Abstract
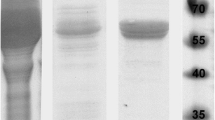
Maximum catalase production by mesophilic bacterium Serratia marcescens SYBC-01 was obtained by an optimization of culture medium and conditions. A novel cold-adapted catalase from the strain was purified and characterized. The Cat-2 without peroxidase activity was a homodimer with a molecular mass of 154 kDa, consisting of two identical subunits of about 70 kDa. Its apparent Km and Vmax value were 29.7 mM and 80,925 U/mg of protein, respectively. The Cat-2 exhibited maximal activity at pH 7.0, being relatively stable in alkaline conditions. The enzyme was most active at approximately 20°C and had 73.8% activity at 0°C. After incubation at 60°C for 60 min, the enzyme still maintained 75% of its initial activity. The Cat-2 displayed relatively higher thermostability compared to that of other cold-adapted and some mesophilic catalases.



Similar content being viewed by others
References
Adamo SA (2004) Estimating disease resistance in insects: phenoloxidase and lysozyme-like activity and disease resistance in the cricket Gryllus texensis. J Insect Physiol 50:209–216
Beers RF Jr, Sizer IW (1952) A spectrophotometric method for measuring the breakdown of hydrogen peroxide by Catalase. J Biol Chem 195:133–140
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brown-Peterson NJ, Salin ML (1993) Purification of a catalase-peroxidase from halobacterium halobium: characterization of some unique properties of the halophilic enzyme. J Bacteriol 175:4197–4202
Brown-Peterson NJ, Salin ML (1995) Purification and characterization of a mesohalic catalase from the halophilic bacterium Halobacterium halobium. J Bacteriol 177:378–384
Caridis K-A, Christakopoulos P, Macris BJ (1991) Simultaneous production of glucose oxidase and catalase by Alternaria alternata. Appl Microbiol Biotechnol 34:794–797
Chelikani P, Fita I, Loewen PC (2004) Diversity of structures and properties among catalases. Cell Mol Life Sci 61:192–208
Claiborne A, Fridovich I (1979) Purification of the o-dianisidine peroxidase from Escherichia Coli B. physicochemical characterization and analysis of its dual catalatic and peroxidatic activities. J Biol Chem 254:4245–4252
Duzhak AB, Panfilova ZI, Vasyunina EA (2002) Synthesis of extracellular chitinase by wild-type B-10 and mutant M-1 strains of Serratia marcescens. Appl Biochem Microbiol 38:214–221
Fu JH, Huang HQ, Meng K, Yuan TZ, Yao B, Shi YH, Ouyang PK (2008) A novel cold-adapted phospholipase a(1) from Serratia Sp Xjf1: gene cloning, expression and characterization. Enzyme MicrobTechnol 42:187–194
Georlette D, Bentahir M, Claverie P, Collins T, D 'Amico S, Delille D, Feller G, Gratia E, Hoyoux A, Lonhienne T (2001) Cold-adapted enzymes. In: De Cuyper M, Bulte JWM (eds) Physics and chemistry basis of biotechnology. Kluwer, London, pp 177–196
Goldberg I, Hochman A (1989) Three different types of catalases in Klebsiella pneumoniae. Arch Biochem Biophys 268:124–128
Kagawa M, Murakoshi N, Nishikawa Y, Matsumoto G, Kurata Y, Mizobata T, Kawata Y, Nagai J (1999) Purification and cloning of a thermostable manganese catalase from a thermophilic bacterium. Arch Biochem Biophys 362:346–355
Khardenavis AA, Kapley A, Purohit HJ (2009) Processing of poultry feathers by alkaline keratin hydrolyzing enzyme from Serratia sp. Hpc 1383. Waste Manag 29:1409–1415
Kim H, Lee JS, Hah YC, Roe JH (1994) Characterization of the major catalase from Streptomyces coelicolor Atcc 10147. Microbiology 140:3391–3397
Kobayashi I, Tamura T, Sghaier H, Narumi I, Yamaguchi S, Umeda K, Inagaki K (2006) Characterization of monofunctional catalase KatA from radioresistant bacterium Deinococcus Radiodurans. J Biosci Bioeng 101:315–321
Kurakov AV, Kupletskaya MB, Skrynnikova EV, Somova NG (2001) Search for micromycetes producing extracellular catalase and study of conditions of catalase synthesis. Appl Biochem Microbiol 37:59–64
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666
Loewen PC, Switala J (1987) Multiple catalases in Bacillus subtilis. J Bacteriol 169:3601–360
Lorentzen MS, Moe EH, Jouve M, Willassen NP (2006) Cold dapted features of Vibrio salmonicida catalase: characterisation and comparison to the mesophilic counterpart from Proteus mirabilis. Extremophiles 10:427–440
Margesin R, Schinner F (1999) Cold-adapted organisms-ecology, physiology, enzymology and molecular biology. Springer, Berlin, pp 1–416
Monti D, Baldaro E, Riva S (2003) Separation and characterization of two catalase activities isolated from the yeast Trigonopsis variabilis. Enzyme Microb Technol 32:596–605
Pair SD, Bruton BD, Mitchell F, Fletcher J, Wayadande JA, Melcher U (2004) Overwintering squash bugs harbor and transmit the causal agent of cucurbit yellow vine disease. J Econ Entomol 97:74–78
Patterson KL, Porter JW, Ritchie KB, Polson SW, Mueller E, Peters EC, Santavy DL, Smith GW (2002) The etiology of white pox, a lethal disease of the caribbean elkhorn coral, Acropora palmata. Proc Natl Acad Sci USA 99:8725–8730
Petruccioli M, Fenice M, Piccioni P, Federici F (1995) Effect of stirrer speed and buffering agents on the production of glucose oxidase and catalase by Penicillium variable (P16) in benchtopbioreactor. Enzyme Microb Technol 17:336–339
Qian SL, Cai YJ, Liao XR, Shen W, Wu K, Zhang F, Zhang DB (2008) Screening, identification and enzymatic properties of the thermal stability catalase-producing strain. Acta Agric Boreali-Occident Sin 17:238–242
Richards MJ, Edwards JR, Culver DH, Gaynes RP (2000) Nosocomial infections in combined medical-surgical intensive care units in the United States. Infect Control Hosp Epidemiol 21:510–515
Rochat T, Miyoshi A, Gratadoux JJ, Duwat P, Sourice S, Zevedo VA, Langella P (2005) High-level resistance to oxidative stress in Lactococcus lactis conferred by Bacillus subtilis Catalase Kate. Microbiology 151:3011–3018
Romero FJ, Garcia LA, Salas JA, Diaz M, Quiros LM (2001) Production, purification and partial characterization of two extracellular proteases from Serratia marcescens grown in whey. Proc Biochem 36:507–515
Shin DH, Choi YS, Cho YH (2008) Unusual properties of catalase a (Kata) of Pseudomonas Aeruginosa Pa14 are associated with its biofilm peroxide resistance. J Bacteriol 190:2663–2670
Switala J, Loewen PC (2002) Diversity of properties among catalases. Arch Biochem Biophys 401:145–154
Terzenbach DP, Blaut M (1998) Purification and characterization of a catalase from the nonsulfur phototrophic bacterium Rhodobacter Sphaeroides Ath 2.4.1 and its role in the oxidative stress response. Arch Microbiol 169:503–508
Venkateshwaran G, Somashekar D, Prakash MH, Basappa SC, Richard J (1999) Production and utilisation of catalase using Saccharomyces cerevisiae. Proc Biochem 34:187–191
Wang W, Sun M, Liu W, Zhang B (2008) Purification and characterization of a psychrophilic catalase from Antarctic Bacillus. Can J Microbiol 54:823–828
Woodbury W, Spencer AK, Stahman MA (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44:301–305
Yumoto I, Ichihashi D, Iwata H, Istokovics A, Ichise N, Matsuyama H, Okuyama H, Kawasaki K (2000) Purification and characterization of a catalase from the facultatively psychrophilic bacterium Vibrio rumoiensis S-1(T) exhibiting high catalase activity. J Bacteriol 182:1903–1909
Zámocký M, Koller F (1999) Understanding the structure and function of catalases: clues from molecular evolution and in vitro mutagenesis. Prog Biophys Mol Biol 72:19–66
Zámocký M, Godocikova J, Gasperik J, Koller F, Polek B (2004) Expression, purification, and sequence analysis of catalase-1 from the soil bacterium Comamonas terrigena N3H. Protein Expr Purif 36:115–123
Zhang XQ, Xue YF, Zhao AM, Du GC, Xu ZH, Chen J, Ma YH (2005) Purification and characterization of a monofunctional catalase from an alkaliphilic Bacillus sp. F26. Sheng Wu Gong Cheng Xue Bao 21:71–77
Acknowledgments
This work was financially supported by the National High Technology and Development Program of China (863 Program, grant No., 2010AA101501).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zeng, HW., Cai, YJ., Liao, XR. et al. Optimization of catalase production and purification and characterization of a novel cold-adapted Cat-2 from mesophilic bacterium Serratia marcescens SYBC-01. Ann Microbiol 60, 701–708 (2010). https://doi.org/10.1007/s13213-010-0116-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-010-0116-2