Abstract
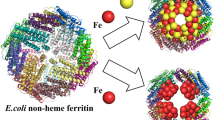
The toxic effects of the essential metal, iron, are controlled within cells through its being bound by proteins or organic chelators [Halliwell and Gutteridge, 1986]. Within mammals, most intracellular iron not utilized for heme-containing enzymes is bound by the iron-storage protein, ferritin [Harrison et al., 1987; Theil, 1987; Joshi and Zimmerman, 1988]. Up to several thousand atoms of ferric iron can be contained within each ferritin molecule as one or more crystallites of the biomineral, ferrihydrite (5Fe2O3·9H2O) [Towe, 1981; Ford et al., 1984]. Unlike commonplace rust, the “biological iron oxide” within ferritin remains soluble. This very special and important capability is a direct consequence of the quaternary structure of the metalloprotein; in each ferritin molecule, the insoluble ferrihydrite forms a core that is contained inside an outer assembly of 24 polypeptide subunits (Mr≈ 20kDa) [Ford et al., 1984]. The core crystallites within the central cavity are attached to the surrounding protein shell [Stuhrmann et al., 1976; Massover, 1978].
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Andersen, M. E., 1971, Sedimentation equilibrium experiments on the self-association of hemoglobin from the lamprey Petromyzon marinus. A model for oxygen transport in the lamprey, J. Biol. Chem., 246:4800–4806.
Bakker, G. R. and Boyer, R. F., 1986, Iron incorporation into apoferritin. The role of apoferritin as a ferroxidase, J. Biol. Chem., 261:13182–13185.
Bolann, B. J. and Ulvik, R. J., 1987, Release of iron from ferritin by xanthine oxidase. Role of the superoxide radical, Biochem. J., 243:55–59.
Bonomi, F. and Pagani, S., 1986, Removal of ferritin-bound iron by DL-dihydrolipoate and DL-dihydrolipoamide, Europ. J. Biochem., 155, 295–300.
Bonomi, F., Cerioli, A. and Pagani, S., 1989, Molecular aspects of the removal of ferritin-bound iron by DL-dihydrolipoate, Biochim. Biophys. Acta., 994:180–186.
Briehl, R. W., 1963, The relation between the oxygen equilibrium and aggregation of subunits in lamprey hemoglobin, J. Biol. Chem., 238:2361–2366.
Crichton, R. R., Roman, F., Roland, F., Paques, E., Paques, A. and Vandamme, E., 1980, Ferritin iron deposition and mobilisation, J. Mol. Catal., 7:267–276.
Dognin, J., Girardet, J. L. and Chapron, Y., 1973, Etude polarographique de la mobilisation du fer de la ferritine, Biochim. Biophys. Acta., 297:276–284.
Farrant, J. L., 1954, An electron microscopic study of ferritin, Biochim. Biophys. Acta., 13:569–576.
Ford, G. C., Harrison, P. M., Rice, D. W., Smith, J. M. A., Treffry, A., White, J. L. and Yariv, J., 1984, Ferritin: design and formation of an iron-storage molecule, Phil. Trans. Roy. Soc. Lond., 304B:551–565.
Halliwell, B. and Gutteridge, J. M. C., 1986, Iron and free radical reactions: two aspects of antioxidant protection, TIBS. 11:372–375.
Harris, D. C., 1978, Iron exchange between ferritin and transferrin in Vitro, Biochem., 17:3071–3078.
Harrison, P. M., Clegg, G. A. and May, K., 1980, Ferritin structure and function, In: “Iron in Biochemistry and Medicine, II”, A. Jacobs and M. Worwood, eds., Academic Press, London, pp. 131–171.
Harrison, P. M., Ford, G. C., Rice, D. W., Smith, J. M. A., Treffry, A. and White, J. L., 1987, Structural and functional studies on ferritins, Biochem. Soc. Trans., 15:744–748.
Hesterberg, L. K. and Lee, J. C., 1982, Self-association of rabbit muscle phosphofructokinase: effect of ligands, Biochem., 21:216–222.
Inoue, S. and Sato, H., 1967, Cell motility by labile association of molecules. The nature of mitotic spindle fibers and their role in chromosome movement, J. Gen Phvsiol., 50:259–288.
Jacobs, D. L., Watt, G. D., Frankel, R. B. and Papaefthymiou, G. C., 1989, Redox reactions associated with iron release from mammalian ferritin, Biochem., 28:1650–1655.
Jones, M. M. and Johnston, D. O., 1967, Rate of release of iron from ferritin to 1,10-phenanthroline, Nature, 216:509–510.
Jones, T., Spencer, R. and Walsh, C., 1978, Mechanism and kinetics of iron release from ferritin by dihydro-flavins and dihydroflavin analogues, Biochem., 17: 4011–4017.
Joshi, J. G. and Zimmerman, A., 1988, Ferritin: an expanded role in metabolic regulation, Toxicol., 48:21–29.
Lee, J. C. K., Lee, S. S., Schlesinger, K. J. and Richter, G. W., 1974, Detection of protein subunits of ferritin in situ in cells by immunofluorescence, Amer. J. Pathol., 75:473–487.
Levi, S., Luzzago, A., Cesareni, G., Cozzi, A., Franceschinelli, F., Albertini, A. and Arosio, P., 1988, Mechanism of ferritin iron uptake: activity of the H-chain and deletion mapping of the ferro-oxidase site. A study of iron uptake and ferro-oxidase activity of human liver, recombinant H-chain ferritins, and of two H-chain deletion mutants, J. Biol. Chem., 263:18086–18092.
Linder, M. C., Nagel, G. M., Roboz, M. and Hungerford, Jr., D. M., 1981, The size and shape of heart and muscle ferritins analyzed by sedimentation, gel filtration, and electrophoresis, J. Biol. Chem., 256:9104–9110.
Luzzago, A., Arosio, P., Iacobello, C., Ruggeri, G., Capucci, L., Brocchi, E., De Simone, F., Gamba, D., Gabri, E., Levi, S., and Albertini, A., 1986, Immunochemical characterization of human liver and heart ferritins with monoclonal antibodies, Biochim. Biophys. Acta., 872:61–71.
Margolis, R. L. and Wilson, L., 1978, Opposite end assembly and disassembly of microtubules at steady state in vitro, Cell, 13:1–8.
Massover, W. H., 1978, The ultrastructure of ferritin macro-molecules. III. Mineralized iron in ferritin is attached to the protein shell, J. Mol. Biol., 123: 721–726.
Massover, W. H., 1979a, Ultrastructural studies on the dissociation of apoferritin into subunits, In: “Proc. 37th. Ann. Meet. Electron Micr. Soc. America”, G. W. Bailey, ed., Claitor’s Publishing Division, Baton Rouge, pp. 22–23.
Massover, W. H., 1979b, The resolution of individual protein subunits in ferritin, In: “Electron Microscopy 1978”, J. M. Sturgess, ed., Microscopical Society of Canada, Toronto, 11:182–183.
Massover, W. H., 1980, Ultrastructural evidence for the dissociation of free subunits from ferritin upon dilution, Biochem. Biophys. Res. Comm., 96:1427–1433.
Massover, W. H. and Cowley, J. M., 1975, Ultrastructure of ferritin and apoferritin, In: “Proteins of Iron Storage and Transport in Biochemistry and Medicine”, R. R. Crichton, ed., North-Holland Publishing Co., Amsterdam, pp. 237–244.
Mazur, A., Baez, S. and Shorr, E., 1955, The mechanism of iron release from ferritin as related to its biological properties, J. Biol. Chem., 213:147–160.
Mitchison, T. and Kirschner, M., 1984, Dynamic instability of microtubule growth, Nature, 312:237–242.
Niederer, W., 1970, Ferritin: iron incorporation and iron release, Experientia. 26:218–220.
Pape, L., Multani, J. S., Stitt, C. and Saltman, P., 1968, In Vitro reconstitution of ferritin, Biochem., 7:606–612.
Stefanini, S., Desideri, A., Vecchini, P., Drakenberg, T. and Chiancone, E., 1989, Identification of the iron entry channels in apoferritin. Chemical modification and spectroscopic studies, Biochem., 28:378–382.
Stuhrmann, H. B., Haas, J., Ibel, K., Koch, M. H. J. and Crichton, R. R., 1976, Low angle neutron scattering of ferritin studied by contrast variation, J. Mol. Biol., 100:399–413.
Theil, E. C., 1987, Ferritin: structure, gene regulation, and cellular function in animals, plants, and microorganisms, Ann. Rev. Biochem., 56:289–315.
Towe, K. M., 1981, Structural distinction between ferritin and iron-dextran (Imferon). An electron diffraction comparison, J. Biol. Chem., 256:9377–9378.
Watt, G. D., Jacobs, D. and Frankel, R. B., 1988, Redox reactivity of bacterial and mammalian ferritin: Is reductant entry into the ferritin interior a necessary step for iron release? Proc. Nat. Acad. Sci. USA., 85:7457–7461.
Yamamoto, S. and Storey, K. B., 1988, Dissociation-association of lactate dehydrogenase isozymes: influences on the formation of tetramers versus dimers of M4-LDH and H4-LDH, Int. J. Biochem., 20:1261–1265.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1991 Springer Science+Business Media New York
About this chapter
Cite this chapter
Massover, W.H. (1991). Dynamic Stability of Apoferritin: A New Model to Explain How Impermeable Reagents Can Reduce/Capture Iron within Ferritin. In: Frankel, R.B., Blakemore, R.P. (eds) Iron Biominerals. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-3810-3_25
Download citation
DOI: https://doi.org/10.1007/978-1-4615-3810-3_25
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6699-7
Online ISBN: 978-1-4615-3810-3
eBook Packages: Springer Book Archive