Abstract
Background
The purpose of this study was to determine the incidence of amenorrhea among breast cancer patients aged 40 years and younger following adjuvant chemotherapy.
Methods
A follow-up questionnaire survey was conducted with premenopausal women with breast cancer who were treated with adjuvant anthracycline and taxane-based chemotherapy.
Results
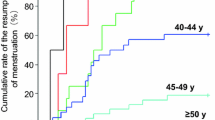
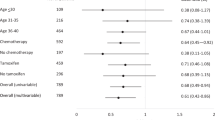
In total, 66 women were retrospectively reviewed. Forty-nine patients were treated with a regimen containing anthracycline followed by taxane and 17 patients with anthracycline alone. Fifty-eight patients (87.9%) experienced amenorrhea during chemotherapy; 14 patients (21.2%) had persistent amenorrhea after chemotherapy. The incidence of amenorrhea during chemotherapy and persistent amenorrhea was higher in patients older than 36 than in younger patients (97.9 vs. 63.2%, 27.7 vs. 5.3%). Additional taxane resulted in a higher incidence of amenorrhea compared with anthracycline-containing regimen alone (93.9 vs. 70.6%). Multivariate analysis showed that age (≥36 years) was independently associated with the incidences of amenorrhea during chemotherapy (p = 0.007).
Conclusion
Age was the strongest predictor of the incidence of amenorrhea during chemotherapy. It is unclear whether additional taxane may contribute to amenorrhea. This information could be useful in deciding whether to use adjuvant chemotherapy.


Similar content being viewed by others
References
Early Breast Cancer Trialists’ Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–1717.
Bines J, Oleske DM, Cobleigh MA. Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer. J Clin Oncol. 1996;114:1718–29.
Partridge AH, Gelber S, Peppercorn J, Sampson E, Knudsen K, Laufer M, et al. Web-based survey of fertility issues in young women with breast cancer. J Clin Oncol. 2004;22:4174–83.
Early Breast Cancer Trialists’ Collaborative Group. Ovarian ablation in early breast cancer: Overview of the randomised trials. Lancet. 1996;348:1189–96.
Pagani O, O’Neill A, Castiglione M, Gelber RD, Goldhirsch A, Rudenstam CM, et al. Prognostic impact of amenorrhoea after adjuvant chemotherapy in premenopausal breast cancer patients with axillary node involvement: results of the International Breast Cancer Study Group (IBCSG) Trial VI. Eur J Cancer. 1998;34:632–40.
Bianco AR, Del Mastro L, Gallo C, Perrone F, Matano E, Pagliarulo C, et al. Prognostic role of amenorrhea induced by adjuvant chemotherapy in premenopausal patients with early breast cancer. Br J Cancer. 1991;63:799–803.
Warne GL, Fairley KF, Hobbs JB, Martin FI. Cyclophosphamide-induced ovarian failure. N Engl J Med. 1973;289:1159–62.
Hortobagyi GN, Buzdar AU, Marcus CE, Smith TL. Immediate and long-term toxicity of adjuvant chemotherapy regimens containing doxorubicin in trials at MD Anderson Hospital and Tumor Institute. NCI Monogr. 1986;1:105–9.
Swain SM, Land SR, Ritter MW, Costantino JP, Cecchini RS, Mamounas EP, et al. Amenorrhea in premenopausal women on the doxorubicin-and-cyclophosphamide-followed-by-docetaxel arm of NSABP B-30 trial. Breast Cancer Res Treat. 2009;113:315–20.
Goldhirsch A, Gelber RD, Castiglione M. The magnitude of endocrine effects of adjuvant chemotherapy for premenopausal breast cancer patients: the International Breast Cancer Study Group. Ann Oncol. 1990;1:183–8.
Goodwin PJ, Ennis M, Pritchard KI, Trudeau M, Hood N. Risk of menopause during the first year after breast cancer diagnosis. J Clin Oncol. 1999;17:2365–70.
Walshe JM, Denduluri N, Swain SM. Amenorrhea in premenopausal women after adjuvant chemotherapy for breast cancer. J Clin Oncol. 2006;24:2651–8.
Han HS, Ro J, Lee KS, Nam BH, Seo JA, Lee DH, et al. Analysis of chemotherapy-induced amenorrhea rates by three different anthracycline and taxane containing regimens for early breast cancer. Breast Cancer Res Treat. 2009;115:335–42.
Berliere M, Dalenc F, Malingret N, Vindevogel A, Piette P, Roche H, et al. Incidence of reversible amenorrhea in women with breast cancer undergoing adjuvant anthracycline-based chemotherapy with or without docetaxel. BMC Cancer. 2008;8:56–64.
Henderson IC, Berry DA, Demetri GD, Cirrincione CT, Goldstein LJ, Martino S, et al. Improved outcomes from adding sequential paclitaxel but not from escalating doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol. 2003;15:976–83.
Martin M, Pienkowski T, Mackey J, Pawlicki M, Guastalla JP, Weaver C, et al. Adjuvant docetaxel for node-positive breast cancer. N Engl J Med. 2005;352:2302–13.
Tham YL, Sexton K, Weiss H, Elledge R, Friedman LC, Kramer R. The rates of chemotherapy-induced amenorrhea in patients treated with adjuvant doxorubicin and cyclophosphamide followed by a taxane. Am J Clin Oncol. 2007;30:126–32.
Fornier MN, Modi S, Panageas KS, Norton L, Hudis C. Incidence of chemotherapy-induced, long-terme amenorrhea in patients with breast carcinoma age 40 years and younger after adjuvant anthracycline and taxane. Cancer. 2005;104:1575–9.
Davis L, Klitus M, Mintzer D. Chemotherapy-induced amenorrhea from adjuvant breast cancer treatment: the effect of the addition of taxanes. Clin Breast Cancer. 2005;6:421–4.
Abusief ME, Missmer SA, Ginsburg ES, Weeks JC, Partridge AH. The effects of pacritaxel, dose density, and trastuzumab on treatment-related amenorrhea in premenopausal women with breast cancer. Cancer. 2010;116:791–8.
Reh A, Oktem O, Oktay K. Impact of breast cancer chemotherapy on ovarian reserve: a prospective observational analysis by menstrual history and ovarian reserve markers. Fertil Steril. 2008;90:1635–9.
Di Cosimo S, Alimonti A, Ferretti G, Sperduti I, Carlini P, Papaldo P, et al. Incidence of chemotherapy-induced amenorrhea depending on the timing of treatment by menstrual cycle phase in women with early breast cancer. Ann Oncol. 2004;15:1065–71.
Bordeleau L, Pritchard K, Goodwin P, Loprinzi C. Therapeutic options for the management of hot flashes in breast cancer survivors: an evidence-based review. Clin Ther. 2007;29:230–41.
Leining MG, Gelber S, Rosenberg R, Przypyszny M, Winer EP, Partridge AH. Menopausal-type symptoms in young breast cancer survivors. Ann Oncol. 2006;17:1777–82.
Nystedt M, Berglund G, Bolund C, Fornander T, Rutqvist LE. Side effects of adjuvant endocrine treatment in premenopausal breast cancer patients: a prospective randomized study. J Clin Oncol. 2003;21:1836–44.
Gerber B, Stehle H, Ricardo F, Maass N, Fischer D, Sommer HL et al. A prospective randomized multicenter study to prevent chemotherapy-induced ovarian failure with the GnRH-agonist goserelin in young hormone-insensitive breast cancer patients receiving anthracycline containing (neo-) adjuvant chemotherapy (GBG 37). J Clin Oncol. 2009;27:Abstr No. 526.
Burger HG. The endocrinology of the menopause. Maturitas. 1996;23:129–36.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Okanami, Y., Ito, Y., Watanabe, C. et al. Incidence of chemotherapy-induced amenorrhea in premenopausal patients with breast cancer following adjuvant anthracycline and taxane. Breast Cancer 18, 182–188 (2011). https://doi.org/10.1007/s12282-011-0256-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-011-0256-7