Abstract
Background
Carcinoma-associated fibroblasts (CAF) are considered to contribute to tumor growth, invasion and metastasis. However, the cell type of origin remains unknown. Since human adipose tissue derived stem cells (hASCs) are locally adjacent to breast cancer cells and might directly interact with tumor cells, we investigated whether CAFs may originate from hASCs.
Methods
hASCs cultured under different conditions were quantified for the expression of alpha smooth muscle actin. ELISA was performed using the human TGFβ1, SDF-1α and CCL5 Quantikine Kit. The invasion potential of MDAMB231 cancer cells was evaluated using a Boyden chamber with filter inserts coated with Matrigel in 24-well dishes.
Results
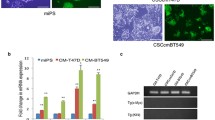
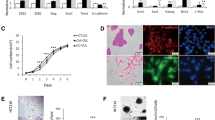
We demonstrated that a significant percentage of hASCs differentiated into a CAF-like myofibroblastic phenotype (e.g. expression of alpha smooth muscle actin and tenascin-C) when exposed to conditioned medium from the human breast cancer lines MDAMB231 and MCF7. The conditioned medium from MDAMB231 and MCF7 contains significant amounts of transforming growth factor-beta 1 (TGFβ1) and the differentiation of hASCs towards CAFs is dependent on TGFβ1 signaling via Smad3 in hASCs. The induction of CAFs can be abolished using a neutralizing antibody to TGFβ1 as well as by pretreatment of the hASCs with SB431542, a TGFβ1 receptor kinase inhibitor. Additionally, we found that these hASC-derived CAF-like cells exhibit functional properties of CAFs, including the ability to promote tumor cell invasion in an in vitro invasion assay, as well as increased expression of stromal-cell derived factor 1 (SDF-1) and CCL5.
Conclusion
Our data suggest that hASCs are a source of CAFs which play an important role in the tumor invasion.








Similar content being viewed by others
References
W.O. De, P. Demetter, M. Mareel, M. Bracke, Stromal myofibroblasts are drivers of invasive cancer growth. Int. J. Cancer 123, 2229–2238 (2008)
L.A. Kunz-Schughart, R. Knuechel, Tumor-associated fibroblasts (part I): Active stromal participants in tumor development and progression? Histol. Histopathol. 17, 599–621 (2002)
A.P. Sappino, O. Skalli, B. Jackson, W. Schurch, G. Gabbiani, Smooth-muscle differentiation in stromal cells of malignant and non-malignant breast tissues. Int. J. Cancer 41, 707–712 (1988)
A. Orimo, P.B. Gupta, D.C. Sgroi, F. Renzana-Seisdedos, T. Delaunay, R. Naeem, V.J. Carey, A.L. Richardson, R.A. Weinberg, Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005)
A. Muller, B. Homey, H. Soto, N. Ge, D. Catron, M.E. Buchanan, T. McClanahan, E. Murphy, W. Yuan, S.N. Wagner, J.L. Barrera, A. Mohar, E. Verastegui, A. Zlotnik, Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001)
W.O. De, Q.D. Nguyen, H.L. Van, M. Bracke, E. Bruyneel, C. Gespach, M. Mareel, Tenascin-C and SF/HGF produced by myofibroblasts in vitro provide convergent pro-invasive signals to human colon cancer cells through RhoA and Rac. FASEB J. 18, 1016–1018 (2004)
T. Tsujino, I. Seshimo, H. Yamamoto, C.Y. Ngan, K. Ezumi, I. Takemasa, M. Ikeda, M. Sekimoto, N. Matsuura, M. Monden, Stromal myofibroblasts predict disease recurrence for colorectal cancer. Clin. Cancer Res. 13, 2082–2090 (2007)
C. Yazhou, S. Wenlv, Z. Weidong, W. Licun, Clinicopathological significance of stromal myofibroblasts in invasive ductal carcinoma of the breast. Tumour. Biol. 25, 290–295 (2004)
I. Haviv, K. Polyak, W. Qiu, M. Hu, I. Campbell, Origin of carcinoma associated fibroblasts. Cell Cycle 8, 589–595 (2009)
L. Ronnov-Jessen, O.W. Petersen, Induction of alpha-smooth muscle actin by transforming growth factor-beta 1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasia. Lab Invest 68, 696–707 (1993)
N.C. Direkze, K. Hodivala-Dilke, R. Jeffery, T. Hunt, R. Poulsom, D. Oukrif, M.R. Alison, N.A. Wright, Bone marrow contribution to tumor-associated myofibroblasts and fibroblasts. Cancer Res. 64, 8492–8495 (2004)
G. Ishii, T. Sangai, T. Oda, Y. Aoyagi, T. Hasebe, N. Kanomata, Y. Endoh, C. Okumura, Y. Okuhara, J. Magae, M. Emura, T. Ochiya, A. Ochiai, Bone-marrow-derived myofibroblasts contribute to the cancer-induced stromal reaction. Biochem. Biophys. Res. Commun. 309, 232–240 (2003)
P.J. Mishra, P.J. Mishra, R. Humeniuk, D.J. Medina, G. Alexe, J.P. Mesirov, S. Ganesan, J.W. Glod, D. Banerjee, Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells. Cancer Res. 68, 4331–4339 (2008)
J.M. Gimble, A.J. Katz, B.A. Bunnell, Adipose-derived stem cells for regenerative medicine. Circ. Res. 100, 1249–1260 (2007)
P.A. Zuk, M. Zhu, H. Mizuno, J. Huang, J.W. Futrell, A.J. Katz, P. Benhaim, H.P. Lorenz, M.H. Hedrick, Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 7, 211–228 (2001)
S.Pinilla, E. Alt, F.J. Bdul Khalek, C. Jotzu, F. Muehlberg, C. Beckmann, Y.H. Song, Tissue resident stem cells produce CCL5 under the influence of cancer cells and thereby promote breast cancer cell invasion. Cancer Lett. (2009)
X. Bai, J. Ma, Z. Pan, Y.H. Song, S. Freyberg, Y. Yan, D. Vykoukal, E. Alt, Electrophysiological properties of human adipose tissue-derived stem cells. Am. J. Physiol Cell Physiol 293, C1539–C1550 (2007)
X. Bai, S. Sadat, S. Gehmert, E. Alt, Y.H. Song, VEGF receptor Flk-1 plays an important role in c-kit expression in adipose tissue derived stem cells. FEBS Lett. 581, 4681–4684 (2007)
B. Hu, Z. Wu, S.H. Phan, Smad3 mediates transforming growth factor-beta-induced alpha-smooth muscle actin expression. Am. J. Respir. Cell Mol. Biol. 29, 397–404 (2003)
J. Massague, TGFbeta in cancer. Cell 134, 215–230 (2008)
E.L. Spaeth, J.L. Dembinski, A.K. Sasser, K. Watson, A. Klopp, B. Hall, M. Andreeff, F. Marini, Mesenchymal stem cell transition to tumor-associated fibroblasts contributes to fibrovascular network expansion and tumor progression. PLoS. One. 4, e4992 (2009)
J.A. Tuxhorn, G.E. Ayala, D.R. Rowley, Reactive stroma in prostate cancer progression. J. Urol. 166, 2472–2483 (2001)
B.A. Teicher, Malignant cells, directors of the malignant process: role of transforming growth factor-beta. Cancer Metastasis Rev. 20, 133–143 (2001)
M.M. Webber, N. Trakul, P.S. Thraves, D. Bello-DeOcampo, W.W. Chu, P.D. Storto, T.K. Huard, J.S. Rhim, D.E. Williams, A human prostatic stromal myofibroblast cell line WPMY-1: a model for stromal-epithelial interactions in prostatic neoplasia. Carcinogenesis 20, 1185–1192 (1999)
D. Wang, J.S. Park, J.S. Chu, A. Krakowski, K. Luo, D.J. Chen, S. Li, Proteomic profiling of bone marrow mesenchymal stem cells upon transforming growth factor beta1 stimulation. J. Biol. Chem. 279, 43725–43734 (2004)
J.A. Burger, T.J. Kipps, CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood 107, 1761–1767 (2006)
F.L. Muehlberg, Y.H. Song, A. Krohn, S.P. Pinilla, L.H. Droll, X. Leng, M. Seidensticker, J. Ricke, A.M. Altman, E. Devarajan, W. Liu, R.B. Arlinghaus, E.U. Alt, Tissue-resident stem cells promote breast cancer growth and metastasis. Carcinogenesis 30, 589–597 (2009)
E.S. Jeon, H.J. Moon, M.J. Lee, H.Y. Song, Y.M. Kim, M. Cho, D.S. Suh, M.S. Yoon, C.L. Chang, J.S. Jung, J.H. Kim, Cancer-derived lysophosphatidic acid stimulates differentiation of human mesenchymal stem cells to myofibroblast-like cells. Stem Cells 26, 789–797 (2008)
M. Allinen, R. Beroukhim, L. Cai, C. Brennan, J. Lahti-Domenici, H. Huang, D. Porter, M. Hu, L. Chin, A. Richardson, S. Schnitt, W.R. Sellers, K. Polyak, Molecular characterization of the tumor microenvironment in breast cancer. Cancer Cell 6, 17–32 (2004)
A. Orimo, R.A. Weinberg, Stromal fibroblasts in cancer: a novel tumor-promoting cell type. Cell Cycle 5, 1597–1601 (2006)
Acknowledgements
This research was supported in part by the Department of Defense Breast Cancer Research Program W81XWH-08-1-0523 01 (to YHS) and by the Alliance of Cardiovascular Researchers (to EA).We thank Feras J Abdul Khalek and Christoph Beckmann for their technical assistance. The authors are also grateful to Dr. Sendurai Mani for helpful comments on this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Constantin Jotzu and Eckhard Alt contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
MCF7 conditioned medium stimulates the secretion of SDF-1α from hASCs. Protein levels of SDF-1α secreted from hASCs exposed to MCF7 conditioned medium over time (12–72 h) were measured by ELISA. The experiment was repeated three times. *P < 0.002 and **P < 0.0003 versus 12 h MCF7 CM. SDF-1α: stromal cell-derived factor 1 alpha; CM: conditioned medium. (PPT 105 kb)
Rights and permissions
About this article
Cite this article
Jotzu, C., Alt, E., Welte, G. et al. Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cellular Onc. 34, 55–67 (2011). https://doi.org/10.1007/s13402-011-0012-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-011-0012-1