Abstract
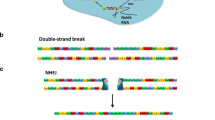
Ex-vivo gene therapy can entail either the replacement or the addition of genes. In gene addition therapy, a therapeutic gene is inserted directly into the host genome, with the abnormal gene remaining intact. In gene replacement therapy, the genome is modified directly. Homologous recombination technology can be used to perform many of these kinds of gene correction. In the past, gene correction therapy has been hampered by the low efficiency of the recombination event. However, recently engineered zinc finger nucleases (ZFNs) were found to have the ability to successfully stimulate homologous recombination by inducing double-strand breaks at specific DNA sites. Another class of enzyme, the transcription activator-like effector nucleases (TALENs), provides an efficient alternative means to induce specific DNA double-strand at breaks. Meanwhile, newly developed gene correction methods using stem cells and induced pluripotent stem (iPS) cells have made gene therapy more feasible in clinical practice. Cells are taken from patients, harvested, and transformed through induction into stem cells, which have the potential to differentiate into a variety of mature cells types for transplant. Further research is needed to develop gene therapy, which may be used in tandem with embryonic and induced pluripotent stem cell therapy, especially to repair preexisting mutations that may be passed on in iPS cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hockemeyer D, Wang H, Kiani S, Lai CS, Gao Q, Cassady JP, Cost GJ, Zhang L, Santiago Y, Miller JC et al (2011) Genetic engineering of human pluripotent cells using TALE nucleases. Nat Biotechnol 29:731–734
Cavazzana-Calvo M, Thrasher A, Mavilio F (2004) The future of gene therapy. Nature 427:779–781
Townes TM (2008) Gene replacement therapy for sickle cell disease and other blood disorders. Am Soc Hematol Educ Program 1:193–196
Thomson JG, Ow DW (2006) Site-specific recombination systems for the genetic manipulation of eukaryotic genomes. Genesis 44:465–476
Schwartzberg PL, Goff SP, Robertson EJ (1989) Germ-line transmission of a c-abl mutation produced by targeted gene disruption in ES cells. Science 246:799–803
Cui X, Ji D, Fisher DA, Wu Y, Briner DM, Weinstein EJ (2011) Targeted integration in rat and mouse embryos with zinc-finger nucleases. Nat Biotechnol 29:64–67
Li MA, Bradley A (2011) Crafting rat genomes with zinc fingers. Nat Biotechnol 29:39–41
Chang RLJ, Xu SM, Meneses J, Chan K, Pedersen R, Kan YW (1996) Inactivation of mouse alpha-globin gene by homologous recombination: mouse model of hemoglobin H disease. Blood 88:1846–1851
Chang Tong PL, Wu NL, Yan Y, Ying QL (2010) Production of p53 gene knockout rats by homologous recombination in embryonic stem cells. Nature 467:211–215
Soldner F, Laganiere J, Cheng AW, Hockemeyer D, Gao Q, Alagappan R, Khurana V, Golbe LI, Myers RH, Lindquist S et al (2011) Generation of isogenic pluripotent stem cells differing exclusively at two early-onset Parkinson point mutations. Cell 146:318–331
Zwaka TP, Thomson JA (2003) Homologous recombination in human embryonic stem cells. Nat Biotechnol 21:319–321
Ruby KM, Zheng B (2009) Gene targeting in a HUES line of human embryonic stem cells via electroporation. Stem Cells 27:1496–1506
Irion S, Luche H, Gadue P, Fehling HJ, Kennedy M, Keller G (2007) Identification and targeting of the ROSA26 locus in human embryonic stem cells. Nat Biotechnol 25:1477–1482
Xue H, Wu S, Papadeas ST, Spusta S, Swistowska AM, MacArthur CC, Mattson MP, Maragakis NJ, Capecchi MR, Rao MS, Zeng X, Liu Y (2009) A targeted neuroglial reporter line generated by homologous recombination in human embryonic stem cells. Stem Cells 27:1836–1846
Davis RP, Ng ES, Costa M, Mossman AK, Sourris K, Elefanty AG, Stanley EG (2008) Targeting a GFP reporter gene to the MIXL1 locus of human embryonic stem cells identifies human primitive streak-like cells and enables isolation of primitive hematopoietic precursors. Blood 111:1876–1884
Costa M, Dottori M, Sourris K, Jamshidi P, Hatzistavrou T, Davis R, Azzola L, Jackson S, Lim SM, Pera M et al (2007) A method for genetic modification of human embryonic stem cells using electroporation. Nat Protoc 2:792–796
Urbach A, Schuldiner M, Benvenisty N (2004) Modeling for Lesch-Nyhan disease by gene targeting in human embryonic stem cells. Stem Cells 22:635–641
Doetschman T, Gregg RG, Maeda N, Hooper ML, Melton DW, Thompson S, Smithies O (1987) Targeted correction of a mutant HPRT gene in mouse embryonic stem cells. Nature 330:576–578
Porteus MH, Carroll D (2005) Gene targeting using zinc finger nucleases. Nat Biotechnol 23:967–973
Song H, Chung SK, Xu Y (2010) Modeling disease in human ESCs using an efficient BAC-based homologous recombination system. Cell Stem Cell 6:80–89
Perez EE, Wang J, Miller JC, Jouvenot Y, Kim KA, Liu O, Wang N, Lee G, Bartsevich VV, Lee YL, Guschin DY, Rupniewski I, Waite AJ, Carpenito C, Carroll RG, Orange JS, Urnov FD, Rebar EJ, Ando D, Gregory PD, Riley JL, Holmes MC, June CH (2008) Establishment of HIV-1 resistance in CD4+ T cells by genome editing using zinc-finger nucleases. Nat Biotechnol 26:808–816
Lombardo A, Genovese P, Beausejour CM, Colleoni S, Lee YL, Kim KA, Ando D, Urnov FD, Galli C, Gregory PD, Holmes MC, Naldini L (2007) Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nat Biotechnol 25:1298–1306
Brenneman M, Gimble FS, Wilson JH (1996) Stimulation of intrachromosomal homologous recombination in human cells by electroporation with site-specific endonucleases. Proc Natl Acad Sci U S A 93:3608–3612
Choulika A, Perrin A, Dujon B, Nicolas JF (1995) Induction of homologous recombination in mammalian chromosomes by using the I-SceI system of Saccharomyces cerevisiae. Mol Cell Biol 15:1968–1973
Rouet P, Smih F, Jasin M (1994) Introduction of double-strand breaks into the genome of mouse cells by expression of a rare-cutting endonuclease. Mol Cell Biol 14:8096–8106
Donoho G, Jasin M, Berg P (1998) Analysis of gene targeting and intrachromosomal homologous recombination stimulated by genomic double-strand breaks in mouse embryonic stem cells. Mol Cell Biol 18:4070–4078
Porteus M, Baltimore D (2003) Chimeric nucleases stimulate gene targeting in human cells. Science 300:763
Morton J, Davis MW, Jorgensen EM, Carroll D (2006) Induction and repair of zinc-finger nuclease-targeted double-strand breaks in Caenorhabditis elegans somatic cells. Proc Natl Acad Sci U S A 103:16370–16375
Meng X, Noyes MB, Zhu LJ, Lawson ND, Wolfe SA (2008) Targeted gene inactivation in zebrafish using engineered zinc-finger nucleases. Nat Biotechnol 26:695–701
Doyon Y, McCammon JM, Miller JC, Faraji F, Ngo C, Katibah GE, Amora R, Hocking TD, Zhang L, Rebar EJ, Gregory PD, Urnov FD, Amacher SL (2008) Heritable targeted gene disruption in zebrafish using designed zinc-finger nucleases. Nat Biotechnol 26:702–708
Maeder ML, Thibodeau-Beganny S, Sander JD, Voytas DF, Joung JK (2009) Oligomerized pool engineering (OPEN): an ‘open-source’ protocol for making customized zinc-finger arrays. Nat Protoc 4:1471–1501
Sander JD, Dahlborg EJ, Goodwin MJ, Cade L, Zhang F, Cifuentes D, Curtin SJ, Blackburn JS, Thibodeau-Beganny S, Qi Y et al (2011) Selection-free zinc-finger-nuclease engineering by context-dependent assembly (CoDA). Nat Methods 8:67–69
Bogdanove AJ, Schornack S, Lahaye T (2010) TAL effectors: finding plant genes for disease and defense. Curr Opin Plant Biol 13:394–401
Romer P, Recht S, Strauss T, Elsaesser J, Schornack S, Boch J, Wang S, Lahaye T (2010) Promoter elements of rice susceptibility genes are bound and activated by specific TAL effectors from the bacterial blight pathogen Xanthomonas oryzae pv. oryzae. New Phytol 187:1048–1057
Boch J, Scholze H, Schornack S, Landgraf A, Hahn S, Kay S, Lahaye T, Nickstadt A, Bonas U (2009) Breaking the code of DNA binding specificity of TAL-type III effectors. Science 326:1509–1512
Christian M, Cermak T, Doyle EL, Schmidt C, Zhang F, Hummel A, Bogdanove AJ, Voytas DF (2010) Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186:757–761
Cermak T, Doyle EL, Christian M, Wang L, Zhang Y, Schmidt C, Baller JA, Somia NV, Bogdanove AJ, Voytas DF (2011) Efficient design and assembly of custom TALEN and other TAL effector-based constructs for DNA targeting. Nucleic Acids Res 39:e82
Doerfler W (1996) Adenoviruses. In: Baron S (ed) Medical microbiology, 4th edn. University of Texas, Galveston
Pacchia A, Mukherjee S, Dougherty JP (2003) Choice and use of appropriate packaging cell types. In: Federico M (ed) Lentivirus gene engineering protocols, 3rd edn. Humana Press, Totowa
Whitley RJ (1996) Herpesviruses. In: Baron S (ed) Medical microbiology, 4th edn. University of Texas, Galveston
Muzyczka N (1992) Use of adeno-associated virus as a general transduction vector for mammalian cells. Curr Top Microbiol Immunol 158:97–129
Choi VW, Asokan A, Haberman RA, Samulski RJ (2007) Production of recombinant adeno-associated viral vectors for in vitro and in vivo use. Curr Protoc Mol Biol. 16.25.11–16.25.24
Lynch CM (1999) Generation of recombinant adeno-associated viruses for delivery of genes into vascular cells. In: Baker S (ed) Vascular disease: molecular biology and gene therapy protocols. Humana Press, Totowa
Hanna J, Wernig M, Markoulaki S, Sun CW, Meissner A, Cassady JP, Beard C, Brambrink T, Wu LC, Townes TM et al (2007) Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science 318:1920–1923
Warren L, Manos PD, Ahfeldt T, Loh YH, Li H, Lau F, Ebina W, Mandal PK, Smith ZD, Meissner A et al (2011) Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7:618–630
Maeder ML, Thibodeau-Beganny S, Osiak A, Wright DA, Anthony RM, Eichtinger M, et al (2008) Rapid “open-source” engineering of customized zinc-finger nucleases for highly efficient gene modification. Mol Cell 31:294–301
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Su, CH., Erol, D. (2013). Current Ex-Vivo Gene Therapy Technologies and Future Developments. In: Tsang, S. (eds) Stem Cell Biology and Regenerative Medicine in Ophthalmology. Stem Cell Biology and Regenerative Medicine. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-5493-9_10
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5493-9_10
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-5492-2
Online ISBN: 978-1-4614-5493-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)