1), 13-16. https://doi.org/10.1083/jcb.149.1.13
Tsukita, S., Furuse, M., & Itoh, M. (2001). Multifunctional strands in tight junctions. Nature Reviews. Molecular Cell Biology, 2(4), 285-293. https://doi.org/10.1038/35067088
Martini, E., Krug, S. M., Siegmund, B., Neurath, M. F., & Becker, C. (2017). Mend Your Fences: The Epithelial Barrier and its Relationship With Mucosal Immunity in Inflammatory Bowel Disease. Cellular and Molecular Gastroenterology and Hepatology, 4(1), 33-46. https://doi.org/10.1016/j.jcmgh.2017.03.007
Somsouk, M., Estes, J. D., Deleage, C., Dunham, R. M., Albright, R., Inadomi, J. M., Martin, J. N., Deeks, S. G., McCune, J. M., & Hunt, P. W. (2015). Gut epithelial barrier and systemic inflammation during chronic HIV infection. AIDS (London, England), 29(1), 43-51. https://doi.org/10.1097/QAD.0000000000000511
Turner, J. R. (2009). Intestinal mucosal barrier function in health and disease. Nature Reviews. Immunology, 9(11), 799-809. https://doi.org/10.1038/nri2653
Crosnier, C., Stamataki, D., & Lewis, J. (2006). Organizing cell renewal in the intestine: Stem cells, signals and combinatorial control. Nature Reviews. Genetics, 7(5), 349-359. https://doi.org/10.1038/nrg1840
Rees, W. D., Tandun, R., Yau, E., Zachos, N. C., & Steiner, T. S. (2020). Regenerative Intestinal Stem Cells Induced by Acute and Chronic Injury: The Saving Grace of the Epithelium? Frontiers in Cell and Developmental Biology, 8, 583919. https://doi.org/10.3389/fcell.2020.583919
Sato, T., Vries, R. G., Snippert, H. J., van de Wetering, M., Barker, N., Stange, D. E., van Es, J. H., Abo, A., Kujala, P., Peters, P. J., & Clevers, H. (2009). Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature, 459(7244), 262-265. https://doi.org/10.1038/nature07935
Camilleri, M., Madsen, K., Spiller, R., Greenwood-Van Meerveld, B., Van Meerveld, B. G., & Verne, G. N. (2012). Intestinal barrier function in health and gastrointestinal disease. Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 24(6), 503-512. https://doi.org/10.1111/j.1365-2982.2012.01921.x
Bischoff, S. C., Barbara, G., Buurman, W., Ockhuizen, T., Schulzke, J.-D., Serino, M., Tilg, H., Watson, A., & Wells, J. M. (2014). Intestinal permeability—A new target for disease prevention and therapy. BMC Gastroenterology, 14, 189. https://doi.org/10.1186/s12876-014-0189-7
Schulzke, J. D., Schulzke, I., Fromm, M., & Riecken, E. O. (1995). Epithelial barrier and ion transport in coeliac sprue: Electrical measurements on intestinal aspiration biopsy specimens. Gut, 37(6), 777-782. https://doi.org/10.1136/gut.37.6.777
Karasov, W. H. (2017). Integrative physiology of transcellular and paracellular intestinal absorption. The Journal of Experimental Biology, 220(Pt 14), 2495-2501. https://doi.org/10.1242/jeb.144048
Camilleri, M., Nadeau, A., Lamsam, J., Nord, S. L., Ryks, M., Burton, D., Sweetser, S., Zinsmeister, A. R., & Singh, R. (2010). Understanding measurements of intestinal permeability in healthy humans with urine lactulose and mannitol excretion. Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 22(1), e15-26. https://doi.org/10.1111/j.1365-2982.2009.01361.x
Fung, K. Y. Y., Fairn, G. D., & Lee, W. L. (2018). Transcellular vesicular transport in epithelial and endothelial cells: Challenges and opportunities. Traffic (Copenhagen, Denmark), 19(1), 5-18. https://doi.org/10.1111/tra.12533
Kellett, G. L., Brot-Laroche, E., Mace, O. J., & Leturque, A. (2008). Sugar absorption in the intestine: The role of GLUT2. Annual Review of Nutrition, 28, 35-54. https://doi.org/10.1146/annurev.nutr.28.061807.155518
Ménard, S., Cerf-Bensussan, N., & Heyman, M. (2010). Multiple facets of intestinal permeability and epithelial handling of dietary antigens. Mucosal Immunology, 3(3), 247-259. https://doi.org/10.1038/mi.2010.5
New, R. (2020). Oral Delivery of Biologics via the Intestine. Pharmaceutics, 13(1), E18. https://doi.org/10.3390/pharmaceutics13010018
Sesorova, I. S., Dimov, I. D., Kashin, A. D., Sesorov, V. V., Karelina, N. R., Zdorikova, M. A., Beznoussenko, G. V., & Mirоnоv, A. A. (2021). Cellular and sub-cellular mechanisms of lipid transport from gut to lymph. Tissue & Cell, 72, 101529. https://doi.org/10.1016/j.tice.2021.101529
Tamura, A., & Tsukita, S. (2014). Paracellular barrier and channel functions of TJ claudins in organizing biological systems: Advances in the field of barriology revealed in knockout mice. Seminars in Cell & Developmental Biology, 36, 177-185. https://doi.org/10.1016/j.semcdb.2014.09.019
Tsukita, S., Tanaka, H., & Tamura, A. (2019b). The Claudins: From Tight Junctions to Biological Systems. Trends in Biochemical Sciences, 44(2), 141-152. https://doi.org/10.1016/j.tibs.2018.09.008
González-Mariscal, L., Betanzos, A., Nava, P., & Jaramillo, B. E. (2003). Tight junction proteins. Progress in Biophysics and Molecular Biology, 81(1), 1-44. https://doi.org/10.1016/s0079-6107(02)00037-8
Kostrewa, D., Brockhaus, M., D’Arcy, A., Dale, G. E., Nelboeck, P., Schmid, G., Mueller, F., Bazzoni, G., Dejana, E., Bartfai, T., Winkler, F. K., & Hennig, M. (2001). X-ray structure of junctional adhesion molecule: Structural basis for homophilic adhesion via a novel dimerization motif. The EMBO Journal, 20(16), 4391-4398. https://doi.org/10.1093/emboj/20.16.4391
Krause, G., Winkler, L., Mueller, S. L., Haseloff, R. F., Piontek, J., & Blasig, I. E. (2008). Structure and function of claudins. Biochimica Et Biophysica Acta, 1778(3), 631-645. https://doi.org/10.1016/j.bbamem.2007.10.018
González-Mariscal, L., Betanzos, A., & Avila-Flores, A. (2000). MAGUK proteins: Structure and role in the tight junction. Seminars in Cell & Developmental Biology, 11(4), 315-324. https://doi.org/10.1006/scdb.2000.0178
Otani, T., & Furuse, M. (2020). Tight Junction Structure and Function Revisited. Trends in Cell Biology, 30(10), 805-817. https://doi.org/10.1016/j.tcb.2020.08.004
Citi, S. (2019). The mechanobiology of tight junctions. Biophysical Reviews, 11(5), 783-793. https://doi.org/10.1007/s12551-019-00582-7
Mitic, L. L., & Anderson, J. M. (1998). Molecular architecture of tight junctions. Annual Review of Physiology, 60, 121-142. https://doi.org/10.1146/annurev.physiol.60.1.121
Rodgers, L. S., & Fanning, A. S. (2011). Regulation of epithelial permeability by the actin cytoskeleton. Cytoskeleton (Hoboken, N.J.), 68(12), 653-660. https://doi.org/10.1002/cm.20547
Heinemann, U., & Schuetz, A. (2019). Structural Features of Tight-Junction Proteins. International Journal of Molecular Sciences, 20(23), E6020. https://doi.org/10.3390/ijms20236020
Morita, K., Furuse, M., Fujimoto, K., & Tsukita, S. (1999). Claudin multigene family encoding four-transmembrane domain protein components of tight junction strands. Proceedings of the National Academy of Sciences of the United States of America, 96(2), 511-516. https://doi.org/10.1073/pnas.96.2.511
Daugherty, B. L., Ward, C., Smith, T., Ritzenthaler, J. D., & Koval, M. (2007). Regulation of heterotypic claudin compatibility. The Journal of Biological Chemistry, 282(41), 30005-30013. https://doi.org/10.1074/jbc.M703547200
Piontek, J., Winkler, L., Wolburg, H., Müller, S. L., Zuleger, N., Piehl, C., Wiesner, B., Krause, G., & Blasig, I. E. (2008). Formation of tight junction: Determinants of homophilic interaction between classic claudins. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, 22(1), 146-158. https://doi.org/10.1096/fj.07-8319com
Colegio, O. R., Van Itallie, C., Rahner, C., & Anderson, J. M. (2003). Claudin extracellular domains determine paracellular charge selectivity and resistance but not tight junction fibril architecture. American Journal of Physiology. Cell Physiology, 284(6), C1346-1354. https://doi.org/10.1152/ajpcell.00547.2002
Suzuki, H., Nishizawa, T., Tani, K., Yamazaki, Y., Tamura, A., Ishitani, R., Dohmae, N., Tsukita, S., Nureki, O., & Fujiyoshi, Y. (2014). Crystal structure of a claudin provides insight into the architecture of tight junctions. Science (New York, N.Y.), 344(6181), 304-307. https://doi.org/10.1126/science.1248571
Reiche, J., & Huber, O. (2020). Post-translational modifications of tight junction transmembrane proteins and their direct effect on barrier function. Biochimica Et Biophysica Acta. Biomembranes, 1862(9), 183330. https://doi.org/10.1016/j.bbamem.2020.183330
Van Itallie, C. M., Gambling, T. M., Carson, J. L., & Anderson, J. M. (2005). Palmitoylation of claudins is required for efficient tight-junction localization. Journal of Cell Science, 118(Pt 7), 1427-1436. https://doi.org/10.1242/jcs.01735
Van Itallie, C. M., Mitic, L. L., & Anderson, J. M. (2012). SUMOylation of claudin-2. Annals of the New York Academy of Sciences, 1258, 60-64. https://doi.org/10.1111/j.1749-6632.2012.06541.x
Barmeyer, C., Schulzke, J. D., & Fromm, M. (2015). Claudin-related intestinal diseases. Seminars in Cell & Developmental Biology, 42, 30-38. https://doi.org/10.1016/j.semcdb.2015.05.006
Fujita, H., Chiba, H., Yokozaki, H., Sakai, N., Sugimoto, K., Wada, T., Kojima, T., Yamashita, T., & Sawada, N. (2006). Differential expression and subcellular localization of claudin-7, -8, -12, -13, and -15 along the mouse intestine. The Journal of Histochemistry and Cytochemistry: Official Journal of the Histochemistry Society, 54(8), 933-944. https://doi.org/10.1369/jhc.6A6944.2006
Garcia-Hernandez, V., Quiros, M., & Nusrat, A. (2017). Intestinal epithelial claudins: Expression and regulation in homeostasis and inflammation. Annals of the New York Academy of Sciences, 1397(1), 66-79. https://doi.org/10.1111/nyas.13360
Holmes, J. L., Van Itallie, C. M., Rasmussen, J. E., & Anderson, J. M. (2006). Claudin profiling in the mouse during postnatal intestinal development and along the gastrointestinal tract reveals complex expression patterns. Gene Expression Patterns: GEP, 6(6), 581-588. https://doi.org/10.1016/j.modgep.2005.12.001
Lameris, A. L., Huybers, S., Kaukinen, K., Mäkelä, T. H., Bindels, R. J., Hoenderop, J. G., & Nevalainen, P. I. (2013). Expression profiling of claudins in the human gastrointestinal tract in health and during inflammatory bowel disease. Scandinavian Journal of Gastroenterology, 48(1), 58-69. https://doi.org/10.3109/00365521.2012.741616
Nakagawa, S., Miyoshi, N., Ishii, H., Mimori, K., Tanaka, F., Sekimoto, M., Doki, Y., & Mori, M. (2011). Expression of CLDN1 in colorectal cancer: A novel marker for prognosis. International Journal of Oncology, 39(4), 791-796. https://doi.org/10.3892/ijo.2011.1102
Yoshida, T., Kinugasa, T., Akagi, Y., Kawahara, A., Romeo, K., Shiratsuchi, I., Ryu, Y., Gotanda, Y., & Shirouzu, K. (2011). Decreased expression of claudin-1 in rectal cancer: A factor for recurrence and poor prognosis. Anticancer Research, 31(7), 2517-2525.
Curry, J. N., Saurette, M., Askari, M., Pei, L., Filla, M. B., Beggs, M. R., Rowe, P. S., Fields, T., Sommer, A. J., Tanikawa, C., Kamatani, Y., Evan, A. P., Totonchi, M., Alexander, R. T., Matsuda, K., & Yu, A. S. (2020). Claudin-2 deficiency associates with hypercalciuria in mice and human kidney stone disease. The Journal of Clinical Investigation, 130(4), 1948-1960. https://doi.org/10.1172/JCI127750
Raju, P., Shashikanth, N., Tsai, P.-Y., Pongkorpsakol, P., Chanez-Paredes, S., Steinhagen, P. R., Kuo, W.-T., Singh, G., Tsukita, S., & Turner, J. R. (2020). Inactivation of paracellular cation-selective claudin-2 channels attenuates immune-mediated experimental colitis in mice. The Journal of Clinical Investigation, 130(10), 5197-5208. https://doi.org/10.1172/JCI138697
Xing, T., Camacho Salazar, R., & Chen, Y.-H. (2017). Animal models for studying epithelial barriers in neonatal necrotizing enterocolitis, inflammatory bowel disease and colorectal cancer. Tissue Barriers, 5(4), e1356901. https://doi.org/10.1080/21688370.2017.1356901
Ding, L., Lu, Z., Foreman, O., Tatum, R., Lu, Q., Renegar, R., Cao, J., & Chen, Y.-H. (2012). Inflammation and disruption of the mucosal architecture in claudin-7-deficient mice. Gastroenterology, 142(2), 305-315. https://doi.org/10.1053/j.gastro.2011.10.025
Tabariès, S., & Siegel, P. M. (2017). The role of claudins in cancer metastasis. Oncogene, 36(9), 1176-1190. https://doi.org/10.1038/onc.2016.289
Tamura, A., Hayashi, H., Imasato, M., Yamazaki, Y., Hagiwara, A., Wada, M., Noda, T., Watanabe, M., Suzuki, Y., & Tsukita, S. (2011). Loss of claudin-15, but not claudin-2, causes Na+ deficiency and glucose malabsorption in mouse small intestine. Gastroenterology, 140(3), 913-923. https://doi.org/10.1053/j.gastro.2010.08.006
Maryan, N., Statkiewicz, M., Mikula, M., Goryca, K., Paziewska, A., Strzałkowska, A., Dabrowska, M., Bujko, M., & Ostrowski, J. (2015). Regulation of the expression of claudin 23 by the enhancer of zeste 2 polycomb group protein in colorectal cancer. Molecular Medicine Reports, 12(1), 728-736. https://doi.org/10.3892/mmr.2015.3378
Tanaka, H., Takechi, M., Kiyonari, H., Shioi, G., Tamura, A., & Tsukita, S. (2015). Intestinal deletion of Claudin-7 enhances paracellular organic solute flux and initiates colonic inflammation in mice. Gut, 64(10), 1529-1538. https://doi.org/10.1136/gutjnl-2014-308419
Fujita, H., Sugimoto, K., Inatomi, S., Maeda, T., Osanai, M., Uchiyama, Y., Yamamoto, Y., Wada, T., Kojima, T., Yokozaki, H., Yamashita, T., Kato, S., Sawada, N., & Chiba, H. (2008). Tight junction proteins claudin-2 and -12 are critical for vitamin D-dependent Ca2+ absorption between enterocytes. Molecular Biology of the Cell, 19(5), 1912-1921. https://doi.org/10.1091/mbc.e07-09-0973
Van Itallie, C. M., Fanning, A. S., & Anderson, J. M. (2003). Reversal of charge selectivity in cation or anion-selective epithelial lines by expression of different claudins. American Journal of Physiology. Renal Physiology, 285(6), F1078-1084. https://doi.org/10.1152/ajprenal.00116.2003
Cummins, P. M. (2012). Occludin: One protein, many forms. Molecular and Cellular Biology, 32(2), 242-250. https://doi.org/10.1128/MCB.06029-11
Feldman, G. J., Mullin, J. M., & Ryan, M. P. (2005). Occludin: Structure, function and regulation. Advanced Drug Delivery Reviews, 57(6), 883-917. https://doi.org/10.1016/j.addr.2005.01.009
Medina, R., Rahner, C., Mitic, L. L., Anderson, J. M., & Van Itallie, C. M. (2000). Occludin localization at the tight junction requires the second extracellular loop. The Journal of Membrane Biology, 178(3), 235-247. https://doi.org/10.1007/s002320010031
Chen, Y., Merzdorf, C., Paul, D. L., & Goodenough, D. A. (1997). COOH terminus of occludin is required for tight junction barrier function in early Xenopus embryos. The Journal of Cell Biology, 138(4), 891-899. https://doi.org/10.1083/jcb.138.4.891
Furuse, M., Itoh, M., Hirase, T., Nagafuchi, A., Yonemura, S., Tsukita, S., & Tsukita, S. (1994). Direct association of occludin with ZO-1 and its possible involvement in the localization of occludin at tight junctions. The Journal of Cell Biology, 127(6 Pt 1), 1617-1626. https://doi.org/10.1083/jcb.127.6.1617
Bamforth, S. D., Kniesel, U., Wolburg, H., Engelhardt, B., & Risau, W. (1999). A dominant mutant of occludin disrupts tight junction structure and function. Journal of Cell Science, 112 ( Pt 12), 1879-1888.
Muresan, Z., Paul, D. L., & Goodenough, D. A. (2000). Occludin 1B, a variant of the tight junction protein occludin. Molecular Biology of the Cell, 11(2), 627-634. https://doi.org/10.1091/mbc.11.2.627
Al-Sadi, R., Khatib, K., Guo, S., Ye, D., Youssef, M., & Ma, T. (2011). Occludin regulates macromolecule flux across the intestinal epithelial tight junction barrier. American Journal of Physiology. Gastrointestinal and Liver Physiology, 300(6), G1054-1064. https://doi.org/10.1152/ajpgi.00055.2011
Kyoko, O., Kono, H., Ishimaru, K., Miyake, K., Kubota, T., Ogawa, H., Okumura, K., Shibata, S., & Nakao, A. (2014). Expressions of tight junction proteins Occludin and Claudin-1 are under the circadian control in the mouse large intestine: Implications in intestinal permeability and susceptibility to colitis. PloS One, 9(5), e98016. https://doi.org/10.1371/journal.pone.0098016
Dörfel, M. J., Westphal, J. K., Bellmann, C., Krug, S. M., Cording, J., Mittag, S., Tauber, R., Fromm, M., Blasig, I. E., & Huber, O. (2013). CK2-dependent phosphorylation of occludin regulates the interaction with ZO-proteins and tight junction integrity. Cell Communication and Signaling: CCS, 11(1), 40. https://doi.org/10.1186/1478-811X-11-40
Kuo, W.-T., Shen, L., Zuo, L., Shashikanth, N., Ong, M. L. D. M., Wu, L., Zha, J., Edelblum, K. L., Wang, Y., Wang, Y., Nilsen, S. P., & Turner, J. R. (2019). Inflammation-induced Occludin Downregulation Limits Epithelial Apoptosis by Suppressing Caspase-3 Expression. Gastroenterology, 157(5), 1323-1337. https://doi.org/10.1053/j.gastro.2019.07.058
Hanning, N., Edwinson, A. L., Ceuleers, H., Peters, S. A., De Man, J. G., Hassett, L. C., De Winter, B. Y., & Grover, M. (2021). Intestinal barrier dysfunction in irritable bowel syndrome: A systematic review. Therapeutic Advances in Gastroenterology, 14, 1756284821993586. https://doi.org/10.1177/1756284821993586
Martínez, C., Lobo, B., Pigrau, M., Ramos, L., González-Castro, A. M., Alonso, C., Guilarte, M., Guilá, M., de Torres, I., Azpiroz, F., Santos, J., & Vicario, M. (2013). Diarrhoea-predominant irritable bowel syndrome: An organic disorder with structural abnormalities in the jejunal epithelial barrier. Gut, 62(8), 1160-1168. https://doi.org/10.1136/gutjnl-2012-302093
Annaházi, A., Ferrier, L., Bézirard, V., Lévêque, M., Eutamène, H., Ait-Belgnaoui, A., Coëffier, M., Ducrotté, P., Róka, R., Inczefi, O., Gecse, K., Rosztóczy, A., Molnár, T., Ringel-Kulka, T., Ringel, Y., Piche, T., Theodorou, V., Wittmann, T., & Bueno, L. (2013). Luminal cysteine-proteases degrade colonic tight junction structure and are responsible for abdominal pain in constipation-predominant IBS. The American Journal of Gastroenterology, 108(8), 1322-1331. https://doi.org/10.1038/ajg.2013.152
Oshitani, N., Watanabe, K., Nakamura, S., Fujiwara, Y., Higuchi, K., & Arakawa, T. (2005). Dislocation of tight junction proteins without F-actin disruption in inactive Crohn’s disease. International Journal of Molecular Medicine, 15(3), 407-410.
Yamamoto-Furusho, J. K., Mendivil, E. J., Mendivil-Rangel, E. J., & Fonseca-Camarillo, G. (2012). Differential expression of occludin in patients with ulcerative colitis and healthy controls. Inflammatory Bowel Diseases, 18(10), E1999. https://doi.org/10.1002/ibd.22835
Zeissig, S., Bürgel, N., Günzel, D., Richter, J., Mankertz, J., Wahnschaffe, U., Kroesen, A. J., Zeitz, M., Fromm, M., & Schulzke, J.-D. (2007). Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut, 56(1), 61-72. https://doi.org/10.1136/gut.2006.094375
Bürgel, N., Bojarski, C., Mankertz, J., Zeitz, M., Fromm, M., & Schulzke, J.-D. (2002). Mechanisms of diarrhea in collagenous colitis. Gastroenterology, 123(2), 433-443. https://doi.org/10.1053/gast.2002.34784
Krug, S. M., Schulzke, J. D., & Fromm, M. (2014). Tight junction, selective permeability, and related diseases. Seminars in Cell & Developmental Biology, 36, 166-176. https://doi.org/10.1016/j.semcdb.2014.09.002
Shen, L., & Turner, J. R. (2005). Actin depolymerization disrupts tight junctions via caveolae-mediated endocytosis. Molecular Biology of the Cell, 16(9), 3919-3936. https://doi.org/10.1091/mbc.e04-12-1089
Utech, M., Ivanov, A. I., Samarin, S. N., Bruewer, M., Turner, J. R., Mrsny, R. J., Parkos, C. A., & Nusrat, A. (2005). Mechanism of IFN-gamma-induced endocytosis of tight junction proteins: Myosin II-dependent vacuolarization of the apical plasma membrane. Molecular Biology of the Cell, 16(10), 5040-5052. https://doi.org/10.1091/mbc.e05-03-0193
Youakim, A., & Ahdieh, M. (1999). Interferon-gamma decreases barrier function in T84 cells by reducing ZO-1 levels and disrupting apical actin. The American Journal of Physiology, 276(5), G1279-1288. https://doi.org/10.1152/ajpgi.1999.276.5.G1279
Liu, H., Li, M., Wang, P., & Wang, F. (2011). Blockade of hypoxia-inducible factor-1α by YC-1 attenuates interferon-γ and tumor necrosis factor-α-induced intestinal epithelial barrier dysfunction. Cytokine, 56(3), 581-588. https://doi.org/10.1016/j.cyto.2011.08.023
Ogawa, M., Osada, H., Hasegawa, A., Ohno, H., Yanuma, N., Sasaki, K., Shimoda, M., Shirai, J., Kondo, H., & Ohmori, K. (2018). Effect of interleukin-1β on occludin mRNA expression in the duodenal and colonic mucosa of dogs with inflammatory bowel disease. Journal of Veterinary Internal Medicine, 32(3), 1019-1025. https://doi.org/10.1111/jvim.15117
Wang, X., Zeng, H.-C., Huang, Y.-R., & He, Q.-Z. (2020b). Chlamydia muridarum Alleviates Colitis via the IL-22/Occludin Signal Pathway. BioMed Research International, 2020, 8894331. https://doi.org/10.1155/2020/8894331
Wiley, J. W., Zong, Y., Zheng, G., Zhu, S., & Hong, S. (2020). Histone H3K9 methylation regulates chronic stress and IL-6-induced colon epithelial permeability and visceral pain. Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 32(12), e13941. https://doi.org/10.1111/nmo.13941
Minagar, A., Long, A., Ma, T., Jackson, T. H., Kelley, R. E., Ostanin, D. V., Sasaki, M., Warren, A. C., Jawahar, A., Cappell, B., & Alexander, J. S. (2003). Interferon (IFN)-beta 1a and IFN-beta 1b block IFN-gamma-induced disintegration of endothelial junction integrity and barrier. Endothelium: Journal of Endothelial Cell Research, 10(6), 299-307. https://doi.org/10.1080/10623320390272299
Lee, J. S., Tato, C. M., Joyce-Shaikh, B., Gulen, M. F., Cayatte, C., Chen, Y., Blumenschein, W. M., Judo, M., Ayanoglu, G., McClanahan, T. K., Li, X., & Cua, D. J. (2015). Interleukin-23-Independent IL-17 Production Regulates Intestinal Epithelial Permeability. Immunity, 43(4), 727-738. https://doi.org/10.1016/j.immuni.2015.09.003
Díaz-Coránguez, M., Liu, X., & Antonetti, D. A. (2019). Tight Junctions in Cell Proliferation. International Journal of Molecular Sciences, 20(23), E5972. https://doi.org/10.3390/ijms20235972
Yaffe, Y., Shepshelovitch, J., Nevo-Yassaf, I., Yeheskel, A., Shmerling, H., Kwiatek, J. M., Gaus, K., Pasmanik-Chor, M., & Hirschberg, K. (2012). The MARVEL transmembrane motif of occludin mediates oligomerization and targeting to the basolateral surface in epithelia. Journal of Cell Science, 125(Pt 15), 3545-3556. https://doi.org/10.1242/jcs.100289
Raleigh, D. R., Marchiando, A. M., Zhang, Y., Shen, L., Sasaki, H., Wang, Y., Long, M., & Turner, J. R. (2010). Tight junction-associated MARVEL proteins marveld3, tricellulin, and occludin have distinct but overlapping functions. Molecular Biology of the Cell, 21(7), 1200-1213. https://doi.org/10.1091/mbc.e09-08-0734
Bauer, H., Stelzhammer, W., Fuchs, R., Weiger, T. M., Danninger, C., Probst, G., & Krizbai, I. A. (1999). Astrocytes and neurons express the tight junction-specific protein occludin in vitro. Experimental Cell Research, 250(2), 434-438. https://doi.org/10.1006/excr.1999.4558
Castro, V., Skowronska, M., Lombardi, J., He, J., Seth, N., Velichkovska, M., & Toborek, M. (2018). Occludin regulates glucose uptake and ATP production in pericytes by influencing AMP-activated protein kinase activity. Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 38(2), 317-332. https://doi.org/10.1177/0271678X17720816
Morgan, S. V., Garwood, C. J., Jennings, L., Simpson, J. E., Castelli, L. M., Heath, P. R., Mihaylov, S. R., Vaquéz-Villaseñor, I., Minshull, T. C., Ince, P. G., Dickman, M. J., Hautbergue, G. M., & Wharton, S. B. (2018). Proteomic and cellular localisation studies suggest non-tight junction cytoplasmic and nuclear roles for occludin in astrocytes. The European Journal of Neuroscience, 47(12), 1444-1456. https://doi.org/10.1111/ejn.13933
Rescigno, M., Urbano, M., Valzasina, B., Francolini, M., Rotta, G., Bonasio, R., Granucci, F., Kraehenbuhl, J. P., & Ricciardi-Castagnoli, P. (2001). Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunology, 2(4), 361-367. https://doi.org/10.1038/86373
Saitou, M., Furuse, M., Sasaki, H., Schulzke, J. D., Fromm, M., Takano, H., Noda, T., & Tsukita, S. (2000). Complex phenotype of mice lacking occludin, a component of tight junction strands. Molecular Biology of the Cell, 11(12), 4131-4142. https://doi.org/10.1091/mbc.11.12.4131
Castro, V., Bertrand, L., Luethen, M., Dabrowski, S., Lombardi, J., Morgan, L., Sharova, N., Stevenson, M., Blasig, I. E., & Toborek, M. (2016). Occludin controls HIV transcription in brain pericytes via regulation of SIRT-1 activation. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, 30(3), 1234-1246. https://doi.org/10.1096/fj.15-277673
Martìn-Padura, I., Lostaglio, S., Schneemann, M., Williams, L., Romano, M., Fruscella, P., Panzeri, C., Stoppacciaro, A., Ruco, L., Villa, A., Simmons, D., & Dejana, E. (1998). Junctional adhesion molecule, a novel member of the immunoglobulin superfamily that distributes at intercellular junctions and modulates monocyte transmigration. The Journal of Cell Biology, 142(1), 117-127. https://doi.org/10.1083/jcb.142.1.117
Ebnet, K., Suzuki, A., Ohno, S., & Vestweber, D. (2004). Junctional adhesion molecules (JAMs): More molecules with dual functions? Journal of Cell Science, 117(Pt 1), 19-29. https://doi.org/10.1242/jcs.00930
Hartmann, C., Schwietzer, Y. A., Otani, T., Furuse, M., & Ebnet, K. (2020). Physiological functions of junctional adhesion molecules (JAMs) in tight junctions. Biochimica Et Biophysica Acta. Biomembranes, 1862(9), 183299. https://doi.org/10.1016/j.bbamem.2020.183299
Luissint, A.-C., Nusrat, A., & Parkos, C. A. (2014). JAM-related proteins in mucosal homeostasis and inflammation. Seminars in Immunopathology, 36(2), 211-226. https://doi.org/10.1007/s00281-014-0421-0
Mandell, K. J., & Parkos, C. A. (2005). The JAM family of proteins. Advanced Drug Delivery Reviews, 57(6), 857-867. https://doi.org/10.1016/j.addr.2005.01.005
Steinbacher, T., Kummer, D., & Ebnet, K. (2018). Junctional adhesion molecule-A: Functional diversity through molecular promiscuity. Cellular and Molecular Life Sciences: CMLS, 75(8), 1393-1409. https://doi.org/10.1007/s00018-017-2729-0
Bazzoni, G., Martinez-Estrada, O. M., Orsenigo, F., Cordenonsi, M., Citi, S., & Dejana, E. (2000). Interaction of junctional adhesion molecule with the tight junction components ZO-1, cingulin, and occludin. The Journal of Biological Chemistry, 275(27), 20520-20526. https://doi.org/10.1074/jbc.M905251199
Monteiro, A. C., Sumagin, R., Rankin, C. R., Leoni, G., Mina, M. J., Reiter, D. M., Stehle, T., Dermody, T. S., Schaefer, S. A., Hall, R. A., Nusrat, A., & Parkos, C. A. (2013). JAM-A associates with ZO-2, afadin, and PDZ-GEF1 to activate Rap2c and regulate epithelial barrier function. Molecular Biology of the Cell, 24(18), 2849-2860. https://doi.org/10.1091/mbc.E13-06-0298
Van Itallie, C. M., & Anderson, J. M. (2018). Phosphorylation of tight junction transmembrane proteins: Many sites, much to do. Tissue Barriers, 6(1), e1382671. https://doi.org/10.1080/21688370.2017.1382671
Iden, S., Misselwitz, S., Peddibhotla, S. S. D., Tuncay, H., Rehder, D., Gerke, V., Robenek, H., Suzuki, A., & Ebnet, K. (2012). APKC phosphorylates JAM-A at Ser285 to promote cell contact maturation and tight junction formation. The Journal of Cell Biology, 196(5), 623-639. https://doi.org/10.1083/jcb.201104143
Braiterman, L. T., Heffernan, S., Nyasae, L., Johns, D., See, A. P., Yutzy, R., McNickle, A., Herman, M., Sharma, A., Naik, U. P., & Hubbard, A. L. (2008). JAM-A is both essential and inhibitory to development of hepatic polarity in WIF-B cells. American Journal of Physiology. Gastrointestinal and Liver Physiology, 294(2), G576-588. https://doi.org/10.1152/ajpgi.00159.2007
Naik, M. U., Caplan, J. L., & Naik, U. P. (2014). Junctional adhesion molecule-A suppresses platelet integrin αIIbβ3 signaling by recruiting Csk to the integrin-c-Src complex. Blood, 123(9), 1393-1402. https://doi.org/10.1182/blood-2013-04-496232
Naik, M. U., Mousa, S. A., Parkos, C. A., & Naik, U. P. (2003). Signaling through JAM-1 and alphavbeta3 is required for the angiogenic action of bFGF: Dissociation of the JAM-1 and alphavbeta3 complex. Blood, 102(6), 2108-2114. https://doi.org/10.1182/blood-2003-04-1114
Mandicourt, G., Iden, S., Ebnet, K., Aurrand-Lions, M., & Imhof, B. A. (2007). JAM-C regulates tight junctions and integrin-mediated cell adhesion and migration. The Journal of Biological Chemistry, 282(3), 1830-1837. https://doi.org/10.1074/jbc.M605666200
Morton, P. E., Hicks, A., Nastos, T., Santis, G., & Parsons, M. (2013). CAR regulates epithelial cell junction stability through control of E-cadherin trafficking. Scientific Reports, 3, 2889. https://doi.org/10.1038/srep02889
Nunbhakdi-Craig, V., Machleidt, T., Ogris, E., Bellotto, D., White, C. L., & Sontag, E. (2002). Protein phosphatase 2A associates with and regulates atypical PKC and the epithelial tight junction complex. The Journal of Cell Biology, 158(5), 967-978. https://doi.org/10.1083/jcb.200206114
Fan, S., Weight, C. M., Luissint, A.-C., Hilgarth, R. S., Brazil, J. C., Ettel, M., Nusrat, A., & Parkos, C. A. (2019). Role of JAM-A tyrosine phosphorylation in epithelial barrier dysfunction during intestinal inflammation. Molecular Biology of the Cell, 30(5), 566-578. https://doi.org/10.1091/mbc.E18-08-0531
Shawki, A., & McCole, D. F. (2017). Mechanisms of Intestinal Epithelial Barrier Dysfunction by Adherent-Invasive Escherichia coli. Cellular and Molecular Gastroenterology and Hepatology, 3(1), 41-50. https://doi.org/10.1016/j.jcmgh.2016.10.004
Stevenson, B. R., Siliciano, J. D., Mooseker, M. S., & Goodenough, D. A. (1986). Identification of ZO-1: A high molecular weight polypeptide associated with the tight junction (zonula occludens) in a variety of epithelia. The Journal of Cell Biology, 103(3), 755-766. https://doi.org/10.1083/jcb.103.3.755
Jesaitis, L. A., & Goodenough, D. A. (1994). Molecular characterization and tissue distribution of ZO-2, a tight junction protein homologous to ZO-1 and the Drosophila discs-large tumor suppressor protein. The Journal of Cell Biology, 124(6), 949-961. https://doi.org/10.1083/jcb.124.6.949
Haskins, J., Gu, L., Wittchen, E. S., Hibbard, J., & Stevenson, B. R. (1998). ZO-3, a novel member of the MAGUK protein family found at the tight junction, interacts with ZO-1 and occludin. The Journal of Cell Biology, 141(1), 199-208. https://doi.org/10.1083/jcb.141.1.199
Bauer, H., Zweimueller-Mayer, J., Steinbacher, P., Lametschwandtner, A., & Bauer, H. C. (2010). The dual role of zonula occludens (ZO) proteins. Journal of Biomedicine & Biotechnology, 2010, 402593. https://doi.org/10.1155/2010/402593
Rodgers, L. S., Beam, M. T., Anderson, J. M., & Fanning, A. S. (2013). Epithelial barrier assembly requires coordinated activity of multiple domains of the tight junction protein ZO-1. Journal of Cell Science, 126(Pt 7), 1565-1575. https://doi.org/10.1242/jcs.113399
Suzuki, T. (2013). Regulation of intestinal epithelial permeability by tight junctions. Cellular and Molecular Life Sciences: CMLS, 70(4), 631-659. https://doi.org/10.1007/s00018-012-1070-x
Umeda, K., Ikenouchi, J., Katahira-Tayama, S., Furuse, K., Sasaki, H., Nakayama, M., Matsui, T., Tsukita, S., Furuse, M., & Tsukita, S. (2006). ZO-1 and ZO-2 independently determine where claudins are polymerized in tight-junction strand formation. Cell, 126(4), 741-754. https://doi.org/10.1016/j.cell.2006.06.043
Capaldo, C. T., Koch, S., Kwon, M., Laur, O., Parkos, C. A., & Nusrat, A. (2011). Tight function zonula occludens-3 regulates cyclin D1-dependent cell proliferation. Molecular Biology of the Cell, 22(10), 1677-1685. https://doi.org/10.1091/mbc.E10-08-0677
Garcia, M. A., Nelson, W. J., & Chavez, N. (2018). Cell-Cell Junctions Organize Structural and Signaling Networks. Cold Spring Harbor Perspectives in Biology, 10(4), a029181. https://doi.org/10.1101/cshperspect.a029181
Das, P., Goswami, P., Das, T. K., Nag, T., Sreenivas, V., Ahuja, V., Panda, S. K., Gupta, S. D., & Makharia, G. K. (2012). Comparative tight junction protein expressions in colonic Crohn’s disease, ulcerative colitis, and tuberculosis: A new perspective. Virchows Archiv: An International Journal of Pathology, 460(3), 261-270. https://doi.org/10.1007/s00428-012-1195-1
Poritz, L. S., Garver, K. I., Green, C., Fitzpatrick, L., Ruggiero, F., & Koltun, W. A. (2007). Loss of the tight junction protein ZO-1 in dextran sulfate sodium induced colitis. The Journal of Surgical Research, 140(1), 12-19. https://doi.org/10.1016/j.jss.2006.07.050
Han, X., Ren, X., Jurickova, I., Groschwitz, K., Pasternak, B. A., Xu, H., Wilson, T. A., Hogan, S. P., & Denson, L. A. (2009). Regulation of intestinal barrier function by signal transducer and activator of transcription 5b. Gut, 58(1), 49-58. https://doi.org/10.1136/gut.2007.145094
Landy, J., Ronde, E., English, N., Clark, S. K., Hart, A. L., Knight, S. C., Ciclitira, P. J., & Al-Hassi, H. O. (2016). Tight junctions in inflammatory bowel diseases and inflammatory bowel disease associated colorectal cancer. World Journal of Gastroenterology, 22(11), 3117-3126. https://doi.org/10.3748/wjg.v22.i11.3117
Prot-Bertoye, C., & Houillier, P. (2020). Claudins in Renal Physiology and Pathology. Genes, 11(3), E290. https://doi.org/10.3390/genes11030290
Kachar, B., & Reese, T. S. (1982). Evidence for the lipidic nature of tight junction strands. Nature, 296(5856), 464-466. https://doi.org/10.1038/296464a0
Nusrat, A., Parkos, C. A., Verkade, P., Foley, C. S., Liang, T. W., Innis-Whitehouse, W., Eastburn, K. K., & Madara, J. L. (2000). Tight junctions are membrane microdomains. Journal of Cell Science, 113 ( Pt 10), 1771-1781.
Pinto da Silva, P., & Kachar, B. (1982). On tight-junction structure. Cell, 28(3), 441-450. https://doi.org/10.1016/0092-8674(82)90198-2
Dragsten, P. R., Blumenthal, R., & Handler, J. S. (1981). Membrane asymmetry in epithelia: Is the tight junction a barrier to diffusion in the plasma membrane? Nature, 294(5843), 718-722. https://doi.org/10.1038/294718a0
van Meer, G., & Simons, K. (1986). The function of tight junctions in maintaining differences in lipid composition between the apical and the basolateral cell surface domains of MDCK cells. The EMBO Journal, 5(7), 1455-1464.
Cao, X., Surma, M. A., & Simons, K. (2012). Polarized sorting and trafficking in epithelial cells. Cell Research, 22(5), 793-805. https://doi.org/10.1038/cr.2012.64
Seo, K., Seo, J., Yeun, J., Choi, H., Kim, Y.-I., & Chang, S.-Y. (2021). The role of mucosal barriers in human gut health. Archives of Pharmacal Research, 44(4), 325-341. https://doi.org/10.1007/s12272-021-01327-5
Camilleri, M. (2019). Leaky gut: Mechanisms, measurement and clinical implications in humans. Gut, 68(8), 1516-1526. https://doi.org/10.1136/gutjnl-2019-318427
Muehler, A., Slizgi, J. R., Kohlhof, H., Groeppel, M., Peelen, E., & Vitt, D. (2020). Clinical relevance of intestinal barrier dysfunction in common gastrointestinal diseases. World Journal of Gastrointestinal Pathophysiology, 11(6), 114-130. https://doi.org/10.4291/wjgp.v11.i6.114
Piche, T. (2014). Tight junctions and IBS--the link between epithelial permeability, low-grade inflammation, and symptom generation? Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 26(3), 296-302. https://doi.org/10.1111/nmo.12315
Brescia, P., & Rescigno, M. (2021). The gut vascular barrier: A new player in the gut-liver-brain axis. Trends in Molecular Medicine, S1471-4914(21)00154-4. https://doi.org/10.1016/j.molmed.2021.06.007
Mu, Q., Kirby, J., Reilly, C. M., & Luo, X. M. (2017). Leaky Gut As a Danger Signal for Autoimmune Diseases. Frontiers in Immunology, 8, 598. https://doi.org/10.3389/fimmu.2017.00598
Mg, L., Porfirio, N., & Asma, N. (2008). Role of the intestinal barrier in inflammatory bowel disease. World Journal of Gastroenterology, 14(3), 401-407. https://doi.org/10.3748/wjg.14.401
Huang, L., Jiang, Y., Sun, Z., Gao, Z., Wang, J., & Zhang, D. (2018). Autophagy Strengthens Intestinal Mucosal Barrier by Attenuating Oxidative Stress in Severe Acute Pancreatitis. Digestive Diseases and Sciences, 63(4), 910-919. https://doi.org/10.1007/s10620-018-4962-2
Ferdinande, L., Demetter, P., Perez-Novo, C., Waeytens, A., Taildeman, J., Rottiers, I., Rottiers, P., De Vos, M., & Cuvelier, C. A. (2008). Inflamed intestinal mucosa features a specific epithelial expression pattern of indoleamine 2,3-dioxygenase. International Journal of Immunopathology and Pharmacology, 21(2), 289-295. https://doi.org/10.1177/039463200802100205
Chelakkot, C., Ghim, J., & Ryu, S. H. (2018). Mechanisms regulating intestinal barrier integrity and its pathological implications. Experimental & Molecular Medicine, 50(8), 1-9. https://doi.org/10.1038/s12276-018-0126-x
Ferrucci, L., & Fabbri, E. (2018). Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nature Reviews. Cardiology, 15(9), 505-522. https://doi.org/10.1038/s41569-018-0064-2
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C., & Santoro, A. (2018). Inflammaging: A new immune-metabolic viewpoint for age-related diseases. Nature Reviews. Endocrinology, 14(10), 576-590. https://doi.org/10.1038/s41574-018-0059-4
Johnson, A. M., & Loftus, E. V. (2021). Obesity in inflammatory bowel disease: A review of its role in the pathogenesis, natural history, and treatment of IBD. Saudi Journal of Gastroenterology: Official Journal of the Saudi Gastroenterology Association. https://doi.org/10.4103/sjg.sjg_30_21
Zhu, X., Li, B., Lou, P., Dai, T., Chen, Y., Zhuge, A., Yuan, Y., & Li, L. (2021). The Relationship Between the Gut Microbiome and Neurodegenerative Diseases. Neuroscience Bulletin. https://doi.org/10.1007/s12264-021-00730-8
Allaire, J. M., Crowley, S. M., Law, H. T., Chang, S.-Y., Ko, H.-J., & Vallance, B. A. (2018). The Intestinal Epithelium: Central Coordinator of Mucosal Immunity. Trends in Immunology, 39(9), 677-696. https://doi.org/10.1016/j.it.2018.04.002
Biagi, E., Nylund, L., Candela, M., Ostan, R., Bucci, L., Pini, E., Nikkïla, J., Monti, D., Satokari, R., Franceschi, C., Brigidi, P., & De Vos, W. (2010). Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PloS One, 5(5), e10667. https://doi.org/10.1371/journal.pone.0010667
Neurath, M. F. (2014). Cytokines in inflammatory bowel disease. Nature Reviews. Immunology, 14(5), 329-342. https://doi.org/10.1038/nri3661
Berkes, J., Viswanathan, V. K., Savkovic, S. D., & Hecht, G. (2003). Intestinal epithelial responses to enteric pathogens: Effects on the tight junction barrier, ion transport, and inflammation. Gut, 52(3), 439-451. https://doi.org/10.1136/gut.52.3.439
Paradis, T., Bègue, H., Basmaciyan, L., Dalle, F., & Bon, F. (2021). Tight Junctions as a Key for Pathogens Invasion in Intestinal Epithelial Cells. International Journal of Molecular Sciences, 22(5), 2506. https://doi.org/10.3390/ijms22052506
Caron, E., Crepin, V. F., Simpson, N., Knutton, S., Garmendia, J., & Frankel, G. (2006). Subversion of actin dynamics by EPEC and EHEC. Current Opinion in Microbiology, 9(1), 40-45. https://doi.org/10.1016/j.mib.2005.12.008
Knutton, S., Lloyd, D. R., & McNeish, A. S. (1987). Adhesion of enteropathogenic Escherichia coli to human intestinal enterocytes and cultured human intestinal mucosa. Infection and Immunity, 55(1), 69-77. https://doi.org/10.1128/iai.55.1.69-77.1987
Ottlinger, M. E., & Lin, S. (1988). Clostridium difficile toxin B induces reorganization of actin, vinculin, and talin in cultured cells. Experimental Cell Research, 174(1), 215-229. https://doi.org/10.1016/0014-4827(88)90156-5
Booth, B. A., Boesman-Finkelstein, M., & Finkelstein, R. A. (1984). Vibrio cholerae hemagglutinin/protease nicks cholera enterotoxin. Infection and Immunity, 45(3), 558-560. https://doi.org/10.1128/iai.45.3.558-560.1984
Moncrief, J. S., Obiso, R., Barroso, L. A., Kling, J. J., Wright, R. L., Van Tassell, R. L., Lyerly, D. M., & Wilkins, T. D. (1995). The enterotoxin of Bacteroides fragilis is a metalloprotease. Infection and Immunity, 63(1), 175-181. https://doi.org/10.1128/iai.63.1.175-181.1995
Myers, L. L., Shoop, D. S., Stackhouse, L. L., Newman, F. S., Flaherty, R. J., Letson, G. W., & Sack, R. B. (1987). Isolation of enterotoxigenic Bacteroides fragilis from humans with diarrhea. Journal of Clinical Microbiology, 25(12), 2330-2333. https://doi.org/10.1128/jcm.25.12.2330-2333.1987
Freedman, J. C., Shrestha, A., & McClane, B. A. (2016). Clostridium perfringens Enterotoxin: Action, Genetics, and Translational Applications. Toxins, 8(3), E73. https://doi.org/10.3390/toxins8030073
Simonovic, I., Rosenberg, J., Koutsouris, A., & Hecht, G. (2000). Enteropathogenic Escherichia coli dephosphorylates and dissociates occludin from intestinal epithelial tight junctions. Cellular Microbiology, 2(4), 305-315. https://doi.org/10.1046/j.1462-5822.2000.00055.x
Viswanathan, V. K., Koutsouris, A., Lukic, S., Pilkinton, M., Simonovic, I., Simonovic, M., & Hecht, G. (2004). Comparative Analysis of EspF from Enteropathogenic and Enterohemorrhagic Escherichia coli in Alteration of Epithelial Barrier Function. Infection and Immunity, 72(6), 3218-3227. https://doi.org/10.1128/IAI.72.6.3218-3227.2004
Muza-Moons, M. M., Schneeberger, E. E., & Hecht, G. A. (2004). Enteropathogenic Escherichia coli infection leads to appearance of aberrant tight junctions strands in the lateral membrane of intestinal epithelial cells. Cellular Microbiology, 6(8), 783-793. https://doi.org/10.1111/j.1462-5822.2004.00404.x
Campbell, J. A., Schelling, P., Wetzel, J. D., Johnson, E. M., Forrest, J. C., Wilson, G. A. R., Aurrand-Lions, M., Imhof, B. A., Stehle, T., & Dermody, T. S. (2005). Junctional Adhesion Molecule A Serves as a Receptor for Prototype and Field-Isolate Strains of Mammalian Reovirus. Journal of Virology, 79(13), 7967-7978. https://doi.org/10.1128/JVI.79.13.7967-7978.2005
Cohen, C. J., Shieh, J. T., Pickles, R. J., Okegawa, T., Hsieh, J. T., & Bergelson, J. M. (2001). The coxsackievirus and adenovirus receptor is a transmembrane component of the tight junction. Proceedings of the National Academy of Sciences of the United States of America, 98(26), 15191-15196. https://doi.org/10.1073/pnas.261452898
Nava, P., López, S., Arias, C. F., Islas, S., & González-Mariscal, L. (2004). The rotavirus surface protein VP8 modulates the gate and fence function of tight junctions in epithelial cells. Journal of Cell Science, 117(Pt 23), 5509-5519. https://doi.org/10.1242/jcs.01425
Guttman, J. A., & Finlay, B. B. (2009). Tight junctions as targets of infectious agents Biochimica et Biophysica Acta (BBA) - Biomembranes, 1788(4), 832-841. https://doi.org/10.1016/j.bbamem.2008.10.028
Simonovic, I., Arpin, M., Koutsouris, A., Falk-Krzesinski, H. J., & Hecht, G. (2001). Enteropathogenic Escherichia coli Activates Ezrin, Which Participates in Disruption of Tight Junction Barrier Function. Infection and Immunity, 69(9), 5679-5688. https://doi.org/10.1128/IAI.69.9.5679-5688.2001
Gasaly, N., Hermoso, M. A., & Gotteland, M. (2021). Butyrate and the Fine-Tuning of Colonic Homeostasis: Implication for Inflammatory Bowel Diseases. International Journal of Molecular Sciences, 22(6), 3061. https://doi.org/10.3390/ijms22063061
Geirnaert, A., Calatayud, M., Grootaert, C., Laukens, D., Devriese, S., Smagghe, G., De Vos, M., Boon, N., & Van de Wiele, T. (2017). Butyrate-producing bacteria supplemented in vitro to Crohn’s disease patient microbiota increased butyrate production and enhanced intestinal epithelial barrier integrity. Scientific Reports, 7(1), 11450. https://doi.org/10.1038/s41598-017-11734-8
Kumari, R., Ahuja, V., & Paul, J. (2013). Fluctuations in butyrate-producing bacteria in ulcerative colitis patients of North India. World Journal of Gastroenterology, 19(22), 3404-3414. https://doi.org/10.3748/wjg.v19.i22.3404
Louis, P., Young, P., Holtrop, G., & Flint, H. J. (2010). Diversity of human colonic butyrate-producing bacteria revealed by analysis of the butyryl-CoA:acetate CoA-transferase gene. Environmental Microbiology, 12(2), 304-314. https://doi.org/10.1111/j.1462-2920.2009.02066.x
Machiels, K., Joossens, M., Sabino, J., De Preter, V., Arijs, I., Eeckhaut, V., Ballet, V., Claes, K., Van Immerseel, F., Verbeke, K., Ferrante, M., Verhaegen, J., Rutgeerts, P., & Vermeire, S. (2014). A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut, 63(8), 1275-1283. https://doi.org/10.1136/gutjnl-2013-304833
Van den Abbeele, P., Belzer, C., Goossens, M., Kleerebezem, M., De Vos, W. M., Thas, O., De Weirdt, R., Kerckhof, F.-M., & Van de Wiele, T. (2013). Butyrate-producing Clostridium cluster XIVa species specifically colonize mucins in an in vitro gut model. The ISME Journal, 7(5), 949-961. https://doi.org/10.1038/ismej.2012.158
Wang, R. X., Lee, J. S., Campbell, E. L., & Colgan, S. P. (2020a). Microbiota-derived butyrate dynamically regulates intestinal homeostasis through regulation of actin-associated protein synaptopodin. Proceedings of the National Academy of Sciences of the United States of America, 117(21), 11648-11657. https://doi.org/10.1073/pnas.1917597117
Bach Knudsen, K. E., Lærke, H. N., Hedemann, M. S., Nielsen, T. S., Ingerslev, A. K., Gundelund Nielsen, D. S., Theil, P. K., Purup, S., Hald, S., Schioldan, A. G., Marco, M. L., Gregersen, S., & Hermansen, K. (2018). Impact of Diet-Modulated Butyrate Production on Intestinal Barrier Function and Inflammation. Nutrients, 10(10), 1499. https://doi.org/10.3390/nu10101499
Coppola, S., Avagliano, C., Calignano, A., & Berni Canani, R. (2021). The Protective Role of Butyrate against Obesity and Obesity-Related Diseases. Molecules, 26(3), 682. https://doi.org/10.3390/molecules26030682
McNabney, S. M., & Henagan, T. M. (2017). Short Chain Fatty Acids in the Colon and Peripheral Tissues: A Focus on Butyrate, Colon Cancer, Obesity and Insulin Resistance. Nutrients, 9(12), 1348. https://doi.org/10.3390/nu9121348
Stoeva, M. K., Garcia-So, J., Justice, N., Myers, J., Tyagi, S., Nemchek, M., McMurdie, P. J., Kolterman, O., & Eid, J.. (2021). Butyrate-producing human gut symbiont, Clostridium butyricum, and its role in health and disease. Gut Microbes, 13(1), 1907272. https://doi.org/10.1080/19490976.2021.1907272
Dinan, T. G., & Cryan, J. F. (2013). Melancholic microbes: A link between gut microbiota and depression? Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 25(9), 713-719. https://doi.org/10.1111/nmo.12198
Finegold, S. M., Dowd, S. E., Gontcharova, V., Liu, C., Henley, K. E., Wolcott, R. D., Youn, E., Summanen, P. H., Granpeesheh, D., Dixon, D., Liu, M., Molitoris, D. R., & Green, J. A. (2010). Pyrosequencing study of fecal microflora of autistic and control children. Anaerobe, 16(4), 444-453. https://doi.org/10.1016/j.anaerobe.2010.06.008
Larsen, N., Vogensen, F. K., van den Berg, F. W. J., Nielsen, D. S., Andreasen, A. S., Pedersen, B. K., Al-Soud, W. A., Sørensen, S. J., Hansen, L. H., & Jakobsen, M. (2010). Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PloS One, 5(2), e9085. https://doi.org/10.1371/journal.pone.0009085
Matt, S. M., Allen, J. M., Lawson, M. A., Mailing, L. J., Woods, J. A., & Johnson, R. W. (2018). Butyrate and Dietary Soluble Fiber Improve Neuroinflammation Associated With Aging in Mice. Frontiers in Immunology, 9, 1832. https://doi.org/10.3389/fimmu.2018.01832
Casén, C., Vebø, H. C., Sekelja, M., Hegge, F. T., Karlsson, M. K., Ciemniejewska, E., Dzankovic, S., Frøyland, C., Nestestog, R., Engstrand, L., Munkholm, P., Nielsen, O. H., Rogler, G., Simrén, M., Öhman, L., Vatn, M. H., & Rudi, K. (2015). Deviations in human gut microbiota: A novel diagnostic test for determining dysbiosis in patients with IBS or IBD. Alimentary Pharmacology & Therapeutics, 42(1), 71-83. https://doi.org/10.1111/apt.13236
Frank, D. N., St Amand, A. L., Feldman, R. A., Boedeker, E. C., Harpaz, N., & Pace, N. R. (2007). Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proceedings of the National Academy of Sciences of the United States of America, 104(34), 13780-13785. https://doi.org/10.1073/pnas.0706625104
Swidsinski, A., Loening-Baucke, V., Vaneechoutte, M., & Doerffel, Y. (2008). Active Crohn’s disease and ulcerative colitis can be specifically diagnosed and monitored based on the biostructure of the fecal flora. Inflammatory Bowel Diseases, 14(2), 147-161. https://doi.org/10.1002/ibd.20330
Teh, J. J., Berendsen, E. M., Hoedt, E. C., Kang, S., Zhang, J., Zhang, F., Liu, Q., Hamilton, A. L., Wilson-O’Brien, A., Ching, J., Sung, J. J. Y., Yu, J., Ng, S. C., Kamm, M. A., & Morrison, M. (2021). Novel strain-level resolution of Crohn’s disease mucosa-associated microbiota via an ex vivo combination of microbe culture and metagenomic sequencing. The ISME Journal. https://doi.org/10.1038/s41396-021-00991-1
Aghapour, M., Raee, P., Moghaddam, S. J., Hiemstra, P. S., & Heijink, I. H. (2018). Airway Epithelial Barrier Dysfunction in Chronic Obstructive Pulmonary Disease: Role of Cigarette Smoke Exposure. American Journal of Respiratory Cell and Molecular Biology, 58(2), 157-169. https://doi.org/10.1165/rcmb.2017-0200TR
Andrews, C., McLean, M. H., & Durum, S. K. (2018). Cytokine Tuning of Intestinal Epithelial Function. Frontiers in Immunology, 9, 1270. https://doi.org/10.3389/fimmu.2018.01270
Andriopoulou, P., Navarro, P., Zanetti, A., Lampugnani, M. G., & Dejana, E. (1999). Histamine induces tyrosine phosphorylation of endothelial cell-to-cell adherens junctions. Arteriosclerosis, Thrombosis, and Vascular Biology, 19(10), 2286-2297. https://doi.org/10.1161/01.atv.19.10.2286
Bayarri, M. A., Milara, J., Estornut, C., & Cortijo, J. (2021). Nitric Oxide System and Bronchial Epithelium: More Than a Barrier. Frontiers in Physiology, 12, 687381. https://doi.org/10.3389/fphys.2021.687381
Bazarganipour, S., Hausmann, J., Oertel, S., El-Hindi, K., Brachtendorf, S., Blumenstein, I., Kubesch, A., Sprinzl, K., Birod, K., Hahnefeld, L., Trautmann, S., Thomas, D., Herrmann, E., Geisslinger, G., Schiffmann, S., & Grösch, S. (2019). The Lipid Status in Patients with Ulcerative Colitis: Sphingolipids are Disease-Dependent Regulated. Journal of Clinical Medicine, 8(7), E971. https://doi.org/10.3390/jcm8070971
Bowie, R. V., Donatello, S., Lyes, C., Owens, M. B., Babina, I. S., Hudson, L., Walsh, S. V., O’Donoghue, D. P., Amu, S., Barry, S. P., Fallon, P. G., & Hopkins, A. M. (2012). Lipid rafts are disrupted in mildly inflamed intestinal microenvironments without overt disruption of the epithelial barrier. American Journal of Physiology. Gastrointestinal and Liver Physiology, 302(8), G781-793. https://doi.org/10.1152/ajpgi.00002.2011
El-Salhy, M., Solomon, T., Hausken, T., Gilja, O. H., & Hatlebakk, J. G. (2017). Gastrointestinal neuroendocrine peptides/amines in inflammatory bowel disease. World Journal of Gastroenterology, 23(28), 5068-5085. https://doi.org/10.3748/wjg.v23.i28.5068
Kessler, E. C., Wall, S. K., Hernandez, L. L., Gross, J. J., & Bruckmaier, R. M. (2019). Short communication: Mammary gland tight junction permeability after parturition is greater in dairy cows with elevated circulating serotonin concentrations. Journal of Dairy Science, 102(2), 1768-1774. https://doi.org/10.3168/jds.2018-15543
Kulkarni, N., Pathak, M., & Lal, G. (2017). Role of chemokine receptors and intestinal epithelial cells in the mucosal inflammation and tolerance. Journal of Leukocyte Biology, 101(2), 377-394. https://doi.org/10.1189/jlb.1RU0716-327R
Luissint, A.-C., Parkos, C. A., & Nusrat, A. (2016). Inflammation and the Intestinal Barrier: Leukocyte-Epithelial Cell Interactions, Cell Junction Remodeling, and Mucosal Repair. Gastroenterology, 151(4), 616-632. https://doi.org/10.1053/j.gastro.2016.07.008
Massironi, S., Zilli, A., Cavalcoli, F., Conte, D., & Peracchi, M. (2016). Chromogranin A and other enteroendocrine markers in inflammatory bowel disease. Neuropeptides, 58, 127-134. https://doi.org/10.1016/j.npep.2016.01.002
Mourad, F. H., Barada, K. A., & Saade, N. E. (2017). Impairment of Small Intestinal Function in Ulcerative Colitis: Role of Enteric Innervation. Journal of Crohn’s & Colitis, 11(3), 369-377. https://doi.org/10.1093/ecco-jcc/jjw162
Zimmerman, N. P., Vongsa, R. A., Wendt, M. K., & Dwinell, M. B. (2008). Chemokines and chemokine receptors in mucosal homeostasis at the intestinal epithelial barrier in inflammatory bowel disease. Inflammatory Bowel Diseases, 14(7), 1000-1011. https://doi.org/10.1002/ibd.20480
Kominsky, D. J., Campbell, E. L., Ehrentraut, S. F., Wilson, K. E., Kelly, C. J., Glover, L. E., Collins, C. B., Bayless, A. J., Saeedi, B., Dobrinskikh, E., Bowers, B. E., MacManus, C. F., Müller, W., Colgan, S. P., & Bruder, D. (2014). IFN-γ-mediated induction of an apical IL-10 receptor on polarized intestinal epithelia. Journal of Immunology (Baltimore, Md.: 1950), 192(3), 1267-1276. https://doi.org/10.4049/jimmunol.1301757
Lee, J., Gonzales-Navajas, J. M., & Raz, E. (2008). The «polarizing-tolerizing» mechanism of intestinal epithelium: Its relevance to colonic homeostasis. Seminars in Immunopathology, 30(1), 3-9. https://doi.org/10.1007/s00281-007-0099-7
Serrano, C., Galán, S., Rubio, J. F., Candelario-Martínez, A., Montes-Gómez, A. E., Chánez-Paredes, S., Cedillo-Barrón, L., Schnoor, M., Meraz-Ríos, M. A., Villegas-Sepúlveda, N., Ortiz-Navarrete, V., & Nava, P. (2019). Compartmentalized Response of IL-6/STAT3 Signaling in the Colonic Mucosa Mediates Colitis Development. Journal of Immunology (Baltimore, Md.: 1950), 202(4), 1239-1249. https://doi.org/10.4049/jimmunol.1801060
Piedra-Quintero, Z. L., Serrano, C., Villegas-Sepúlveda, N., Maravillas-Montero, J. L., Romero-Ramírez, S., Shibayama, M., Medina-Contreras, O., Nava, P., & Santos-Argumedo, L. (2018). Myosin 1F Regulates M1-Polarization by Stimulating Intercellular Adhesion in Macrophages. Frontiers in Immunology, 9, 3118. https://doi.org/10.3389/fimmu.2018.03118
Suzuki, T., Yoshinaga, N., & Tanabe, S. (2011). Interleukin-6 (IL-6) regulates claudin-2 expression and tight junction permeability in intestinal epithelium. The Journal of Biological Chemistry, 286(36), 31263-31271. https://doi.org/10.1074/jbc.M111.238147
Bertheloot, D., Latz, E., & Franklin, B. S. (2021). Necroptosis, pyroptosis and apoptosis: An intricate game of cell death. Cellular and Molecular Immunology, 18(5), 1106-1121. https://doi.org/10.1038/s41423-020-00630-3
Castro-Martinez, F., Candelario-Martinez, A., Encarnacion-Garcia, M. D. R., Piedra-Quintero, Z., Bonilla-Moreno, R., Betanzos, A., Perez-Orozco, R., Hernandez-Cueto, M. de L. A., Muñoz-Medina, J. E., Patiño-Lopez, G., Schnoor, M., Villegas-Sepulveda, N., & Nava, P. (2021). Rictor/Mammalian Target of Rapamycin Complex 2 Signaling Protects Colonocytes from Apoptosis and Prevents Epithelial Barrier Breakdown. The American Journal of Pathology, S0002-9440(21)00259-5. https://doi.org/10.1016/j.ajpath.2021.06.004
Günther, C., Martini, E., Wittkopf, N., Amann, K., Weigmann, B., Neumann, H., Waldner, M. J., Hedrick, S. M., Tenzer, S., Neurath, M. F., & Becker, C. (2011). Caspase-8 regulates TNF-α-induced epithelial necroptosis and terminal ileitis. Nature, 477(7364), 335-339. https://doi.org/10.1038/nature10400
Piedra-Quintero, Z. L., Wilson, Z., Nava, P., & Guerau-de-Arellano, M. (2020). CD38: An Immunomodulatory Molecule in Inflammation and Autoimmunity. Frontiers in Immunology, 11, 597959. https://doi.org/10.3389/fimmu.2020.597959
Tanzer, M. C., Frauenstein, A., Stafford, C. A., Phulphagar, K., Mann, M., & Meissner, F. (2020). Quantitative and Dynamic Catalogs of Proteins Released during Apoptotic and Necroptotic Cell Death. Cell Reports, 30(4), 1260-1270.e5. https://doi.org/10.1016/j.celrep.2019.12.079
Garside, P. (1999). Cytokines in experimental colitis. Clinical and Experimental Immunology, 118(3), 337-339. https://doi.org/10.1046/j.1365-2249.1999.01088.x
Bhat, A. A., Uppada, S., Achkar, I. W., Hashem, S., Yadav, S. K., Shanmugakonar, M., Al-Naemi, H. A., Haris, M., & Uddin, S. (2018). Tight Junction Proteins and Signaling Pathways in Cancer and Inflammation: A Functional Crosstalk. Frontiers in Physiology, 9, 1942. https://doi.org/10.3389/fphys.2018.01942
Balda, M. S., & Matter, K. (2009). Tight junctions and the regulation of gene expression. Biochimica Et Biophysica Acta, 1788(4), 761-767. https://doi.org/10.1016/j.bbamem.2008.11.024
Mankertz, J., Hillenbrand, B., Tavalali, S., Huber, O., Fromm, M., & Schulzke, J. D. (2004). Functional crosstalk between Wnt signaling and Cdx-related transcriptional activation in the regulation of the claudin-2 promoter activity. Biochemical and Biophysical Research Communications, 314(4), 1001-1007. https://doi.org/10.1016/j.bbrc.2003.12.185
Miwa, N., Furuse, M., Tsukita, S., Niikawa, N., Nakamura, Y., & Furukawa, Y. (2001). Involvement of claudin-1 in the beta-catenin/Tcf signaling pathway and its frequent upregulation in human colorectal cancers. Oncology Research, 12(11-12), 469-476. https://doi.org/10.3727/096504001108747477
Bhat, A. A., Sharma, A., Pope, J., Krishnan, M., Washington, M. K., Singh, A. B., & Dhawan, P. (2012). Caudal homeobox protein Cdx-2 cooperates with Wnt pathway to regulate claudin-1 expression in colon cancer cells. PloS One, 7(6), e37174. https://doi.org/10.1371/journal.pone.0037174
Dahan, S., Rabinowitz, K. M., Martin, A. P., Berin, M. C., Unkeless, J. C., & Mayer, L. (2011). Notch-1 signaling regulates intestinal epithelial barrier function, through interaction with CD4+ T cells, in mice and humans. Gastroenterology, 140(2), 550-559. https://doi.org/10.1053/j.gastro.2010.10.057
Mees, S. T., Mennigen, R., Spieker, T., Rijcken, E., Senninger, N., Haier, J., & Bruewer, M. (2009). Expression of tight and adherens junction proteins in ulcerative colitis associated colorectal carcinoma: Upregulation of claudin-1, claudin-3, claudin-4, and beta-catenin. International Journal of Colorectal Disease, 24(4), 361-368. https://doi.org/10.1007/s00384-009-0653-y
Nilsson, A., Peric, A., Strimfors, M., Goodwin, R. J. A., Hayes, M. A., Andrén, P. E., & Hilgendorf, C. (2017). Mass Spectrometry Imaging proves differential absorption profiles of well-characterised permeability markers along the crypt-villus axis. Scientific Reports, 7(1), 6352. https://doi.org/10.1038/s41598-017-06583-4
Pearce, S. C., Al-Jawadi, A., Kishida, K., Yu, S., Hu, M., Fritzky, L. F., Edelblum, K. L., Gao, N., & Ferraris, R. P. (2018). Marked differences in tight junction composition and macromolecular permeability among different intestinal cell types. BMC Biology, 16(1), 19. https://doi.org/10.1186/s12915-018-0481-z
Pope, J. L., Ahmad, R., Bhat, A. A., Washington, M. K., Singh, A. B., & Dhawan, P. (2014a). Claudin-1 overexpression in intestinal epithelial cells enhances susceptibility to adenamatous polyposis coli-mediated colon tumorigenesis. Molecular Cancer, 13, 167. https://doi.org/10.1186/1476-4598-13-167
Pope, J. L., Bhat, A. A., Sharma, A., Ahmad, R., Krishnan, M., Washington, M. K., Beauchamp, R. D., Singh, A. B., & Dhawan, P. (2014b). Claudin-1 regulates intestinal epithelial homeostasis through the modulation of Notch-signalling. Gut, 63(4), 622-634. https://doi.org/10.1136/gutjnl-2012-304241
Huber, P. (2020). Targeting of the apical junctional complex by bacterial pathogens. Biochimica Et Biophysica Acta. Biomembranes, 1862(6), 183237. https://doi.org/10.1016/j.bbamem.2020.183237
Lechuga, S., & Ivanov, A. I. (2021). Actin cytoskeleton dynamics during mucosal inflammation: A view from broken epithelial barriers. Current Opinion in Physiology, 19, 10-16. https://doi.org/10.1016/j.cophys.2020.06.012
Rusu, A. D., & Georgiou, M. (2020). The multifarious regulation of the apical junctional complex. Open Biology, 10(2), 190278. https://doi.org/10.1098/rsob.190278
Spadaro, D., Tapia, R., Pulimeno, P., & Citi, S. (2012). The control of gene expression and cell proliferation by the epithelial apical junctional complex. Essays in Biochemistry, 53, 83-93. https://doi.org/10.1042/bse0530083
Tsukita, K., Yano, T., Tamura, A., & Tsukita, S. (2019a). Reciprocal Association between the Apical Junctional Complex and AMPK: A Promising Therapeutic Target for Epithelial/Endothelial Barrier Function? International Journal of Molecular Sciences, 20(23), E6012. https://doi.org/10.3390/ijms20236012
Garber, J. J., Mallick, E. M., Scanlon, K. M., Turner, J. R., Donnenberg, M. S., Leong, J. M., & Snapper, S. B. (2018). Attaching-and-Effacing Pathogens Exploit Junction Regulatory Activities of N-WASP and SNX9 to Disrupt the Intestinal Barrier. Cellular and Molecular Gastroenterology and Hepatology, 5(3), 273-288. https://doi.org/10.1016/j.jcmgh.2017.11.015
Citalán-Madrid, A. F., Vargas-Robles, H., García-Ponce, A., Shibayama, M., Betanzos, A., Nava, P., Salinas-Lara, C., Rottner, K., Mennigen, R., & Schnoor, M. (2017). Cortactin deficiency causes increased RhoA/ROCK1-dependent actomyosin contractility, intestinal epithelial barrier dysfunction, and disproportionately severe DSS-induced colitis. Mucosal Immunology, 10(5), 1237-1247. https://doi.org/10.1038/mi.2016.136
Wang, D., Naydenov, N. G., Feygin, A., Baranwal, S., Kuemmerle, J. F., & Ivanov, A. I. (2016). Actin-Depolymerizing Factor and Cofilin-1 Have Unique and Overlapping Functions in Regulating Intestinal Epithelial Junctions and Mucosal Inflammation. The American Journal of Pathology, 186(4), 844-858. https://doi.org/10.1016/j.ajpath.2015.11.023
Naydenov, N. G., Feygin, A., Wang, D., Kuemmerle, J. F., Harris, G., Conti, M. A., Adelstein, R. S., & Ivanov, A. I. (2016). Nonmuscle Myosin IIA Regulates Intestinal Epithelial Barrier in vivo and Plays a Protective Role During Experimental Colitis. Scientific Reports, 6, 24161. https://doi.org/10.1038/srep24161
Hegan, P. S., Chandhoke, S. K., Barone, C., Egan, M., Bähler, M., & Mooseker, M. S. (2016). Mice lacking myosin IXb, an inflammatory bowel disease susceptibility gene, have impaired intestinal barrier function and superficial ulceration in the ileum. Cytoskeleton (Hoboken, N.J.), 73(4), 163-179. https://doi.org/10.1002/cm.21292
McAlpine, W., Wang, K., Choi, J. H., San Miguel, M., McAlpine, S. G., Russell, J., Ludwig, S., Li, X., Tang, M., Zhan, X., Choi, M., Wang, T., Bu, C. H., Murray, A. R., Moresco, E. M. Y., Turer, E. E., & Beutler, B. (2018). The class I myosin MYO1D binds to lipid and protects against colitis. Disease Models & Mechanisms, 11(9), dmm035923. https://doi.org/10.1242/dmm.035923
Tyska, M. J., Mackey, A. T., Huang, J.-D., Copeland, N. G., Jenkins, N. A., & Mooseker, M. S. (2005). Myosin-1a is critical for normal brush border structure and composition. Molecular Biology of the Cell, 16(5), 2443-2457. https://doi.org/10.1091/mbc.e04-12-1116
Grover, M., Camilleri, M., Hines, J., Burton, D., Ryks, M., Wadhwa, A., Sundt, W., Dyer, R., & Singh, R. J. (2016). (13) C mannitol as a novel biomarker for measurement of intestinal permeability. Neurogastroenterology and Motility: The Official Journal of the European Gastrointestinal Motility Society, 28(7), 1114-1119. https://doi.org/10.1111/nmo.12802
Khoshbin, K., Khanna, L., Maselli, D., Atieh, J., Breen-Lyles, M., Arndt, K., Rhoten, D., Dyer, R. B., Singh, R. J., Nayar, S., Bjerkness, S., Harmsen, W. S., Busciglio, I., & Camilleri, M. (2021). Development and Validation of Test for «Leaky Gut» Small Intestinal and Colonic Permeability Using Sugars in Healthy Adults. Gastroenterology, S0016-5085(21)00642-9. https://doi.org/10.1053/j.gastro.2021.04.020
Gupta, R. B., Harpaz, N., Itzkowitz, S., Hossain, S., Matula, S., Kornbluth, A., Bodian, C., & Ullman, T. (2007). Histologic inflammation is a risk factor for progression to colorectal neoplasia in ulcerative colitis: A cohort study. Gastroenterology, 133(4), 1099-1105; quiz 1340-1341. https://doi.org/10.1053/j.gastro.2007.08.001
Weber, C. R., Nalle, S. C., Tretiakova, M., Rubin, D. T., & Turner, J. R. (2008). Claudin-1 and claudin-2 expression is elevated in inflammatory bowel disease and may contribute to early neoplastic transformation. Laboratory Investigation; a Journal of Technical Methods and Pathology, 88(10), 1110-1120. https://doi.org/10.1038/labinvest.2008.78
Trier, J. S., & Rubin, C. E. (1964). Electron microscopy of the gut: a word of caution. Gastroenterology, 47, 313-315.
Severs, N. J. (2007). Freeze-fracture electron microscopy. Nature Protocols, 2(3), 547-576. https://doi.org/10.1038/nprot.2007.55
Suenaert, P., Bulteel, V., Lemmens, L., Noman, M., Geypens, B., Van Assche, G., Geboes, K., Ceuppens, J. L., & Rutgeerts, P. (2002). Anti-tumor necrosis factor treatment restores the gut barrier in Crohn’s disease. The American Journal of Gastroenterology, 97(8), 2000-2004. https://doi.org/10.1111/j.1572-0241.2002.05914.x
Schmitz, H., Barmeyer, C., Gitter, A. H., Wullstein, F., Bentzel, C. J., Fromm, M., Riecken, E. O., & Schulzke, J. D. (2000). Epithelial barrier and transport function of the colon in ulcerative colitis. Annals of the New York Academy of Sciences, 915, 312-326. https://doi.org/10.1111/j.1749-6632.2000.tb05259.x
Crespo, M., Vilar, E., Tsai, S.-Y., Chang, K., Amin, S., Srinivasan, T., Zhang, T., Pipalia, N. H., Chen, H. J., Witherspoon, M., Gordillo, M., Xiang, J. Z., Maxfield, F. R., Lipkin, S., Evans, T., & Chen, S. (2017). Colonic organoids derived from human induced pluripotent stem cells for modeling colorectal cancer and drug testing. Nature Medicine, 23(7), 878-884. https://doi.org/10.1038/nm.4355
McCracken, K. W., Howell, J. C., Wells, J. M., & Spence, J. R. (2011). Generating human intestinal tissue from pluripotent stem cells in vitro. Nature Protocols, 6(12), 1920-1928. https://doi.org/10.1038/nprot.2011.410
Spence, J. R., Mayhew, C. N., Rankin, S. A., Kuhar, M. F., Vallance, J. E., Tolle, K., Hoskins, E. E., Kalinichenko, V. V., Wells, S. I., Zorn, A. M., Shroyer, N. F., & Wells, J. M. (2011). Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature, 470(7332), 105-109. https://doi.org/10.1038/nature09691
Sarvestani, S. K., Signs, S., Hu, B., Yeu, Y., Feng, H., Ni, Y., Hill, D. R., Fisher, R. C., Ferrandon, S., DeHaan, R. K., Stiene, J., Cruise, M., Hwang, T. H., Shen, X., Spence, J. R., & Huang, E. H. (2021). Induced organoids derived from patients with ulcerative colitis recapitulate colitic reactivity. Nature Communications, 12(1), 262. https://doi.org/10.1038/s41467-020-20351-5
Liu, Y., & Chen, Y.-G. (2018). 2D- and 3D-Based Intestinal Stem Cell Cultures for Personalized Medicine. Cells, 7(12), E225. https://doi.org/10.3390/cells7120225
Roodsant, T., Navis, M., Aknouch, I., Renes, I. B., van Elburg, R. M., Pajkrt, D., Wolthers, K. C., Schultsz, C., van der Ark, K. C. H., Sridhar, A., & Muncan, V. (2020). A Human 2D Primary Organoid-Derived Epithelial Monolayer Model to Study Host-Pathogen Interaction in the Small Intestine. Frontiers in Cellular and Infection Microbiology, 10, 272. https://doi.org/10.3389/fcimb.2020.00272
Acknowledgments
We want to extend our gratitude to Dr. Zayda Piedra Quintero for sharing her expertise in the inflammation field and for her critical discussion of the manuscript. The project was supported by the sectoral funding for research and education via the grant for Basic Science from Conacyt (No. A1-S-20887 to PND) and the SEP-Conacyt grant (No.179 to NV/PND). MREG is the recipient of a doctoral fellowship from Conacyt (860508).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
del Rocio Encarnacion-Garcia, M., Nava, P. (2022). Tight Junctions in the Inflamed Gut. In: González-Mariscal, L. (eds) Tight Junctions. Springer, Cham. https://doi.org/10.1007/978-3-030-97204-2_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-97204-2_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-97203-5
Online ISBN: 978-3-030-97204-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)