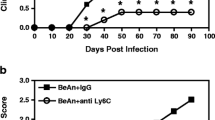
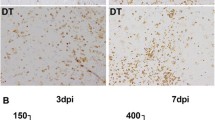
Abstract
Pathogenesis induced by mouse hepatitis virus (MHV) infection of rodents is characterized by acute viral encephalomyelitis and demyelination which progresses to a persistent CNS infection associated with ongoing myelin loss, pathologically similar to multiple sclerosis (MS). Although humoral immunity appears redundant for the control of acute virus replication, it is vital in maintaining virus at levels detectable only by RNA analysis. T cell mediated control of acute infection cannot be sustained in antibody (Ab) deficient mice, resulting in virus reactivation. The protective role of Ab during persistence is strongly supported by detection of Ab in the cerebrospinal fluid of MHV infected rodents and maintenance of virus specific Ab secreting cells (ASC) in the CNS long after virus clearance. Ab mediated neutralization constitutes the major mechanism of protection, although fusion inhibition also plays a minor role. Delayed accumulation of ASC, concomitant with a decline in T cell function, assures control of residual virus while minimizing T cell mediated pathology. Although there is little evidence for a detrimental role of Ab in demyelination, an association between Ab mediated protection and remyelination is unclear.
Chapter PDF
Similar content being viewed by others
References
Correale J., de Los Milagros Bassani Molinas M. Oligoclonal bands and antibody responses in Mulitple sclerosis. J Neurol. 2002. 249: 375–89.
Connor O., Kevin C, Bar-Or A., Hafler D. A. The Neuroimmunology of Multiple Sclerosis: Possible roles of T and B lymphocytes in Immunopathogenesis. J Clin Immunol. 2001. 21: 81–92.
Kabat E. A., Glusman M., Knaub V. Quantitative estimation of the albumin and gamma globulin in normal and pathologic cerebrospinal fluid by immunochemical methods. Am J Med. 1948. 4: 653–62.
Bornstein M. B., Appel S. H. The application of tissue culture to the study of experimental allergic encephalomyelitis. I. Patterns of demyelination. J Neuropathol Exp Neurol. 1961. 20: 141–7.
Esiri M. M., Immunoglobulin-containing cells in multiple sclerosis plaques. Lancet. 1977. ii: 478–80.
Genain C. P., Cannella B., Hauser S. L., Raine C. S. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat. Med. 1999. 5: 170–5.
Cross A. H., Trotter J. L., Lyons J-A. B cells and antibodies in CNS demyelinating disease. J Neurolmm. 2001. 112: 1–14.
Colombo M., Dono M, Gazzola P., Roncella S., Valetto A., Chiorazi N., Mancardi G. L., Ferrarini M. Accumulation of clonally related B lymphocytes in the cerebrospinal fluid of multiple sclerosis patients. 2001. J Immunol. 164: 2782–89.
Gilden D. H., Devlin M. E., Burgoon M. P., Owens G. P. The search for virus in multiple sclerosis brain. Mult Scler. 1996. 2: 179–83.
Archelos J. J., Storch M. K., Hartung H-P. The role of B cells and autoantibodies in Multiple sclerosis. Annals Neurol 2000. 47: 694–706.
Lumsden C. E. The immunopathogenesis of the multiple sclerosis plaque. Brain Res. 1971. 28: 365–90.
Stohlman, S. A., Bergmann C. C, Perlman S. “Mouse hepatitis virus.” In Persistent Viral Infections R. Ahmed, I. Chen. ed. New York, NY: Wiley Publishers. 1999.
Marten N. W., Stohlman S. A., Bergmann C. C. MHV infection of the CNS: Mechanisms of immune-mediated control. Viral Immunol. 2001. 14: 1–18.
Lin M. T., Hinton D. R., Marten N. W., Bergmann C. C, Stohlman S. A. Antibody prevents virus reactivation within the central nervous system. J Immunol. 1999. 162: 7358–68.
Ramakrishna, C, Stohlman S. A., tkinson R., Shlomchik M. J., Bergmann C. C. Mechanisms of central nervous system viral persistence: the critical role of antibody and B cells. J Immunol. 2002. 168: 1204–11.
Matthews A. E., Weiss S. R., Shlomchik M. J., Hannum L. G., Gombold J. L., Paterson Y. Antibody is required for clearance of infectious murine hepatitis virus A59 from the central nervous system, but not the liver. J Immunol. 2001. 167: 5254–63.
Tschen, S., Bergmann C. C., Ramakrishna C., Morales S., Atkinson R., Stohlman S. A. 2002. Recruitment kinetics and composition of antibody secreting cells within the central nervous system following viral encephalomyelitis. J. Immunol. 168: 2922–29.
Fleming J. O., Shubin R. A., Sussman M. A., Casteel N., Stohlman S. A. Monoclonal antibodies to the matrix (El) glycoprotein of mouse hepatitis virus protect mice from encephalitis. Virology. 1989. 168: 162–7.
Buchmeier M. J., Lewicki H. A., Talbot P. J., Knobler R. L. Murine hepatitis virus-4 (strain JHM)-induced neurologic disease is modulated in vivo by monoclonal antibody. Virology. 1984. 132: 261–70.
Nakanaga K., Yamanouchi K., Fujiwara K. Protective effect of monoclonal antibodies on lethal mouse hepatitis virus infection in mice. J Virol. 1986. 59: 168–71.
Perlman S., Schelper R., Bolger E., Ries D. Late onset, symptomatic, demyelinating encephalomyelitis in mice infected with MHV-JHM in the presence of maternal antibody. Microb Pathog. 1987. 2: 185–94.
Kolb A. J., Pewe L., Webster J., Perlman S., Whitelaw C. B., Siddel S. G. Virus-neutralizing monoclonal antibody expressed in milk of transgenic mice provides full protection against virus induced encephalitis. J. Virol. 2001. 75: 2803–9.
Morales S., Parra B., Ramakrishna C., Blau D. M., Stohlman S. A. B cell mediated lysis of cells infected with the neurotropic JHM strain of mouse hepatitis virus. Virol. 2001. 286: 160–7.
Slifka M. K., Ahmed R. Long lived plasma cells: a mechanism for maintaining persistent antibody production. Curr Opin Immunol. 1998. 10: 252–8.
Knopf P.M., Harling-Berg C. J., Cserr H. F., Basu B., Sirulnick E. J., Nolan S. C, Park J. T., Keir G., Thompson E. T., Hickey W. F. Antigen-Dependent intrathceal antibody synthesis in the normal rat brain: Tissue Entry and local retention of Antigen specific B cells. J Immunol. 1998. 161: 692–701.
Griffin D., Levine B., Tyor W., Ubol S., Despres P. The role of antibody in recovery from alphavirus encephalitis. Immunol Rev. 1997. 159: 155–61.
Schwender S., Imrich H., Dorries R. The pathogenic role of virus-specific antibody secreting cells in the central nervous system of rats with different susceptibility to coronavirus-induced demyelinating encephalitis. Immunol. 1991. 74: 533–8.
Bergmann C. C., Ramakrishna C., Kornacki M., Stohlman S. A. Impaired T cell immunity in B cell-deficient mice following viral central nervous system infection. J. Immunol. 2001. 167: 1575–83.
Smith-Norowitz T. A., Sobel R. A., Mokhatarian F. B cells and antibodies in the pathogenesis of myelin injury in Semliki Forest Virus encephalomyelitis. Cell Immunol. 2000. 200: 27–35.
Lin M. T., Stohlman S. A., Hinton D. R. Mouse hepatitis virus is cleared from the central nervous systems of mice lacking perforin-mediated cytolysis. J Virol. 1997. 71: 383–91.
Ramakrishna C, Bergmann C. C., Atkinson R., Stohlman S. A. Control of central nervous system persistence by neutralizing antibody. J. Virol. Submitted.
Furrer, E., T. Bilzer, L. Stitz, and O. Planz. Neutralizing antibodies in persistent Borna disease virus infection: prophylactic effect of gp94-specific monoclonal antibodies in preventing encephalitis. J Virol. 2001. 75: 943–51.
Lamarre A., Talbot P. J. Protection from lethal Coronavirus infection by immunoglobulin fragments. J Immunol. 1995. 154: 3975–84.
Houtman J. J., Fleming J. O. Dissociation of demyelination and viral clearance in congenitally immunodeficient mice infected with murine coronavirus JHM. J Neurovirol. 1996. 2: 101–10.
Zimprich F., Winter J., Wege H., Lassmann H. Coronavirus induced primary demyelination: indications for the involvement of a humoral immune response. Neuropath Applied Neurobiol. 1991. 17: 469–84.
Drescher K. M., Pease L. R., Rodriguez M. Antiviral immune responses modulate the nature of central nervous system (CNS) disease in a murine model of multiple sclerosis. Immunol Rev. 1997. 159: 177–93.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2005 Springer Science+Business Media, Inc.
About this chapter
Cite this chapter
Ramakrishna, C., Tschen, S., Bergmann, C.C., Stohlman, S.A. (2005). The Role of Humoral Immunity in Mouse Hepatitis Virus Induced Demyelination. In: Lavi, E., Constantinescu, C.S. (eds) Experimental Models of Multiple Sclerosis. Springer, Boston, MA. https://doi.org/10.1007/0-387-25518-4_41
Download citation
DOI: https://doi.org/10.1007/0-387-25518-4_41
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-25517-0
Online ISBN: 978-0-387-25518-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)