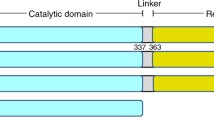
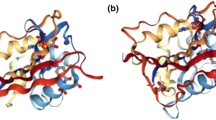
Abstract
Naturally occurring pteridines in eukaryotic systems usually contain 2-amino, 4-hydroxy 6-alkyl substituents composed of either a methylene-p-aminobenzoylglutamate (or polutamate) or a dihydroxypropyl group. The former class are known collectively as the folates and occur widely as reduced and 5-alkylated derivatives, in which form they participate in important metabolic one-carbon transfers (Blakley, 1984). The latter, known as biopterin,, also occurs in reduced forms and is an important cofactor in aromatic amino acid hydroxylations en route to the catecholamines (Shiman, 1985; Kaufman and Kaufman, 1985; n and Lovenberg, 1985) and in the nitrite synthase pathway (Marletta,, 1993). Dihydrofolate reductase (DHFR), dihydropteridine reductase (DHPR) and pteridine reductase (PTR1), best characterised from Leishmania, are three enzymes that initiate the reduction of a pteridine in association with a reduced dinucleotide cofactor. Their comparative reaction pathways are illustrated in Figure 1.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Armarego, W. L. F. (1979) Hydrogen transfer from 4-R and 4-S (4-3H) NADH in the reduction of d,l-cis-6,1-di-methyl-6,7 (8H)-dihydropterin with dihydropteridine reductase from human liver and sheep liver. Biochem. Biophys. Res. Commun. 89, 246–249.
Baccanari, D., Phillips, A., Smith, S., Sinski, D., and Burchall, J. (1975) Purification and properties of Escherichia coli dihydrofolate reductase. Biochemistry 14, 5267–5273.
Blakley, R. L. (1984) Benkovic, S. J. New York, John Wiley & Sons
Bystroff, C, Oatley, S. J., and Kraut, J. (1990) Crystal structure of Escherichia coli dihydrofolate reductase: the NADP+ holoenzyme and the folate-NADP+ ternary complex. Substrate binding and a model for the transition state. Biochemistry 29, 3263–3277.
Chang, G., Shiao, M., Liaw, J., and Lee, H. (1989) Periodate-oxidized 3-aminopyridine adenine dinucleotide phosphate as a fluorescent affinity lable for pigeon liver malic enzyme. J. Biol Chem. 264, 280–287.
Färber, S., Diamond, L. K., Mercer, R. D., Sylvester, R. F., Jr., and Wolff, J. A. (1948) Temporary remissions in acute leukemia in children. N. E. J. Med. 238, 787–793.
Filman, D. J., Bolin, J. T., Matthews, D. A., and Kraut, J. (1982) Crystal structures of Escherichia coli and Lactoba-cillus casei dihydrofolate reductase refined at 1. 7 A resolution. II. Environment of bound NADPH and implications for catalysis. J Biol Chem 257, 13663–13672.
Jörnvall, H., Persson, B., Krook, M., Atrian, S., Gonzàlez-Duarte, R., Jeffery, J., and Ghosh, D. (1995) Short-chain dehydrogenases/reductases (SDR). Biochemistry 34, 6003–6013.
Kaufman, S. and Kaufman, E. E. (1985) Tyrosine Hydroxylase. In: Folates and Pterins, Vol. 2, 251–352. Edited by Blakley, R. L. and Benkovic, S. J. New York, Wiley Interscience.
Kuhn, D. M. and Lovenberg, W. (1985) Tryptophan Hydroxylase. In: Folates and Pterins, Vol. 2, 353–382. Edited by Blakley, R. L. and Benkovic, S. J. New York, Wiley Interscience.
Luba, J., Nare, B., Liang, P.-H., Anderson, K. S., Beverley, S. M., and Hardy, L. (1998) Leishmania major pteridine reductase 1 belongs to the short chain dehydrogenase family: Stereochemical and kinetic evidence. Biochemistry 37, 4093–4104.
Marietta, M. A. (1993) Nitric oxide synthase structure and mechanism. J. Biol. Chem. 268, 12231–12234.
Nare, B., Hardy, L. W., and Beverley, S. M. (1997) The roles of pteridine reductase 1 and dihydrofolate reductase-thymidylate synthase in pteridine metabolism in the protozoan parasite Leishmania major. J. Biol. Chem. 272, 13883–138
Rosenkranz-Weiss, P., Sessa, W. C., Milstien, S., Kaufman, S., Watson, C. A., and Prober, J. S. (1994) Regulation of nitric oxide synthesis by proinflammatory cytokines in human umbilical vein endothelial cells. Elevations in tetrahydrobiopterin levels enhance endothelial nitric oxide synthase specific activity. J. Clin. Invest. 93, 2236–2243.
Shiman, R. (1985) Phenylalanine Hydroxylase and Dihydropteridine Reductase. In: Folates and Pterins, Vol. 2, 179–249. Edited by Blakley, R. L. and Benkovic, S. J. New York, Wiley Interscience.
Varughese, K. I., Skinner, M. M., Whiteley, J. M., Matthews, D. A., and Xuong, N. H. (1992) Crystal structure of rat liver dihydropteridine reductase. Proc. Natl. Acad. Sci., USA 89, 6080–6084.
Varughese, K. I., Xuong, N. H., Kiefer, P. M., Matthews, D. A., and Whiteley, J. M. (1994) Structural and mechanistic characteristics of dihydropteridine reductase: A member of the Tyr-(Xaa)3-Lys-containing family of re-ductases and dehydrogenases. Proc. Natl. Acad. Sci.,USA 91, 5582–5586.
Wang, J. Y., Leblanc, E., Chang, CF., Papadopoulou, B., Bray, T., Whiteley, J. M., Lin, S. X., and Ouellette, M. (1997) Pterin and folate reduction by the Leishmania tarentolae H locus short-chain dehydrogenase/reduc-tase PTR1. Arch. Biochem. Biophys. 342, 197–202.
Whiteley, J. M., Varughese, K. I., Xuong, N. H., Matthews, D. A., and Grimshaw, C. E. (1993) Dihydropteridine reductase. Pteridines 4, 159–173.
Wierenga, R. K., Terpstra, P., and Hol, W. G. J. (1986) Prediction of the occurrence of the ADP-binding βαβ-fold in proteins, using an amino acid sequence fingerprint. J. Mol. Biol. 187, 101–107.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer Science+Business Media New York
About this chapter
Cite this chapter
Chang, CF., Bray, T., Varughese, K.I., Whiteley, J.M. (1999). Comparative Properties of Three Pteridine Reductases. In: Weiner, H., Maser, E., Crabb, D.W., Lindahl, R. (eds) Enzymology and Molecular Biology of Carbonyl Metabolism 7. Advances in Experimental Medicine and Biology, vol 463. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-4735-8_50
Download citation
DOI: https://doi.org/10.1007/978-1-4615-4735-8_50
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-7146-5
Online ISBN: 978-1-4615-4735-8
eBook Packages: Springer Book Archive