Abstract
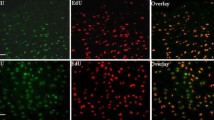
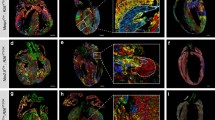
Neonatal mice display a remarkable ability to regenerate their heart following an injury during the first week of life. A key facet of successful cardiac regeneration is the proliferation of cardiomyocytes to replace the lost cells. Stimulating cardiomyocyte proliferation in the adult heart is a very promising approach to restore cardiac structure and function following injury. Here, we outline our approach to assess cardiomyocyte proliferation following a myocardial injury via the cell cycle markers phospho-histone H3 and Aurora B. We additionally discuss how we assess successful regeneration using wheat germ agglutinin to measure cardiomyocyte size, nuclear staining to quantify cardiomyocyte nucleation, and Trichrome staining to identify myocardial regeneration and scarring in the myocardium.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jopling C, Sleep E, Raya M, Marti M, Raya A, Izpisua Belmonte JC (2010) Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 464(7288):606–609. https://doi.org/10.1038/nature08899
Laube F, Heister M, Scholz C, Borchardt T, Braun T (2006) Re-programming of newt cardiomyocytes is induced by tissue regeneration. J Cell Sci 119(22):4719–4729. https://doi.org/10.1242/jcs.03252
Becker RO, Chapin S, Sherry R (1974) Regeneration of the ventricular myocardium in amphibians. Nature 248(5444):145–147. https://doi.org/10.1038/248145a0
Lutgens E, Daemen MJAP, de Muinck ED, Debets J, Leenders P, Smits JFM (1999) Chronic myocardial infarction in the mouse: cardiac structural and functional change1. Cardiovasc Res 41(3):586–593. https://doi.org/10.1016/s0008-6363(98)00216-8
Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, Sadek HA (2011) Transient regenerative potential of the neonatal mouse heart. Science 331(6020):1078–1080. https://doi.org/10.1126/science.1200708
Mahmoud AI, Porrello ER, Kimura W, Olson EN, Sadek HA (2014) Surgical models for cardiac regeneration in neonatal mice. Nat Protoc 9(2):305–311. https://doi.org/10.1038/nprot.2014.021
Li Y, He L, Huang X, Bhaloo SI, Zhao H, Zhang S, Pu W, Tian X, Li Y, Liu Q, Yu W, Zhang L, Liu X, Liu K, Tang J, Zhang H, Cai D, Ralf AH, Xu Q, Lui KO, Zhou B (2018) Genetic lineage tracing of nonmyocyte population by dual recombinases. Circulation 138(8):793–805. https://doi.org/10.1161/CIRCULATIONAHA.118.034250
Alkass K, Panula J, Westman M, Wu T-D, Guerquin-Kern J-L, Bergmann O (2015) No evidence for cardiomyocyte number expansion in preadolescent mice. Cell 163(4):1026–1036. https://doi.org/10.1016/j.cell.2015.10.035
Derks W, Bergmann O (2020) Polyploidy in cardiomyocytes. Circ Res 126(4):552–565. https://doi.org/10.1161/CIRCRESAHA.119.315408
Mohamed TMA, Ang Y-S, Radzinsky E, Zhou P, Huang Y, Elfenbein A, Foley A, Magnitsky S, Srivastava D (2018) Regulation of cell cycle to stimulate adult cardiomyocyte proliferation and cardiac regeneration. Cell 173(1):104–116. e112. https://doi.org/10.1016/j.cell.2018.02.014
Alvarez R Jr, Wang BJ, Quijada PJ, Avitabile D, Ho T, Shaitrit M, Chavarria M, Firouzi F, Ebeid D, Monsanto MM, Navarrete N, Moshref M, Siddiqi S, Broughton KM, Bailey BA, Gude NA, Sussman MA (2019) Cardiomyocyte cell cycle dynamics and proliferation revealed through cardiac-specific transgenesis of fluorescent ubiquitinated cell cycle indicator (FUCCI). J Mol Cell Cardiol 127:154–164. https://doi.org/10.1016/j.yjmcc.2018.12.007
Uribe V, Ramadass R, Dogra D, Rasouli SJ, Gunawan F, Nakajima H, Chiba A, Reischauer S, Mochizuki N, Stainier DYR (2018) In vivo analysis of cardiomyocyte proliferation during trabeculation. Development 145(14):dev164194. https://doi.org/10.1242/dev.164194
Choi W-Y, Gemberling M, Wang J, Holdway JE, Shen M-C, Karlstrom RO, Poss KD (2013) In vivo monitoring of cardiomyocyte proliferation to identify chemical modifiers of heart regeneration. Development 140(3):660–666. https://doi.org/10.1242/dev.088526
Patterson M, Barske L, Van Handel B, Rau CD, Gan P, Sharma A, Parikh S, Denholtz M, Huang Y, Yamaguchi Y, Shen H, Allayee H, Crump JG, Force TI, Lien CL, Makita T, Lusis AJ, Kumar SR, Sucov HM (2017) Frequency of mononuclear diploid cardiomyocytes underlies natural variation in heart regeneration. Nat Genet 49(9):1346–1353. https://doi.org/10.1038/ng.3929
Gonzalez-Rosa JM, Sharpe M, Field D, Soonpaa MH, Field LJ, Burns CE, Burns CG (2018) Myocardial polyploidization creates a barrier to heart regeneration in Zebrafish. Dev Cell 44(4):433–446. e437. https://doi.org/10.1016/j.devcel.2018.01.021
Han L, Choudhury S, Mich-Basso JD, Ammanamanchi N, Ganapathy B, Suresh S, Khaladkar M, Singh J, Maehr R, Zuppo DA, Kim J, Eberwine JH, Wyman SK, Wu YL, Kuhn B (2020) Lamin B2 levels regulate polyploidization of cardiomyocyte nuclei and myocardial regeneration. Dev Cell 53(1):42–59.e11. https://doi.org/10.1016/j.devcel.2020.01.030
Kolk MV, Meyberg D, Deuse T, Tang-Quan KR, Robbins RC, Reichenspurner H, Schrepfer S (2009) LAD-ligation: a murine model of myocardial infarction. J Vis Exp (32). https://doi.org/10.3791/1438
Reichert K, Colantuono B, McCormack I, Rodrigues F, Pavlov V, Abid MR (2017) Murine Left Anterior Descending (LAD) coronary artery ligation: an improved and simplified model for myocardial infarction. J Vis Exp 122:55353. https://doi.org/10.3791/55353
Lugrin J, Parapanov R, Krueger T, Liaudet L (2019) Murine myocardial infarction model using permanent ligation of left anterior descending coronary artery. J Vis Exp (150). https://doi.org/10.3791/59591
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Brandt, E.B., Mahmoud, A.I. (2022). Quantifying Cardiomyocyte Proliferation and Nucleation to Assess Mammalian Cardiac Regeneration. In: Coulombe, K.L., Black III, L.D. (eds) Cardiac Tissue Engineering. Methods in Molecular Biology, vol 2485. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2261-2_16
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2261-2_16
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2260-5
Online ISBN: 978-1-0716-2261-2
eBook Packages: Springer Protocols