Abstract
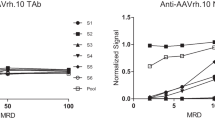
The field of cardiac gene therapy has seen the rising use of adeno-associated viral (AAV) vectors as a promising therapeutic option for cardiac diseases and heart failure. To achieve intended results of AAV delivery, a majority of clinical studies screen patients for existing neutralizing antibodies that could inhibit the effects of the administered AAV and confound treatment efficacy. The cell-based neutralizing antibody assay offers a method of quantifying and identifying a patient’s existing neutralizing antibodies against specific serotypes. Combined with the luciferase assay, the neutralizing antibody assay tests the ability of patient antibodies in the blood to prevent gene transduction of AAV-encoded luciferase gene at ranging serial dilutions. This chapter provides a protocol and experimental techniques to determine the presence of neutralizing antibodies against AAV in the blood.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP (2020) Heart disease and stroke statistics 2020 update. American Heart Association. Circulation 141(9):139–596
Chamberlain K, Riyad JM, Weber T (2017) Cardiac gene therapy with adeno-associated virus-based vectors. Curr Opin Cardiol 32(3):275–282
Rapti K, Hajjar RJ, Weber T (2012) Novel approaches to deliver molecular therapeutics in cardiac disease using adeno-associated virus vectors. Translational cardiology. In: Molecular and translational medicine. Humana Press, Totowa, pp 391–458. https://doi.org/10.1007/978-1-61779-891-7
Rapti K, Stillitano F, Karakikes I, Nonnenmacher M, Weber T, Hulot JS, Hajjar RJ (2015) Effectiveness of gene delivery systems for pluripotent and differentiated cells. Mol Ther Methods Clin Dev 2:14067. https://doi.org/10.1038/mtm.2014.67
Ronzitti G, Gross DA, Mingozzi F (2020) Human immune responses to Adeno-Associated Virus (AAV) vectors. Front Immunol 11:670. https://doi.org/10.3389/fimmu.2020.00670
Kuranda K, Jean-Alphonse P, Leborgne C, Hardet R, Collaud F, Marmier S, Costa Verdera H, Ronzitti G, Veron P, Mingozzi F (2018) Exposure to wild-type AAV drives distinct capsid immunity profiles in humans. J Clin Invest 128(12):5267–5279. https://doi.org/10.1172/JCI122372
Mingozzi F, High KA (2013) Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood 122(1):23–36. https://doi.org/10.1182/blood-2013-01-306647
Jungmann A, Müller O, Rapti K (2017) Cell-based measurement of neutralizing antibodies against Adeno-Associated Virus (AAV). Methods Mol Biol (Clifton, NJ) 1521:109–126
Acknowledgments
This work was supported by NIH R01 HL139963 (K.I). We acknowledge the Gene Therapy Resource Program (GTRP) of the National Heart, Lung, and Blood Institute and National Institutes of Health for providing the gene vectors used in this study.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ravichandran, A.J., Mazurek, R., Ishikawa, K. (2022). Cell-Based Determination of Neutralizing Antibodies Against Adeno-Associated Virus in Cardiac Gene Therapy. In: Ishikawa, K. (eds) Cardiac Gene Therapy. Methods in Molecular Biology, vol 2573. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2707-5_22
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2707-5_22
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2706-8
Online ISBN: 978-1-0716-2707-5
eBook Packages: Springer Protocols