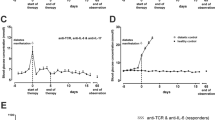
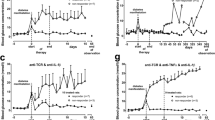
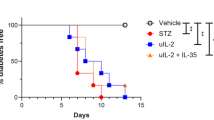
Abstract
There is convincing evidence that the destruction of pancreatic B-cells leading to insulin-dependent diabetes mellitus (IDDM) is immunologically mediated1–3 and occurs in all mammals.4 Investigations of animal strains predominantly used in diabetes research, the NOD mouse and the BB rat, demonstrated that the acute onset of hyperglycemia is the final result of a chronic preclinical phase (prediabetes). Already during prediabetes and definitely at the onset of the disease, the number of pancreatic B-cells is reduced.5–11 In addition to the diminished number of B-cells, the pancreatic islets of prediabetic or newly diagnosed diabetic animals or patients are characterized by an invasion of immunocytes, so-called insulitis.5,6,9–14 The immunocytochemistry of invading mononuclear cells (MNC) reveals the presence of all major subsets, such as T-lymphocytes (CD4 and CD8 T-cells), B-lymphocytes, macrophages, and NK-cells.8,10–15 Electron microscopic investigations of inflafhed pancreatic islets demonstrated a close contact between immune cells and pancreatic B-cells. As a result of B-cell destruction, B-cell granules have been observed in the extracellular space.15–17 Although the molecular mechanism of B-cell killing is still not established, cytotoxic reactivities of CD4 T-cells,18 CD8 T-cells,19 NK cells,20 and macrophages21 against pancreatic B-cells have been demonstrated. In addition, it has been shown that the secretory products of immunocytes, the cytokines, may directly disrupt B-cells,22 or may induce surface proteins as MHC-antigens or adhesion molecules, which allow or facilitate cell-mediated killing of B-cells.23
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Hahn HJ. Tierexperimenteller Diabetes. In: Diabetes mellitus. Bibergeil H, ed. Fischer Verlag, Jena, pp 108–17, 1989.
Leiter EH, Prochazka M, Coleman DL. Animal model of human disease. The non-obese diabetes (NOD) mouse. Am J Pathol 128:380–3, 1987.
Like AA, Rossini AA. Spontaneous autoimmune diabetes mellitus in the Bio-Breeding/Worcester rat. Surv Synth Pathol 3:131–8, 1984.
Buschard K. The thymus-dependent immune system in the pathogenesis of type 1 (insulin-dependent) diabetes mellitus. Animal model and human studies. Dan Med Bull 32:139–51, 1985.
Foulis AK, Farquharson MA. Aberrant expression of HLA-DR antigens by insulin-containing beta-cells in recent onset type I diabetes mellitus. Diabetes 35:1215–24, 1986.
Gepts W, De Mey J. Islet cell survival determined by morphology: An immuno-cytochemical study of the islets of Langerhans in juvenile diabetes mellitus. Diabetes 27(Suppl 1):251–61, 1978.
Gepts W. Pathologic anatomy of the pancreas in juvenile diabetes mellitus. Diabetes 14:619–33, 1965.
Bottazzo GF, Path MRC, Dean BM, et al. In situ characterization of autoimmune phenomena and expression of HLA molecules in the pancreas in diabetic insulitis. N Engl J Med 313:353–60, 1985.
Hanafusa T, Miyazaki A, Miyagawa J, et al. Examination of islets in the pancreas biopsy specimens from newly diagnosed type I (insulin-dependent) diabetic patients. Diabetologia 33:105–11, 1990.
Lucke S, Besch W, Kauert C, Hahn HJ. The endocrine pancreas of BB/OK-rats before and at diagnosis of hyperglycemia. Exp Clin Endocrinol 91:161–70, 1988.
Hanenberg H, Kolb-Bachofen V, Kantwerk-Funke G, Kolb H. Macrophage infiltration precedes and is a prerequisite for lymphocytic insulitis in pancreas of pre-diabetic BB rats. Diabetologia 32:126–34, 1989.
Dean BM, Walker R, Bone AJ, et al. Pre-diabetes in the spontaneously diabetic BB/E rat: Lymphocyte subpopulations in the pancreatic infiltrate and expression of rat MHC class II molecules in endocrine cells. Diabetologia 28:464–6, 1985.
Hahn HJ, Gerdes J, Lücke S, et al. Phenotypical characterization of the cells invading pancreatic islets of diabetic BB/OK rats: Effect of interleukin 2 receptor-targeted immunotherapy. Eur J Immunol 18:2037–42, 1988.
Lucke S, Diamantstein T, Hahn HJ. Different lymphocyte subset distribution within “insulitis” islets of normoglycaemic and prediabetic BB/OK rats of similar age. Exp Clin Endocrinol 95:57–63, 1990.
Diamantstein T, Hahn HJ. Interleukin-2 receptor-targeted immune therapy. In: Monoclonal antibodies and peptide therapy in autoimmune diseases. Bach JF, ed. Marcel Dekker, New York, pp 295–318, 1993.
Maruyama T, Ogawa M, Kobayashi F, et al. Electron microscopic studies on the interaction of pancreatic islet cells and splenic lymphocytes in non-obese diabetic (NOD) mice. Biomed Res 9:67–73, 1988.
Miyagawa J, Hanafusa T, Itoh N, et al. Ultrastructural pathology of insulitis in the pancreas biopsy specimens from newly diagnosed type I diabetic patients. Abstract 15th IDF Congress, 1994, Kobe, Japan, p 434.
Haskins K, McDuffie M. Acceleration of diabetes in young NOD mice with a CD4+ islet-specific T-cell clone. Science 249:1433–36, 1990.
Nagata M, Yoon JW. Studies on autoimmunity for T-cell mediated β-cell destruction. Distinct differences in β-cell destruction between CD4+ and CD8 + T-cell clones erived from lymphocytes infiltrating the islets of NOD mice. Diabetes 41:998–1008, 1992.
Hosszufalus N, Chan E, Teruya M, et al. Quantitative pheotypic and functional analysis of islet immune cells before and after diabetes onset in the BB rat. Diabetologia 36:1146–54, 1993.
Appels B, Burkhart V, Kantwerk-Funke G, et al. Spontaneous cytotoxicity of macrophages against pancreatic islet cells. J Immunol 142:3803–8, 1989.
Nerup J, Mandrup-Poulsen T, Molvig J. The HLA-IDDM association for etiology and pathogenesis of IDDM. Diabetes Metab Rev 3:779–802, 1987.
Kuttler B, Wanka H, Dunger A, Hahn HJ. Expression of MHC antigens on pancreatic islet cells. In: Frontiers of insulin secretion and pancreatic B-cell research. Flatt PR, Lenzen S, eds. Smith-Gordon, London, pp 509–15, 1994.
Skyler JS. Immune intervention studies in insulin-dependent diabetes mellitus. Diabetes Metab Rev 3:1017–35, 1987.
Vialettes B, Atlan-Gepner C, Hermitte L. Specific immuno-therapy for the prevention of diabetes: Theoretical and practical aspects. Diabetes Metab Rev 9:251–5, 1993.
Muir A, Schatz DA, Pozzilli P, MacLaren NK. Intervention therapies for insulin-dependent diabetes. Autoimmunity 16:301–10, 1993.
Smith KA. Interleukin-2: Inception, impact and implications. Science 240:1169–76, 1988.
Waldmann TA. Multichain interleukin-2 receptor: A target for immunotherapy in lymphoma. Immunol Rev 81:914–23, 1989.
Diamantstein T, Osawa H. The interleukin-2 receptor, its physiology and a new approach to a selective immuosuppressive therapy by anti-interleukin-2 receptor monoclonal antibodies. Immuol Rev 92:5–27, 1986.
Diamantstein T, Tilney NL, Strom TB, et al. Interleukin-2 receptor targeted immunosuppressive therapy with anti-interleukin-2 receptor monoclonal antibodies directed against the L-chain of the interleukin 2 receptor: Studies on selectivity and mode of action. In: Advances in immuopharmacology 4. Hadden JW, Spreafico F, Yamamura Y, et al., eds. Pergamon Press, UK, pp 95–104, 1988.
Hahn HJ, Kuttler B, Dunger A, et al. Prolongation of rat pancreatic islet allograft survival by teatment of recipient rats with monoclonal anti-interleukin-2 receptor antibody and cyclosporine. Diabetologia 30:44–6, 1987.
Kupiec-Weglinski JW, Hahn HJ, et al. Cyclosporin potentiates the immunosuppressive effects of anti-interleukin 2 receptor monoclonal antibody therapy. Transplant Proc 2:207–16, 1988.
Kupiec-Weglinski JW, Diamantstein T, Tilney NL. Interleukin 2 receptor-targeted therapy—rationale and applications in organ transplantations. Transplantation 46:785–92, 1988.
Kuttler B, Heym S, Volk HD, et al. No correlation between changes in lymphocyte subsets and pancreatic B-cell destruction. Horm Metab Res 22(Suppl 25): 182–7, 1990.
Wanka H, Kuttler B, Hahn HJ. Detection of β-cell autoreactive cells from BB rats after sensibilization of lymphocytes by syngeneic pancreatic islets. Diabetologia 35(Suppl 1):A213, 1992.
Wanka H, Kuttler B, Hahn HJ: Is the in vitro destruction of pancreatic islet cells MHC-dependent? Diabetologia 36(Suppl 1):A9, 1993.
Hahn HJ, Kuttler B, Volk HD, et al. Interleukin 2 receptor-targeted therapy in islet transplantation research. Horm Metab Res 25(Suppl 1): 177–80, 1989.
Tellides G, Dallman MJ, Morris PJ: Synergistic interaction of cyclosporine A with interleukin 2 receptor monoclonal antibody therapy. Transplant Proc 20(Suppl 2):202, 1988.
Granelli-Piperno A, Keane M, Steinman RM: Evidence that cyclosporine inhibits cell-mediated immunity primarily at the level of the T lymphocytes rather than the accessory cell. Transplantation 46:53S-60S, 1988.
Hahn HJ, Lucke S, Klöting I, et al. Curing BB rats of freshly manifested diabetes by short-term treatment with a combination of a monoclonal anti-interleukin 2 receptor antibody and a subtherapeutic dose of cyclosporin A. Eur J Immunol 17:1075–78, 1987.
Kelley VE, Gailton GN, Hattori M, et al. Anti-interleukin 2 receptor antibody suppresses murine diabetic insulitis and lupus nephritis. J Immunol 140:59–61, 1988.
Stark O, Klöting I, Reiher K, Kohnert KD: The major histocompatibility complex and insulin-dependent diabetes in BB rats. Acta Biol Med Germ 41:1129–33, 1982.
Klöting I, Stark O, Hahn HJ. Animal model of the insulin-dependent diabetes mellitus in BB rats: Their RT1U homogeneity and prolonged survival of allogeneic skin grafts. Folia Biol 30:24–32, 1984.
Klöting I, Stark O, Brdicka R. Incidence of insulin-dependent mellitus in BB rats: Their genetic heterogeneity and susceptibility to infection. Folia Biol 30:33–42, 1984.
Klöting I, Stark O. Occurrence of antibodies against gene products of the major histocompatibility complex in normal sera of diabetes-prone BB rats. Folia Biol 31:34–7, 1985.
Knospe S, Köhler E, Klöting I. Cell-mediated immune reactions against islets of Langerhans in diabetes-prone BB rats. Exp Clin Endocrinol 89:290–6, 1987.
Ziegler B, Klöting I, Besch W, Ziegler M, Hahn HJ. Cytotoxic activity of sera from diabetic BB rats against BB rat islet—a functional study. Diabetes Res 4:67–72, 1987.
Schröder D, Hehmke B, Klöting I, Besch W. Effect of sera from diabetes-prone BB/OK rats on neonatal rat pancreatic islets and islet cell suspensions. Exp Clin Endocrinol 93:187–92, 1989.
Hehmke B, Lücke S, Schröder D, Klöting I, Kohnert KD. Complement-dependent antibody-mediated cytotoxicity in the spontaneously diabetic BB/OK rat: Association with beta-cell volume density. Eur J Immunol 20:1091–96, 1990.
Klöting I, Reiher K. Einige Aspekte zur Haltung, Zucht und Reproduktion spontandiabetischer BB-Ratten. Z Versuchstierkd 27:5–12, 1985.
Klöting I, Vogt L. Breeding problems in diabetes-prone BB rats after “wet hysterectomy.” Z Versuchstierkd 31:19–22, 1988.
Klöting I, Vogt L, Stark O, Fischer U. Genetic heterogeneity in different BB rat subpopulations. Diabetes Res 6:145–9, 1987.
Klöting I, Vogt L. BB/O(ttawa)K(arlsburg) rats: Features of a subline of diabetes-prone BB rats. Diabetes Res Clin Exp 18:79–87, 1991.
Klöting I, Vogt L. On the influence of metabolic state of parents at conception and pregnancy on incidence and age at onset of diabetes in the progeny of BB/OK rats. Diabetes Res 19:37–40, 1992.
Vogt L, Klöting I. Model-based prediction of diabetes incidence in BB/OK rats. Diabetes Metab 19:183–7, 1993.
Vogt L, Klöting I. BB-RADABA: A computer program as a tool for breeding and management of laboratory animals. Comp Method Progr Biomed 38:73–4, 1992.
Sibley RK, Sutherland DER, Goetz F, Michael AF. Recurrent diabetes mellitus in the pancreas iso- and allograft. A light and electron microscopic and immunohistochemical analysis of four cases. Lab Invest 53:132–44, 1985.
Hahn HJ, Kauert C, Dunger A, et al. Interleukin 2 receptor targeted immunotherapy in the prevention of autoimmune beta cell destruction as observed in diabetic BB/OK rats grafted with BB/OK rat islets. Autoimmunity 5:139–42, 1989.
Gottlieb PA, Berrios JP, Mariani G, et al. Autoimmune destruction of islets transplanted into RT6-depleted resistant BB/Wor rats. Diabetes 39:643–5, 1990.
Mathieu C. Immune intervention in animal models of type I diabetes: Pathogenetical and therapeutical aspects. Thesis, Katholieke Universiteit Leuven, Faculteit Geneeskunde, Leuven, 1994.
Wang Y, Pontesilli O, Gill RG, et al. The role of CD4+ and CD8+ T cells in the destruction of islet grafts by spontaneously diabetic mice. Proc Natl Acad Sci USA 88:527–31, 1991.
Hahn HJ. Die isolierte Langerhanssche Insel, ein Modell zur Untersuchung der Insulinsekretion in vitro. Endokrinologie 71:308–24, 1978.
Kuttler B, Mathieu C, Waer M, et al. Lack of disease recurrence in diabetic BB/Pfd rats after syngeneic islet transplantation. Autoimmunity 15:107–12, 1993.
Mathieu C, Kuttler B, Waer M, et al. Spontaneous reestablishment of self-tolerance in BB/Pfd rats. Transplantation 58:349–54, 1994.
Woehrle M, Pullmann J, Stuenkel KG, et al. Effect of anti-interleukin-2 receptor antibody treatment on recurrence autoimmune insulitis in the BB rat. Transplant Proc 26:734–5, 1994.
Holowachuk EW, Greer MK, Martin DR. The complete sequence of the MHC class II chain RT.1D alpha u of the diabetic BB rat: mRNA levels of RT1.D alpha in lymphocytes. Nucleic Acids Res 15:10551–567, 1987.
Hahn HJ, Ziegler B, Lücke S. Streptozocin diabetic BB/OK rats accept permanently BB/OK-islet grafts without immunosuppression. Z Versuchstierkd 32:135–40, 1989.
Woehrle M, Pullmann J, Bretzel RG, Federlin K. Prevention of recurrent autoimmune diabetes in the BB rat by islet transplantation under the renal capsule. Transplantation 53:1099–102, 1992.
Kuttler B, Dunger A, Volk, et al. Prevention and suppression of autoimmune pancreatic beta-cell destruction in BB rats by syngeneic lymphocytes obtained from long-term normoglycaemic donors. Diabetologia 34:74–7, 1991.
Mac Kay P. Adoptive transfer of diabetes to and from old normoglycaemic BB rats. Diabetologia 38:145–52, 1995.
Hahn HJ, Kuttler B. Influence on the pancreatic β-cell of manipulations with therapeutic potential for prevention of insulin-dependent diabetes. In: Frontiers of insulin secretion and pancreatic B-cell research. Flatt PR, Lenzen S, eds. Smith-Gordon, London, pp 597–604, 1994.
Hutchings P, O’Reilly L, Parish NM, et al. The use of a non-depleting anti-CD4 monoclonal antibody to re-establish tolerance to ß-cells in NOD mice. Eur J Immunol 22:1913–18, 1992.
Charlton B, Mandel TE. Recurrence of insulitis in the NOD mouse after early prolonged anti-CD4 monoclonal antibody treatment. Autoimmunity 4:1–7, 1989.
Steinbriichel DA, Larsen S, Kristensen T, et al. Survival, function, morphology and serological aspects of rat renal allografts. Effect of short-term treatment with cyclosporin A, anti-CD4 and anti-interleukin-2 receptor monoclonal antibodies. AP-MIS 100:682–94, 1992.
Kuttler B, Kauert C, Diamantstein T, Hahn HJ. Long-term immunosuppression inhibits the development of tolerance in BB rats. Symposium Intern Diabetes Immunother Group (IDIG) Istanbul, 1993.
Vialettes B, Bardet S, Hirn D, et al. Pilot study of combination anti IL2R mc antibody-low dose of Cyclosporin A in recently diagnosed type 1 diabetes. Diabetologia 34(Suppl 2):A180, 1991.
Ueda H, Hancock WW, Cheung YC, et al. The mechanism of synergistic interaction between anti-interleukin 2 receptor monoclonal antibody and cyclosporine therapy in rat recipients of organ allografts. Transplantation 50:545–50, 1990.
Sido B, Morris PJ, Dallman M. Phenotypic and functional analysis of graft-infiltrating cells following therapy with anti-interleukin-2 receptor monoclonal antibody and cyclosporine A in the rat. Transplant Proc 24:2549–50, 1992.
Soulillou JP, Cantarovich D, Dantal J, et al. Anti-IL-2 receptor and anti-LFA-1 monoclonal antibodies in transplantation: Rationale and Nantes clinical experience. In: A critical analysis of monoclonal antibody therapy in transplantation. Burlingham WJ, ed. CRC Press, Boca Raton, FL, pp 47–66, 1992.
Wood MJA, Sloan DJ, Dallman MJ, Charlton HM. Specific tolerance to neural allografts induced with an antibody to the interleukin 2 receptor. J Exp Med 177:597–603, 1993.
Waldmann H, Cobbold S. Monoclonal antibodies for the induction of transplantation tolerance. Curr Opin Immunol 5:753–8, 1993.
Leong LYW, Qin S, Cobbold SP, Waldmann H. Classical transplantation tolerance in the adult: The interaction between myeloablation and immunosuppression. Eur J Immunol 22:2825–30, 1992.
Qin S, Cobbold SP, Pope H, et al. Infectious transplantation tolerance. Science 259:974–77, 1993.
Tomonari K. Tolerance in vivo and in vitro. Immunol Rev 116:139–57, 1990.
Alters SE, Grossman D, Fathmann CG. Anti-CD4-mediated transplantation tolerance: Mechanisms and memory. In: A critical analysis of monoclonal antibody therapy in transplantation. Burlingham WJ, ed. CRC Press, Boca Raton, FL, pp 79–99, 1992.
Hahn HJ, Lucke S, Kuttler B, et al. Investigation of diabetes-prone normoglycaemic BB rats. In: Lessons from animal diabetes. Shafrir E, ed. Smith-Gordon, London, 3:30–3, 1990.
Hahn HJ, Lücke S, Klöting I, Besch W. Prospective investigations of long-term normoglycaemic BB/OK rats: Serial determination of glucose tolerance, insulitis, B-cell volume density and pancreatic insulin content. Exp Clin Endocrinol 98:54–61, 1991.
Hahn HJ, Diamantstein T. Monoclonals and/or immunotoxims as new therapeutic agents in the treatment of autoimmune diseases. In: Diabetes mellitus. Wojcikowski C, ed. Verlag Normex, Gdansk, pp 1–10, 1992.
Hahn HJ, Kauert C, Kuttler B. Efficacy of FK506 in the prevention of autoaggression in syngeneicly grafted diabetic BB rats. Horm Metab Res 25:56(A24), 1993.
Chen Z, Cobbold S, Waldmann H, Metealf S. Stability of tolerance in mice generated by CD4 and CD8 monoclonal antibody treatment: Cell transfer experiments. Transplant Proc 25:790–1, 1993.
Kupiec-Weglinski JW, Diamanstein T, Tilney NL, Strom TB. Therapy with monoclonal antibody to interleukin 2 receptor spares suppressor T cells and prevents or reverses acute allograft rejection in rats. Proc Natl Acad Sci USA 83:2624–27, 1986.
Rashba Ej, Reich EP, Janeway CA, Sherwin RS. Type 1 diabetes mellitus: An imbalance between effector and regulatory T cells? Acta Diabetol 30:61–9, 1993.
Kuttler B, Kauert C, Wanka H, et al. Tolerance to MHC-identical allogeneic rat islets is mediated by CD4+ helper T-lymphocytes. Diabetologia 38(Suppl 1):A84, 1995.
Fowell D, Mason D. Evidence that the T cell repertoire of normal rats contains cells with the potential to cure diabetes. Characterization of the CD4+ T cell subset that inhibits this autoimune potential. J Exp Med 177:627–36, 1993.
Hahn HJ, Hellman B, Lernmark A, et al. The pancreatic B-cell recognition of insulin secretagogues. X. Influence of neuraminidase treatment on the release of insulin, and the islet content of insulin, sialic acid, and adenosine 3’5’-cyclic monophosphate. J Biol Chem 249:5275–84, 1974.
Ziegler B, Hahn HJ, Ziegler M. Insulin recovery in pancrease and host organs of islet grafts. Exp Clin Endocrinol 85:53–60, 1985.
Lucke S, Ziegler B, Diaz-Alonso JM, Hahn HJ. Eignung spezifischer Färbemethoden für die Bestimmung des ß-Zellvolumens im Rattenpankreas mit normalem und reduziertem Insulingehalt. Acta Histochem 45:107–16, 1985.
Cordeil UL, Falini B, Erber WN, et al. Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase anti-alkaline phosphatase (APAAP complexes). J Histochem 32:219–29, 1984.
Kuttler B, Lehmann M, Lacha J, et al. Anti-CD4 therapy induces a donor-specific, organ-unspecific tolerance in allogeneic kidney graft recipients. Transplant Proc 26:728–9, 1994.
Logothetopoulus J, Valiquette N, Madura E, Cvet D. Onset and progression of pancreatic insulitis in the overt, spontaneously diabetic, young adult BB rat studied by pancreatic biopsy. Diabetes 33:33–6, 1984.
Vogt L, Klöting I. Genetic analysis of frequencies of phenotypes in the spontaneously diabetic BB rat—a main animal model of autoimmune type-I-diabetes. Diabetes Res 22:105–13, 1993.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1996 Birkhäuser Boston
About this chapter
Cite this chapter
Kuttler, B., Lucke, S., Vogt, L., Klöting, I., Diamanstein, T., Hahn, HJ. (1996). Immunotherapy of insulin-dependent diabetes mellitus: Tolerance after short-term anti—;IL-2 receptor/cyclosporine therapy in BB/OK rats. In: Shafrir, E. (eds) Lessons from Animal Diabetes VI. Rev.Ser.Advs.Research Diab.Animals (Birkhäuser), vol 6. Birkhäuser Boston. https://doi.org/10.1007/978-1-4612-4112-6_4
Download citation
DOI: https://doi.org/10.1007/978-1-4612-4112-6_4
Publisher Name: Birkhäuser Boston
Print ISBN: 978-1-4612-8658-5
Online ISBN: 978-1-4612-4112-6
eBook Packages: Springer Book Archive