Abstract
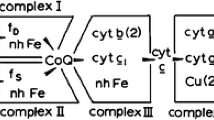
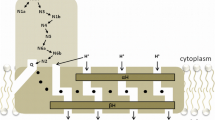
In this chapter we provide a review with a focus on the function of Coenzyme Q (CoQ, ubiquinone) in mitochondria. The notion of a mobile pool of CoQ in the lipid bilayer as the vehicle of electrons from respiratory complexes has somewhat changed with the discovery of respiratory supramolecular units, in particular the supercomplex comprising Complexes I and III; in such assembly the electron transfer is thought to be mediated by direct channelling, and we provide evidence for a kinetic advantage on the transfer based on random collisions. The CoQ pool, however, has a fundamental function in establishing a dissociation equilibrium with bound CoQ, besides being required for electron transfer from other dehydrogenases to Complex III. CoQ bound to Complex I and to Complex III is also involved in proton translocation; although the mechanism of the Q-cycle is well established for Complex III, the involvement of CoQ in proton translocation by Complex I is still debated. This review also briefly examines some additional roles of CoQ, such as the antioxidant effect of its reduced form and its postulated action at the transcriptional level.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Acín-Peréz R, Enriquez JA (2014) The function of respiratory supercomplexes: the plasticity model. Biochim Biophys Acta 1837:444–450
Acín-Peréz R, Fernández-Silva P, Peleato ML et al (2008) Respiratory active mitochondrial supercomplexes. Mol Cell 32:529–539
Althoff T, Mills DJ, Popot JL et al (2011) Arrangement of electron transport chain components in bovine mitochondrial supercomplex I1III2IV1. EMBO J 30:4652–4664
Arroyo A, Navarro F, Navas P et al (1998) Ubiquinol regeneration by plasma membrane ubiquinone reductase. Protoplasma 205:107–113
Baradaran R, Berrisford JM, Minhas GS et al (2013) Crystal structure of the entire respiratory complex I. Nature 494:443–448
Battino M, Fahmy T, Lenaz G (1986) Determination of the critical micelle concentration of short-chain ubiquinones in model systems. Biochim Biophys Acta 851:377–384
Battino M, Ferri E, Villa RF et al (1990) Natural distribution and occurrence of coenzyme Q homologues. Membr Biochem 9:179–190
Benard G, Faustin B, Galinier A et al (2008) Functional dynamic compartmentalization of respiratory chain intermediate substrates: implications for the control of energy production and mitochondrial diseases. Int J Biochem Cell Biol 40:1543–1554
Bergamini C, Moruzzi N, Sblendido A et al (2012) A water soluble CoQ10 formulation improves intracellular distribution and promotes mitochondrial respiration in cultured cells. PLoS One 7:e33712
Bernardi P, Forte M (2007) The mitochondrial permeability transition pore. Novartis Found Symp 287:157–164; discussion 164-169
Beyer RE, Segura-Aguilar J, Di Bernardo S et al (1996) The role of DT-diaphorase in the maintenance of the reduced antioxidant form of coenzyme Q in membrane systems. Proc Natl Acad Sci U S A 93:2528–2532
Bianchi C, Fato R, Genova ML et al (2003) Structural and functional organization of Complex I in the mitochondrial respiratory chain. Biofactors 18:3–9
Bianchi C, Genova ML, Parenti Castelli G et al (2004) The mitochondrial respiratory chain is partially organized in a supercomplex assembly: kinetic evidence using flux control analysis. J Biol Chem 279:36562–36569
Blaza JN, Serrelli R, Jones AJ et al (2014) Kinetic evidence against partitioning of the ubiquinone pool and the catalytic relevance of respiratory chain supercomplexes. Proc Natl Acad Sci U S A 111:15735–15740
Boumans H, Grivell LA, Berden JA (1998) The respiratory chain in yeast behaves as a single functional unit. J Biol Chem 273:4872–4877
Brandt U, Kerscher S, Drose S et al (2003) Proton pumping by NADH:ubiquinone oxidoreductase. A redox driven conformational change mechanism? FEBS Lett 545:9–17
Braun BS, Benbow U, Lloyd-Williams P et al (1986) Determination of partition coefficients of quinones by high-performance liquid chromatography. Methods Enzymol 125:119–129
Capaldi RA (1982) Arrangement of proteins in the mitochondrial inner membrane. Biochim Biophys Acta 694:292–306
Chacko BK, Reily C, Srivastava A et al (2010) Prevention of diabetic nephropathy in Ins2(+/)-(AkitaJ) mice by the mitochondria-targeted therapy MitoQ. Biochem J 432:9–19
Chance B, Williams GR (1955) A method for the localization of sites for oxidative phosphorylation. Nature 176:250–254
Cordero MD, Moreno-Fernández AM, Gomez-Skarmeta JL et al (2009) Coenzyme Q10 and alpha-tocopherol protect against amitriptyline toxicity. Toxicol Appl Pharmacol 235:329–337
Cramer WA, Hasan SS, Yamashita E (2011) The Q cycle of cytochrome bc complexes: a structure perspective. Biochim Biophys Acta 1807:788–802
Crane FL, Hatefi Y, Lester RL et al (1957) Isolation of a quinone from beef heart mitochondria. Biochim Biophys Acta 25:220–221
Crane FL, Widmer C, Lester RL et al (1959) Studies on the electron transport system. XV. Coenzyme Q (Q275) and the succinoxidase activity of the electron transport particle. Biochim Biophys Acta 31:476–489
Crofts AR (2004) The Q-cycle, a personal perspective. Photosynth Res 80:223–243
Crofts AR, Hong S, Wilson C et al (2013) The mechanism of ubihydroquinone oxidation at the Qo-site of the cytochrome bc1 complex. Biochim Biophys Acta 1827:1362–1377
Degli Esposti M (1998) Inhibitors of NADH-ubiquinone reductase: an overview. Biochim Biophys Acta 1364:222–235
Degli Esposti M, Ngo A, Ghelli A et al (1996) The interaction of Q analogs, particularly hydroxydecyl benzoquinone (idebenone), with the respiratory complexes of heart mitochondria. Arch Biochem Biophys 330:395–400
Devun F, Walter L, Belliere J et al (2010) Ubiquinone analogs: a mitochondrial permeability transition pore-dependent pathway to selective cell death. PLoS One 5:e11792
Di Bernardo S, Fato R, Casadio R et al (1998) A high diffusion coefficient for coenzyme Q10 might be related to a folded structure. FEBS Lett 426:77–80
Dudkina NV, Kudryashev M, Stahlberg H et al (2011) Interaction of complexes I, III, and IV within the bovine respirasome by single particle cryoelectron tomography. Proc Natl Acad Sci U S A 108:15196–15200
Durán-Prado M, Frontiñán J, Santiago-Mora R et al (2014) Coenzyme Q10 protects human endothelial cells from β-amyloid uptake and oxidative stress-induced injury. PLoS One 9:e109223
Duveau DY, Arce PM, Schoenfeld RA et al (2010) Synthesis and characterization of mitoQ and idebenone analogues as mediators of oxygen consumption in mitochondria. Bioorg Med Chem 18:6429–6441
Echtay KS, Winkler E, Klingenberg M (2000) Coenzyme Q is an obligatory cofactor for uncoupling protein function. Nature 408:609–613
Efremov RG, Sazanov LA (2011) Structure of the membrane domain of respiratory complex I. Nature 476:414–420
Efremov RG, Sazanov LA (2012) The coupling mechanism of respiratory complex I - a structural and evolutionary perspective. Biochim Biophys Acta 1817:1785–1795
Enriquez JA, Lenaz G (2014) Coenzyme Q and the respiratory chain: coenzyme Q pool and mitochondrial supercomplexes. Mol Syndromol 5:119–140
Ernster L, Dallner G (1995) Biochemical, physiological and medical aspects of ubiquinone function. Biochim Biophys Acta 1271:195–204
Estornell E, Fato R, Castelluccio C et al (1992) Saturation kinetics of coenzyme Q in NADH and succinate oxidation in beef heart mitochondria. FEBS Lett 311:107–109
Fato R, Battino M, Degli Esposti M et al (1986) Determination of partition and lateral diffusion coefficients of ubiquinones by fluorescence quenching of n-(9-anthroyloxy)stearic acids in phospholipid vesicles and mitochondrial membranes. Biochemistry 25:3378–3390
Fato R, Castelluccio C, Palmer G et al (1988) A simple method for the determination of the kinetic constants of membrane enzymes utilizing hydrophobic substrates: Ubiquinol cytochrome c reductase. Biochim Biophys Acta 932:216–222
Fato R, Estornell E, Di Bernardo S et al (1996) Steady- state kinetics of the reduction of coenzyme Q analogs by complex I (NADH:ubiquinone oxidoreductase) in bovine heart mitochondria and submitochondrial particles. Biochemistry 35:2705–2716
Flemming D, Stolpe S, Schneider D et al (2005) A possible role for iron-sulfur cluster N2 in proton translocation by the NADH: ubiquinone oxidoreductase (complex I). J Mol Microbiol Biotechnol 10:208–222
Friedrich T (2001) Complex I: a chimaera of a redox and conformation-driven proton pump? J Bioenerg Biomembr 33:169–177
Galassi VV, Arantes GM (2015) Partition, orientation and mobility of ubiquinones in a lipid bilayer. Biochim Biophys Acta 1847:1560–1573
Genova ML, Lenaz G (2011) New developments on the functions of coenzyme Q in mitochondria. Biofactors 37:330–354
Genova ML, Lenaz G (2013) A critical appraisal of the role of respiratory supercomplexes in mitochondria. Biol Chem 394:631–639
Genova ML, Lenaz G (2014) Functional role of mitochondrial respiratory supercomplexes. Biochim Biophys Acta 1837:427–443
Genova ML, Baracca A, Biondi A et al (2008) Is supercomplex organization of the respiratory chain required for optimal electron transfer activity? Biochim Biophys Acta 1777:740–746
Gorelick C, Lopez-Jones M, Goldberg GL et al (2004) Coenzyme Q10 and lipid-related gene induction in HeLa cells. Am J Obstet Gynecol 190:1432–1434
Green DE, Tzagoloff A (1966) The mitochondrial electron transfer chain. Arch Biochem Biophys 116:293–304
Grivennikova VG, Roth R, Zakharova NV et al (2003) The mitochondrial and prokaryotic proton-translocating NADH:ubiquinone oxidoreductases: similarities and dissimilarities of the quinone-junction sites. Biochim Biophys Acta 1607:79–90
Groneberg DA, Kindermann B, Althammer M et al (2005) Coenzyme Q10 affects expression of genes involved in cell signalling, metabolism and transport in human CaCo-2 cells. Int J Biochem Cell Biol 37:1208–1218
Gu J, Wu M, Guo R et al (2016) The architecture of the mammalian respirasome. Nature 537:639–643
Gunner MR, Madeo J, Zhu Z (2008) Modification of quinone electrochemistry by the proteins in the biological electron transfer chains: examples from photosynthetic reaction centers. J Bioenerg Biomembr 40:509–519
Gutman M (1985) Kinetic analysis of electron flux through the quinones in the mitochondrial system. In: Lenaz G (ed) Coenzyme Q. Wiley, Chichester, pp 215–234
Gutman M, Silman N (1972) Mutual inhibition between NADH oxidase and succinoxidase activities in respiring submitochondrial particles. FEBS Lett 26:207–210
Gutman M, Coles CJ, Singer TP et al (1971) On the functional organization of the respiratory chain at the dehydrogenase-coenzyme Q junction. Biochemistry 10:2036–2043
Hackenbrock CR, Chazotte B, Gupte SS (1986) The random collision model and a critical assessment of diffusion and collision in mitochondrial electron transport. J Bioenerg Biomembr 18:331–368
Hatefi Y, Haavik AG, Fowler LR et al (1962a) Studies on the electron transfer system. XLII. Reconstitution of the electron transfer system. J Biol Chem 237:2661–2669
Hatefi Y, Haavik AG, Griffiths DE (1962b) Studies on the electron transfer system. XL. Preparation and properties of mitochondrial DPNH-Coenzyme Q reductase. J Biol Chem 237:1676–1680
Heron C, Ragan CI, Trumpower BL (1978) The interaction between mitochondrial NADH- ubiquinone oxidoreductase and ubiquinol-cytochrome c oxidoreductase. Restoration of ubiquinone-pool behavior. Biochem J 174:791–800
Hochman J, Ferguson-Miller S, Schindler M (1985) Mobility in the mitochondrial electron transport chain. Biochemistry 24:2509–2516
Imada I, Fujita T, Sugiyama Y et al (1989) Effects of idebenone and related compounds on respiratory activities of brain mitochondria, and on lipid peroxidation of their membranes. Arch Gerontol Geriatr 8:323–341
Imada I, Sato EF, Kira Y et al (2008) Effect of CoQ homologues on reactive oxygen generation by mitochondria. Biofactors 32:41–48
Iwata S, Lee JW, Okada K et al (1998) Complete structure of the 11-subunit bovine mitochondrial cytochrome bc1 complex. Science 281:64–71
Jackman MR, Willis WT (1996) Characteristics of mitochondria isolated from type I and type IIb skeletal muscle. Am J Physiol 270:C673–C678
James AM, Cochemé HM, Smith RA et al (2005) Interactions of mitochondria-targeted and untargeted ubiquinones with the mitochondrial respiratory chain and reactive oxygen species. Implications for the use of exogenous ubiquinones as therapies and experimental tools. J Biol Chem 280:21295–21312
Joela H, Kasa S, Lehtovuori P et al (1997) EPR, ENDOR and TRIPLE resonance and MO studies on ubiquinones (Q-n): comparison of radical anions and cations of coenzymes Q-10 and Q-6 with the model compounds Q-2 and Q-0. Acta Chem Scand 51:233–241
Jones AJ, Blaza JN, Bridges HR et al (2016) A self-assembled respiratory chain that catalyzes NADH oxidation by ubiquinone-10 cycling between complex I and the alternative oxidase. Angew Chem Int Ed Engl 55:728–731
Jørgensen BM, Rasmussen HN, Rasmussen UF (1985) Ubiquinone reduction pattern in pigeon heart mitochondria. Identification of three distinct ubiquinone pools. Biochem J 229:621–629
Kaambre T, Chekulayev V, Shevchuk I et al (2012) Metabolic control analysis of cellular respiration in situ in intraoperational samples of human breast cancer. J Bioenerg Biomembr 44:539–558
Kaambre T, Chekulayev V, Shevchuk I et al (2013) Metabolic control analysis of respiration in human cancer tissue. Front Physiol 4:151(1–6)
Kaltschmidt B, Sparna T, Kaltschmidt C (1999) Activation of NF-kappa B by reactive oxygen intermediates in the nervous system. Antioxid Redox Signal 1:129–144
Kaurola P, Sharma V, Vonk A et al (2016) Distribution and dynamics of quinones in the lipid bilayer mimicking the inner membrane of mitochondria. Biochim Biophys Acta 1858:2116–2122
Kelso GF, Porteous CM, Coulter CV et al (2001) Selective targeting of a redox-active ubiquinone to mitochondria within cells: antioxidant and antiapoptotic properties. J Biol Chem 276:4588–4596
Kholodenko NB, Westerhoff HV (1993) Metabolic channelling and control of the flux. FEBS Lett 320:71–74
Kröger A, Klingenberg M (1973a) The kinetics of the redox reactions of ubiquinone related to the electron-transport activity in the respiratory chain. Eur J Biochem 34:358–368
Kröger A, Klingenberg M (1973b) Further evidence of the pool function of ubiquinone as derived from the inhibition of the electron transport by antimycin. Eur J Biochem 39:313–323
Landi L, Pasquali P, Cabrini L et al (1984) On the mechanism of inhibition of NADH oxidase by ubiquinone-3. J Bioenerg Biomembr 16:153–166
Lapuente-Brun E, Moreno-Loshuertos R, Acín-Peréz R et al (2013) Supercomplex assembly determines electron flux in themitochondrial electron transport chain. Science 340:1567–1570
Lass A, Sohal RS (1999) Comparisons of coenzyme Q bound to mitochondrial membrane proteins among different mammalian species. Free Radic Biol Med 27:220–226
Lehninger AL (1965) Bionergetics: the molecular basis of biological energy transformations. WA Benjamin Inc, New York
Lenaz G (1988) Role of mobility of redox components in the inner mitochondrial membrane. J Membrane Biol 104:193–209
Lenaz G (1998) Quinone specificity of complex I. Biochim Biophys Acta 1364:207–221
Lenaz G, Fato R (1986) Is ubiquinone diffusion rate-limiting for electron transfer? J Bioenerg Biomembr 18:369–401
Lenaz G, Genova ML (2007) Kinetics of integrated electron transfer in the mitochondrial respiratory chain: random collisions vs solid state electron channeling. Am J Physiol Cell Physiol 292:C1221–C1239
Lenaz G, Genova ML (2009a) Structural and functional organization of the mitochondrial respiratory chain: a dynamic super-assembly. Int J Biochem Cell Biol 41:1750–1772
Lenaz G, Genova ML (2009b) Mobility and function of Coenzyme Q (ubiquinone) in the mitochondrial respiratory chain. Biochim Biophys Acta 1787:563–573
Lenaz G, Genova ML (2010) Structure and organization of mitochondrial respiratory complexes: a new understanding of an old subject. Antioxid Redox Signal 12:961–1008
Lenaz G, Daves GD, Folkers K (1968) Organic structural specificity and sites of coenzyme Q in succinoxidase and DPNH-oxidase systems. Arch Biochem Biophys 123:539–550
Lenaz G, Fato R, Castelluccio C et al (1994) An updating of the biochemical function of coenzyme Q in mitochondria. Mol Aspects Med 15(Suppl):S29–S36
Lenaz G, Bovina C, Castelluccio C et al (1997) Mitochondrial complex I defects in aging. Mol Cell Biochem 174:329–333
Lenaz G, Fato R, Di Bernardo S et al (1999) Localization and mobility of coenzyme Q in lipid bilayers and membranes. Biofactors 9:87–93
Lenaz G, Baracca A, Barbero G et al (2010) Mitochondrial respiratory chain super-complex I-III in physiology and pathology. Biochim Biophys Acta 1797:633–640
Lenaz G, Tioli G, Falasca AI et al (2016) Complex I function in mitochondrial supercomplexes. Biochim Biophys Acta 1857:991–1000
Letts JA, Fiedorczuk K, Sazanov LA (2016) The architecture of respiratory supercomplexes. Nature 537:644–648
Letts JA, Fiedorczuk K, Degliesposti G, Skehel M, Sazanov LA (2019) Structures of respiratory rupercomplex I+III2 reveal functional and conformational crosstalk. Mol Cell 75:1131–1146.e6
Li G, Zou LY, Cao CM et al (2005) Coenzyme Q10 protects SHSY5Y neuronal cells from beta amyloid toxicity and oxygen-glucose deprivation by inhibiting the opening of the mitochondrial permeability transition pore. Biofactors 25:97–107
Linnane AW, Kopsidas G, Zhang C et al (2002) Cellular redox activity of coenzyme Q10: effect of CoQ10 supplementation on human skeletal muscle. Free Radic Res 36:445–453
Maranzana E, Barbero G, Falasca AI et al (2013) Mitochondrial respiratory supercomplex association limits production of reactive oxygen species from Complex I. Antioxid Redox Signal 19:1469–1480
Mashima Y, Hiida Y, Oguchi Y (1992) Remission of Leber’s hereditary optic neuropathy with idebenone. Lancet 340:368–369
McDaniel DH, Neudecker BA, Di Nardo JC et al (2005) Idebenone: a new antioxidant – Part I. Relative assessment of oxidative stress protection capacity compared to commonly known antioxidants. J Cosmet Dermatol 4:10–17
McKenzie M, Lazarou M, Thorburn DR et al (2006) Mitochondrial respiratory chain supercomplexes are destabilized in Barth syndrome patients. J Mol Biol 361:462–469
Mileykovskaya E, Dowhan W (2014) Cardiolipin-dependent formation of mitochondrial respiratory supercomplexes. Chem Phys Lipids 17:942–948
Mileykovskaya E, Penczek PA, Fang J et al (2012) Arrangement of the respiratory chain complexes in Saccharomyces cerevisiae supercomplex III2IV2 revealed by single particle cryo-electron microscopy (EM). J Biol Chem 287:23095–23103
Mitchell P (1975) The protonmotive Q cycle: a general formulation. FEBS Lett 59:137–139
Moreno-Sanchez S, Devars S, Lopez-Gomez F et al (1991) Distribution of control of oxidative phosphorylation in mitochondria oxidizing NAD-linked substrates. Biochim Biophys Acta 1060:284–292
Mráček T, Holzerová E, Drahota Z et al (2014) ROS generation and multiple forms of mammalian mitochondrial glycerol-3-phosphate dehydrogenase. Biochim Biophys Acta 1837:98–111
Naderi J, Somayajulu-Nitu M, Mukerji A et al (2006) Water-soluble formulation of Coenzyme Q10 inhibits Bax-induced destabilization of mitochondria in mammalian cells. Apoptosis 11:1359–1369
Nohl H, Rohr-Udilova N, Gille L et al (2005) Suppression of tumour-promoting factors in fat-induced colon carcinogenesis by the antioxidants caroverine and ubiquinone. Anticancer Res 25:2793–2800
Ohnishi T (1998) Iron-sulfur clusters/semiquinones in complex I. Biochim Biophys Acta 1364:186–206
Ohnishi T, Salerno JC (2005) Conformation-driven and semiquinone-gated proton-pump mechanism in the NADH-ubiquinone oxidoreductase (complex I). FEBS Lett 579:4555–4561
Ohnishi ST, Salerno JC, Ohnishi T (2010) Possible roles of two quinone molecules in direct and indirect proton pumps of bovine heart NADH-quinone oxidoreductase (complex I). Biochim Biophys Acta 1797:1891–1893
Ovàdi J (1991) Physiological significance of metabolic channelling. J Theor Biol 152:135–141
Ozawa T, Nishikimi M, Suzuki H et al (1987) Structure and assembly of mitochondrial electron-transfer complexes. In: Ozawa T, Papa S (eds) Bioenergetics: structure and function of energy-transducing systems. Japan Sci Soc Press, Tokyo, pp 101–119
Papucci L, Schiavone N, Witort E et al (2003) Coenzyme Q10 prevents apoptosis by inhibiting mitochondrial depolarization independently of its free radical scavenging property. J Biol Chem 278:28220–28228
Parenti Castelli G, Fato R, Castelluccio C et al (1987) Kinetic studies on the pool function of ubiquinone in mitochondrial systems. Chem Scr 27:161–166
Park SK, Kim K, Page GP et al (2009) Gene expression profiling of aging in multiple mouse strains: identification of aging biomarkers and impact of dietary antioxidants. Aging Cell 8:484–495
Pfeiffer K, Gohil V, Stuart RA et al (2003) Cardiolipin stabilizes respiratory chain supercomplexes. J Biol Chem 278:52873–52880
Piccoli C, Scrima R, Boffoli D et al (2006) Control by cytochrome c oxidase of the cellular oxidative phosphorylation system depends on the mitochondrial energy state. Biochem J 396:573–583
Plecitá-Hlavatá L, Jezek J, Jezek P (2009) Pro-oxidant mitochondrial matrix-targeted ubiquinone MitoQ10 acts as anti-oxidant at retarded electron transport or proton pumping within Complex I. Int J Biochem Cell Biol 41:1697–1707
Quarato G, Piccoli C, Scrima R et al (2011) Variation of flux control coefficient of cytochrome c oxidase and of the other respiratory chain complexes at different values protonmotive force occurs by a threshold mechanism. Biochim Biophys Acta 1807:1114–1124
Quinzii CM, Hirano M (2010) Coenzyme Q and mitochondrial disease. Dev Disabil Res Rev 16:183–188
Ragan CI, Cottingham IR (1985) The kinetics of quinone pools in electron transport. Biochim Biophys Acta 811:13–31
Ragan CI, Heron C (1978) The interaction between mitochondrial NADH-ubiquinone oxidoreductase and ubiquinol-cytochrome c oxidoreductase – evidence for stoicheiometric association. Biochem J 174:783–790
Rajarathnam K, Hochman J, Schindler M et al (1989) Synthesis, location, and lateral mobility of fluorescently labeled ubiquinone 10 in mitochondrial and artificial membranes. Biochemistry 28:3168–3176
Rauchová H, Fato R, Drahota Z et al (1997) Steady-state kinetics of reduction of Coenzyme Q analogs by glycerol-3-phosphate dehydrogenase in brown adipose tissue mitochondria. Arch Biochem Biophys 344:235–241
Redfearn ER, Pumphrey AM (1960) The kinetics of ubiquinone reactions in heart-muscle preparations. Biochem J 76:64–71
Rich PR, Harper R (1990) Partition coefficients of quinones and hydroquinones and their relation to biochemical reactivity. FEBS Lett 269:139–144
Rieske JS (1967) Preparation and properties of reduced coenzyme Q-cytochrome c reductase (complex III of the respiratory chain). Methods Enzymol 10:239–245
Ringler RL, Singer TP (1959) Studies on the mitochondrial alpha-glycerophosphate dehydrogenase. I. Reaction of the dehydrogenase with electron acceptors and the respiratory chain. J Biol Chem 234:2211–2217
Rosca MG, Vazquez EJ, Kerner J et al (2008) Cardiac mitochondria in heart failure: decrease in respirasomes and oxidative phosphorylation. Cardiovasc Res 80:30–39
Rosenfeldt F, Marasco S, Lyon W et al (2005) Coenzyme Q10 therapy before cardiac surgery improves mitochondrial function and in vitro contractility of myocardial tissue. J Thorac Cardiovasc Surg 129:25–32
Sahach VF, Vavilova HL, Rudyk OV et al (2007) Inhibition of mitochondrial permeability transition pore is one of the mechanisms of cardioprotective effect of coenzyme Q10. Fiziol Zh 53:35–42
Samorì B, Lenaz G, Battino M et al (1992) On coenzyme Q orientation in membranes: a linear dichroism study of ubiquinones in a model bilayer. J Membr Biol 128:193–203
Sarewicz M, Osyczka A (2015) Electronic connection between the quinone and cytochrome c redox pools and its role in regulation of mitochondrial electron transport and redox signaling. Physiol Rev 95:219–243
Sazanov LA (2015) A giant molecular proton pump: structure and mechanism of respiratory complex I. Nat Rev Mol Cell Biol 16:375–388
Schäfer E, Seelert H, Reifschneider NH et al (2006) Architecture of active mammalian respiratory chain supercomplexes. J Biol Chem 281:15370–15375
Schägger H, Pfeiffer K (2000) Supercomplexes in the respiratory chains of yeast and mammalian mitochondria. EMBO J 19:1777–1783
Schägger H, Pfeiffer K (2001) The ratio of oxidative phosphorylation complexes I–V in bovine heart mitochondria and the composition of respiratory chain supercomplexes. J Biol Chem 276:37861–37867
Schmelzer C, Döring F (2010) Identification of LPS-inducible genes downregulated by ubiquinone in human THP-1 monocytes. Biofactors 36:222–228
Schmelzer C, Kitano M, Rimbach G et al (2009) Effects of ubiquinol-10 on microRNA-146a expression in vitro and in vivo. Mediators Inflamm 2009:415437
Schneider H, Lemasters JJ, Hackenbrock CR (1982) Lateral diffusion of ubiquinone during electron transfer in phospholipid- and ubiquinone-enriched mitochondrial membranes. J Biol Chem 257:10789–10793
Schönfeld P, Wieckowski MR, Lebiedzińska M et al (2010) Mitochondrial fatty acid oxidation and oxidative stress: lack of reverse electron transfer-associated production of reactive oxygen species. Biochim Biophys Acta 1797:929–938
Schwerzmann K, Cruz-Orive LM, Eggman R et al (1986) Molecular architecture of the inner membrane of mitochondria from rat liver: a combined biochemical and stereological study. J Cell Biol 102:97–103
Seelert H, Dani DN, Dante S et al (2009) From protons to OXPHOS supercomplexes and Alzheimer’s disease: structure-dynamics-function relationships of energy-transducing membranes. Biochim Biophys Acta 1787:657–671
Sherwood S, Hirst J (2006) Investigation of the mechanism of proton trasnslocation by NADH:ubiquinone oxidoreductase (complex I) from bovine heart mitochondria: does the enzyme operate by a Q-cycle mechanism? Biochem J 400:541–550
Shinzawa-Itoh K, Shimomura H, Yanagisawa S et al (2016) Purification of active respiratory supercomplex from bovine heart mitochondria enables functional studies. J Biol Chem 291:4178–4184
Singer SJ, Nicolson GL (1972) The fluid mosaic model of the structure of cell membranes. Science 175:720–731
Sohet FM, Neyrinck AM, Pachikian BD et al (2009) Coenzyme Q10 supplementation lowers hepatic oxidative stress and inflammation associated with diet-induced obesity in mice. Biochem Pharmacol 78:1391–1400
Stoner CD (1984) Steady-state kinetics of the overall oxidative phosphorylation reaction in heart mitochondria. Determination of the coupling relationships between the respiratory reactions and miscellaneous observations concerning rate-limiting steps. J Bioenerg Biomembr 16:115–141
Stroh A, Anderka O, Pfeiffer K et al (2004) Assembly of respiratory complexes I, III, and IV into NADH oxidase supercomplex stabilizes complex I in Paracoccus denitrificans. J Biol Chem 279:5000–5007
Sun F, Huo X, Zhai Y et al (2005) Crystal structure of mitochondrial respiratory membrane protein complex II. Cell 121:1043–1057
Szarkowska L (1966) The restoration of DPNH oxidase activity by coenzyme Q (ubiquinone). Arch Biochem Biophys 113:519–525
Takahashi T, Yamaguchi T, Shitashige M et al (1995) Reduction of ubiquinone in membrane lipids by rat liver cytosol and its involvement in the cellular defence system against lipid peroxidation. Biochem J 309:883–890
Tan AK, Ramsay RR, Singer TP et al (1993) Comparison of the structures of the quinone-binding sites in beef heart mitochondria. J Biol Chem 268:19328–19333
Tian G, Sawashita J, Kubo H et al (2014) Ubiquinol-10 supplementation activates mitochondria functions to decelerate senescence in senescence-accelerated mice. Antioxid Redox Signal 20:2606–2620
Tocilescu MA, Zickermann V, Zwicker K et al (2010) Quinone binding and reduction by respiratory complex I. Biochim Biophys Acta 1797:1883–1890
Van Raam BJ, Sluiter W, de Wit E et al (2008) Mitochondrial membrane potential in human neutrophils is maintained by complex III activity in the absence of supercomplex organisation. PLoS One 3:e2013
Vanderkooi G (1978) Organization of protein and lipid components in membranes. In: Fleischer S, Hatefi Y, MacLennan D, Tzagoloff A (eds) Molecular biology of membranes. Plenum Publishing Corp, New York, pp 25–55
Various authors (2015) In: Louro RO, Diaz-Moreno I (eds) Redox proteins in supercomplexes and signalosomes. CRC Press, Boca Raton
Vinogradov AD (2001) Respiratory complex I: structure, redox components, and possible mechanisms of energy transduction. Biochemistry (Mosc) 66:1086–1097
Walter L, Nogueira V, Leverve X et al (2000) Three classes of ubiquinone analogs regulate the mitochondrial permeability transition pore through a common site. J Biol Chem 275:29521–29527
Walter L, Miyoshi H, Leverve X et al (2002) Regulation of the mitochondrial permeability transition pore by ubiquinone analogs. A progress report. Free Radic Res 36:405–412
Wang Y, Hekimi S (2016) Understanding ubiquinone. Trends Cell Biol 26:367–378
Xia D, Yu CA, Kim H et al (1997) Crystal structure of the cytochrome bc1 complex from bovine heart mitochondria. Science 277:60–66
Yamamura T, Otani H, Nakao Y et al (2001) Dual involvement of coenzyme Q10 in redox signaling and inhibition of death signaling in the rat heart mitochondria. Antioxid Redox Signal 3:103–112
Yano T, Ohnishi T (2001) The origin of cluster N2 of the energy-transducing NADH-quinone oxidoreductase: comparisons of phylogenetically related enzymes. J Bioenerg Biomembr 33:213–222
Zhang Z, Huang L, Shulmeister VM et al (1998) Electron transfer by domain movement in cytochrome bc1. Nature 392:677–684
Zhang M, Mileykovskaya E, Dowhan W (2002) Gluing the respiratory chain together Cardiolipin is required for supercomplex formation in the inner mitochondrial membrane. J Biol Chem 277:43553–43556
Zhu QS, Berden JA, De Vries S et al (1982) On the role of ubiquinone in the respiratory chain. Biochim Biophys Acta 680:69–79
Zickermann V, Dröse S, Tocilescu MA et al (2008) Challenges in elucidating structure and mechanism of proton pumping NADH:ubiquinone oxidoreductase (complex I). J Bioenerg Biomembr 40:475–483
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Genova, M.L., Lenaz, G. (2020). Coenzyme Q Function in Mitochondria. In: López Lluch, G. (eds) Coenzyme Q in Aging. Springer, Cham. https://doi.org/10.1007/978-3-030-45642-9_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-45642-9_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-45641-2
Online ISBN: 978-3-030-45642-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)