Abstract
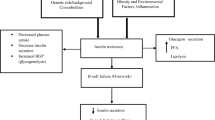
The role of mutations in the melanocortin-3 receptor (MC3R) gene, which is implicated in the regulation of energy homeostasis, is still under debate. Animal studies have clearly proven that, together with the melanocortin-4 receptor (MC4R), the MC3R is a critical receptor for melanocortin peptides within the leptin-melanocortin signaling cascade. However, as several mutations have been found in lean individuals and not all mutations seem to cause receptor dysfunction, results from mutation screens in obese humans remain controversial. In the present study, we screened for rare variants in the MC3R gene of obese children and lean controls to assess the prevalence of MC3R mutations in the Belgian population. We screened 249 severely overweight and obese children and adolescents and 239 lean adults for mutations in the coding region of MC3R. Mutation screening was performed by high resolution melting curve analysis and direct sequencing. We identified four non-synonymous coding variations in the obese population, all of which had been reported previously. In addition, we also found four novel rare MC3R variants in the lean control population, suggesting that not all MC3R mutations are disease-causing. Overall, the total prevalence of rare MC3R variants was 1 % in Belgian obese children and adolescents compared to 1.02 % in lean controls. Ultimately, cosegregation studies combined with comprehensive functional analysis is required to determine the potential pathogenic role of rare MC3R variants in causing human obesity.

Similar content being viewed by others
References
S. Beckers, D. Zegers, L.F. Van Gaal, W. Van Hul, The role of the leptin-melanocortin signalling pathway in the control of food intake. Crit. Rev. Eukaryot. Gene Expr. 19(4), 267–287 (2009)
Y. Zhang, G.E. Kilroy, T.M. Henagan, V. Prpic-Uhing, W.G. Richards, A.W. Bannon, R.L. Mynatt, T.W. Gettys, Targeted deletion of melanocortin receptor subtypes 3 and 4, but not CART, alters nutrient partitioning and compromises behavioral and metabolic responses to leptin. Faseb J. 19(11), 1482–1491 (2005)
A.A. Butler, R.A. Kesterson, K. Khong, M.J. Cullen, M.A. Pelleymounter, J. Dekoning, M. Baetscher, R.D. Cone, A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology 141(9), 3518–3521 (2000)
A.S. Chen, D.J. Marsh, M.E. Trumbauer, E.G. Frazier, X.M. Guan, H. Yu, C.I. Rosenblum, A. Vongs, Y. Feng, L. Cao, J.M. Metzger, A.M. Strack, R.E. Camacho, T.N. Mellin, C.N. Nunes, W. Min, J. Fisher, S. Gopal-Truter, D.E. MacIntyre, H.Y. Chen, L.H. Van der Ploeg, Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat. Genet. 26(1), 97–102 (2000)
C. Vaisse, K. Clement, B. Guy-Grand, P. Froguel, A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nat. Genet. 20(2), 113–114 (1998)
G.S. Yeo, I.S. Farooqi, S. Aminian, D.J. Halsall, R.G. Stanhope, S. O’Rahilly, A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat. Genet. 20(2), 111–112 (1998)
Y.X. Tao, The melanocortin-4 receptor: physiology, pharmacology, and pathophysiology. Endocr. Rev. 31(4), 506–543 (2010)
D. Zegers, S. Beckers, F. de Freitas, A.V. Peeters, I.L. Mertens, S.L. Verhulst, R.P. Rooman, J.P. Timmermans, K.N. Desager, G. Massa, L.F. Van Gaal, W. Van Hul, Identification of three novel genetic variants in the melanocortin-3 receptor of obese children. Obesity (Silver Spring) 19(1), 152–159 (2011)
M.A. Calton, B.A. Ersoy, S. Zhang, J.P. Kane, M.J. Malloy, C.R. Pullinger, Y. Bromberg, L.A. Pennacchio, R. Dent, R. McPherson, N. Ahituv, C. Vaisse, Association of functionally significant Melanocortin-4 but not Melanocortin-3 receptor mutations with severe adult obesity in a large North American case-control study. Hum. Mol. Genet. 18(6), 1140–1147 (2009)
Y.S. Lee, L.K. Poh, K.Y. Loke, A novel melanocortin 3 receptor gene (MC3R) mutation associated with severe obesity. J. Clin. Endocrinol. Metab. 87(3), 1423–1426 (2002)
Y.S. Lee, L.K. Poh, B.L. Kek, K.Y. Loke, The role of melanocortin 3 receptor gene in childhood obesity. Diabetes 56(10), 2622–2630 (2007)
M. Mencarelli, G.E. Walker, S. Maestrini, L. Alberti, B. Verti, A. Brunani, M.L. Petroni, M. Tagliaferri, A. Liuzzi, A.M. Di Blasio, Sporadic mutations in melanocortin receptor 3 in morbid obese individuals. Eur. J. Hum. Genet. 16(5), 581–586 (2008)
M. Mencarelli, B. Dubern, R. Alili, S. Maestrini, L. Benajiba, M. Tagliaferri, P. Galan, M. Rinaldi, C. Simon, P. Tounian, S. Hercberg, A. Liuzzi, A.M. Di Blasio, K. Clement, Rare melanocortin-3 receptor mutations with in vitro functional consequences are associated with human obesity. Hum. Mol. Genet. 20(2), 392–399 (2011)
M. Roelants, R. Hauspie, K. Hoppenbrouwers, References for growth and pubertal development from birth to 21 years in Flanders. Belgium Ann. Hum. Biol. 36(6), 680–694 (2009)
S.A. Miller, D.D. Dykes, H.F. Polesky, A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 16(3), 1215 (1988)
Y.X. Tao, Mutations in the melanocortin-3 receptor (MC3R) gene: impact on human obesity or adiposity. Curr. Opin. Investig. Drugs 11(10), 1092–1096 (2010)
Y.X. Tao, D.L. Segaloff, Functional characterization of melanocortin-3 receptor variants identify a loss-of-function mutation involving an amino acid critical for G protein-coupled receptor activation. J. Clin. Endocrinol. Metab. 89(8), 3936–3942 (2004)
M. Rached, A. Buronfosse, M. Begeot, A. Penhoat, Inactivation and intracellular retention of the human I183N mutated melanocortin 3 receptor associated with obesity. Biochim. Biophys. Acta 1689(3), 229–234 (2004)
Y.X. Tao, Functional characterization of novel melanocortin-3 receptor mutations identified from obese subjects. Biochim. Biophys. Acta 1772(10), 1167–1174 (2007). doi:10.1016/j.bbadis.2007.09.002
F. Yang, Y.X. Tao, Functional characterization of nine novel naturally occurring human melanocortin-3 receptor mutations. Biochim. Biophys. Acta 1822(11), 1752–1761 (2012). doi:10.1016/j.bbadis.2012.07.017
Acknowledgments
Research was funded by a PhD grant of the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT-Vlaanderen) to DZ. SB is a postdoctoral fellow of the Research Foundation - Flanders (FWO Vlaanderen). This work was supported by a grant (G0028.05) from the Research Foundation Flanders (FWO Vlaanderen) to LVG and WVH and by a TOP-research grant from the University of Antwerp to WVH.
Conflict of interest
None disclosed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zegers, D., Beckers, S., Hendrickx, R. et al. Prevalence of rare MC3R variants in obese cases and lean controls. Endocrine 44, 386–390 (2013). https://doi.org/10.1007/s12020-012-9862-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9862-1