Abstract
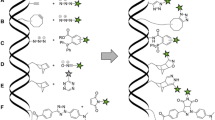
Oligonucleotide conjugates have already reached considerable importance in life science research and oligonucleotide drug development. Since the preparation of oligonucleotide conjugates depends critically on the chemical nature of the used ligand and linker, there is no general and universal procedure. Here, we present a detailed, quick, and facile protocol for attaching fluorescent dyes or cross-linkers of variable chemical stability to oligonucleotides at 3′- or 5′-aminoalkyl handles. Purification and removal of educts and side-products and structural verification by gel electrophoresis and mass spectrometry are presented. Aspects for adapting this protocol for other reaction sites at the oligonucleotide are discussed. We highlight important issues for generating oligonucleotide conjugates with other molecules, including peptide, proteins, and small molecules for receptor-targeting applications. The methodology is suitable for oligonucleotides with various modifications, including stabilized antisense, siRNAs, and miRNAs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Khvorova A, Watts JK (2017) The chemical evolution of oligonucleotide therapies of clinical utility. Nat Biotechnol 35(3):238–248. https://doi.org/10.1038/nbt.3765
Baumann V, Lorenzer C, Thell M, Winkler A-M, Winkler J (2017) RNAi-mediated knockdown of protein expression. Methods Mol Biol 1654:351–360. https://doi.org/10.1007/978-1-4939-7231-9_26
Dirin M, Winkler J (2013) Influence of diverse chemical modifications on the ADME characteristics and toxicology of antisense oligonucleotides. Expert Opin Biol Ther 13(6):875–888. https://doi.org/10.1517/14712598.2013.774366
Baumann V, Winkler J (2014) miRNA-based therapies: strategies and delivery platforms for oligonucleotide and non-oligonucleotide agents. Future Med Chem 6(17):1967–1984. https://doi.org/10.4155/fmc.14.116
Asseline U (2006) Development and applications of fluorescent oligonucleotides. Curr Org Chem 10(4):491–518. https://doi.org/10.2174/138527206776055349
Winkler J (2013) Oligonucleotide conjugates for therapeutic applications. Ther Deliv 4(7):791–809. https://doi.org/10.4155/tde.13.47
Patel PL, Rana NK, Patel MR, Kozuch SD, Sabatino D (2016) Nucleic acid bioconjugates in cancer detection and therapy. ChemMedChem 11(3):252–269. https://doi.org/10.1002/cmdc.201500502
Lorenzer C, Dirin M, Winkler A-M, Baumann V, Winkler J (2015) Going beyond the liver: progress and challenges of targeted delivery of siRNA therapeutics. J Control Release 203:1–15. https://doi.org/10.1016/j.jconrel.2015.02.003
Ming X, Laing B (2015) Bioconjugates for targeted delivery of therapeutic oligonucleotides. Adv Drug Deliv Rev 87(0):81–89. https://doi.org/10.1016/j.addr.2015.02.002
Nair JK, Willoughby JLS, Chan A, Charisse K, Alam MR, Wang Q, Hoekstra M, Kandasamy P, Kel’in AV, Milstein S, Taneja N, O’Shea J, Shaikh S, Zhang L, van der Sluis RJ, Jung ME, Akinc A, Hutabarat R, Kuchimanchi S, Fitzgerald K, Zimmermann T, van Berkel TJC, Maier MA, Rajeev KG, Manoharan M (2014) Multivalent N-acetylgalactosamine-conjugated siRNA localizes in hepatocytes and elicits robust RNAi-mediated gene silencing. J Am Chem Soc 136(49):16958–16961. https://doi.org/10.1021/ja505986a
Prakash TP, Graham MJ, Yu J, Carty R, Low A, Chappell A, Schmidt K, Zhao C, Aghajan M, Murray HF, Riney S, Booten SL, Murray SF, Gaus H, Crosby J, Lima WF, Guo S, Monia BP, Swayze EE, Seth PP (2014) Targeted delivery of antisense oligonucleotides to hepatocytes using triantennary N-acetyl galactosamine improves potency 10-fold in mice. Nucleic Acids Res 42(13):8796–8807. https://doi.org/10.1093/nar/gku531
Winkler J (2015) Therapeutic oligonucleotides with polyethylene glycol modifications. Future Med Chem 7(13):1721–1731. https://doi.org/10.4155/fmc.15.94
Gaziova Z, Baumann V, Winkler A-M, Winkler J (2014) Chemically defined polyethylene glycol siRNA conjugates with enhanced gene silencing effect. Bioorg Med Chem 22(7):2320–2326. https://doi.org/10.1016/j.bmc.2014.02.004
Shokrzadeh N, Winkler A-M, Dirin M, Winkler J (2014) Oligonucleotides conjugated with short chemically defined polyethylene glycol chains are efficient antisense agents. Bioorg Med Chem Lett 24(24):5758–5761. https://doi.org/10.1016/j.bmcl.2014.10.045
Winkler J, Urban E, Noe C (2005) Oligonucleotides conjugated to short lysine chains. Bioconjug Chem 16(4):1038–1044. https://doi.org/10.1021/bc049729d
Winkler J, Gilbert M, Kocourkova A, Stessl M, Noe C (2008) 2 '-O-lysylaminohexyl oligonucleotides: modifications for antisense and siRNA. ChemMedChem 3(1):102–110. https://doi.org/10.1002/cmdc.200700169
Winkler J, Saadat K, Diaz-Gavilan M, Urban E, Noe C (2009) Oligonucleotide-polyamine conjugates: influence of length and position of 2 '-attached polyamines on duplex stability and antisense effect. Eur J Med Chem 44(2):670–677. https://doi.org/10.1016/j.ejmech.2008.05.012
Dirin M, Urban E, Lachmann B, Noe CR, Winkler J (2015) Concise postsynthetic preparation of oligonucleotide-oligopeptide conjugates through facile disulfide bond formation. Future Med Chem 7(13):1657–1673. https://doi.org/10.4155/fmc.15.109
Dirin M, Urban E, Noe CR, Winkler J (2016) Fragment-based solid-phase assembly of oligonucleotide conjugates with peptide and polyethylene glycol ligands. Eur J Med Chem 121:132–142. https://doi.org/10.1016/j.ejmech.2016.05.001
Winkler J (2011) Nanomedicines based on recombinant fusion proteins for targeting therapeutic siRNA oligonucleotides. Ther Deliv 2(7):891–905. https://doi.org/10.4155/tde.11.56
Lonnberg H (2009) Solid-phase synthesis of oligonucleotide conjugates useful for delivery and targeting of potential nucleic acid therapeutics. Bioconjug Chem 20(6):1065–1094. https://doi.org/10.1021/bc800406a
Shah S, Friedman SH (2008) An ESI-MS method for characterization of native and modified oligonucleotides used for RNA interference and other biological applications. Nature Prot 3(3):351–356
SantaLucia J Jr (1998) A unified view of polymer, dumbbell, and oligonucleotide DNA nearest-neighbor thermodynamics. Proc Natl Acad Sci U S A 95(4):1460–1465
Kibbe WA (2007) OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res 35(suppl_2):W43–W46. https://doi.org/10.1093/nar/gkm234
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Lorenzer, C., Winkler, J. (2019). Covalent Fluorophore Labeling of Oligonucleotides and Generation of Other Oligonucleotide Bioconjugates. In: Ogris, M., Sami, H. (eds) Nanotechnology for Nucleic Acid Delivery. Methods in Molecular Biology, vol 1943. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-9092-4_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9092-4_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-9091-7
Online ISBN: 978-1-4939-9092-4
eBook Packages: Springer Protocols