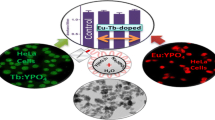
Abstract
Stable and non-invasive fluorescent probes for nanotoxicological investigations are greatly needed to track the fate of nanoparticles in biological systems. The potential for terbium (Tb) to act as a fluorescent probe and its effect on layered double hydroxide (LDH) nanoparticle morphology are presented in this study. Incorporation of Tb during synthesis offers a simple methodology to easily tailor LDH nanoparticle thickness. A three-fold reduction in the average crystallite thickness (from 13 to 4 nm) has been achieved, whilst preferential lateral growth of LDH nanoparticles in the a-b crystal plane has been observed with increasing Tb loadings. Remarkably, Tb–LDH nanoparticles have emitted green fluorescence with a fluorescence quantum yield of 0.044.









Similar content being viewed by others
References
Adachi-Pagano M, Forano C, Besse J-P (2000) Delamination of layered double hydroxides by use of surfactants. Chem Commun (Camb) (1):91–92. doi:10.1039/a908251d
Baraton MI, Chen X, Gonsalves KE (1997) FTIR study of a nanostructured aluminium nitride powder surface: determination of the acidic/basic sites by CO, CO2 and acetic acid absorptions. Nanostruct Mater 8(4):435–445. doi:10.1016/S0965-9773(97)00189-X
Braterman PS, Xu ZP, Yarberry F (2004) Layered double hydroxides (LDHs). In: Auerback SM, Carrado KA, Dutta PK (eds) Handbook of layered materials. Marcell Dekker, New York, pp 373–474
Calvin S, Miller MM, Goswami R, Cheng SF, Mulvaney SP, Whitman LJ, Harris VG (2003) Determination of crystallite size in a magnetic nanocomposite using extended X-ray absorption fine structure. J Appl Phys 94(1):778–783. doi:10.1063/1.1581344
Cavani F, Trifiro F, Vaccari A (1991) Hydrotalcite-type anionic clays: preparation, properties and applications. Catal Today 11(2):173–301. doi:10.1016/0920-5861(91)80068-K
Chang Z, Evans D, Duan X, Boutinaud P, de Roy M, Forano C (2006) Preparation and characterization of rare earth-containing layered double hydroxide. J Phys Chem Solids 67:1054–1057. doi:10.1016/j.jpcs.2006.01.025
Choy J-H, Kwak S-Y, Jeong Y-J, Park J-S (2000) Inorganic layered double hydroxides as nonviral vectors. Angew Chem Int Ed 39(22):4042–4045. doi:10.1002/1521-3773(20001117)39:22<4041::AID-ANIE4041>3.0.CO;2-C
Greenwell HC, Bindley LA, Unwin PR, Holliman PJ, Jones W, Coveney PV, Barnes SL (2006) In situ monitoring of crystal growth and dissolution of oriented layered double-hydroxide crystals immobilized on silicon. J Cryst Growth 294(1):53–59. doi:10.1016/j.jcrysgro.2006.05.048
Hernandez-Moreno MJ, Uilbarri MA, Rendon JL, Serna CJ (1985) IR characteristics of hydrotalcite-like compounds. Phys Chem Miner 12:34–38
Hibino T, Jones W (2001) New approach to the delamination of layered double hydroxides. J Mater Chem 11(5):1321–1323. doi:10.1039/b101135i
Iannou PC, Rusakova NV, Andrikopoulou DA, Glynou KM, Tzompanaki GM (1998) Spectrofluorimetric determination of anthranilic acid derivatives based on terbium sensitized fluorescence. Analyst (Lond) 123:2839–2843. doi:10.1039/a806093b
Kannan S (2004) Influence of synthesis methodology and post treatments on structural and textural variations in MgAlCO3 hydrotalcite. J Mater Sci 39(21):6591–6596. doi:10.1023/B:JMSC.0000044900.22280.93
Kovanda F, Kolousek D, Cilova Z, Hulinsky V (2005) Crystallization of synthetic hydrotalcite under hydrothermal conditions. Appl Clay Sci 28(1–4):101–109. doi:10.1016/j.clay.2004.01.009
Li B, He J, Evans DG, Duan X (2006a) Morphology and size control of Ni-Al layered double hydroxides using chitosan as template. J Phys Chem Solids 67(5–6):1067–1070. doi:10.1016/j.jpcs.2006.01.027
Li L, Ma R, Iyi N, Ebina Y, Takada K, Sasaki T (2006b) Hollow nanoshell of layered double hydroxide. Chem Commun (Camb) (29):3125–3127. doi:10.1039/b605889b
Martini JL, Tetreau C, Pochon F, Tourbez H, Lentz JM, Lavalette D (1993) On the mechanism of energy transfer to Tb3+ ions in proteins. A time-resolved luminescence study of the Tb-elastase complex. Eur J Biochem 211(3):467–473. doi:10.1111/j.1432-1033.1993.tb17572.x
Mohmel S, Kurzawski I, Uecker D, Muller D, Gessner W (2002) The influence of a hydrothermal treatment using microwave heating on the crystallinity of layered double hydroxides. Cryst Res Technol 37(4):359–369. doi:10.1002/1521-4079(200204)37:4<359::AID-CRAT359>3.0.CO;2-I
Nijs H, Clearfield A, Vansant EF (1998) The intercalation of phenylphosphonic acid in layered double hydroxides. Microporous Mesoporous Mater 23(1–2):97–108. doi:10.1016/S1387-1811(98)00056-0
Oh J-M, Hwang S-H, Choy J-H (2002) The effect of synthetic conditions on tailoring the size of hydrotalcite particles. Solid State Ion 151(1–4):285–291. doi:10.1016/S0167-2738(02)00725-7
Oh J-M, Park M, Kim S-T, Jung J-Y, Kang Y-G, Choy J-H (2006) Efficient delivery of anticancer drug MTX through MTX-LDH nanohybrid system. J Phys Chem Solids 67(5–6):1024–1027. doi:10.1016/j.jpcs.2006.01.033
Stumpf T, Curtius H, Walther C, Dardenne K, Ufer K, Fanghael T (2007) Incorporation of Eu(III) into hydrotalcite: a TRLFS and EXAFS study. Environ Sci Technol 41:3186–3191. doi:10.1021/es0624873
Thomas GS, Kamath PV (2006) Line broadening in the PXRD patterns of layered hydroxides: the relative effects of crystallite size and structural disorder. J Chem Sci 118(1):127–133. doi:10.1007/BF02708774
Trifiro F, Vaccari A (1996) Comprehensive supramolecular chemistry, solid state supramolecular chemistry: two and three-dimensional inorganic networks. Pergamon Press, Oxford
Wu Q, Sjastad AO, Vistad OB, Knudsen KD, Roots J, Pedersen JS, Norby P (2007) Characterization of exfoliated (LDH, Mg/Al = 3) nanosheets at high concentrations in formamide. J Mater Chem 17(10):965–971. doi:10.1039/b612389a
Xu ZP, Zeng HC (2001) Abrupt structural transformation in hydrotalcite-like compounds Mg1-xAlx(OH)2(NO3)x · nH2O as a continuous function of nitrate anions. J Phys Chem B 105:1743–1749. doi:10.1021/jp0029257
Xu ZP, Lu GQ (Max) (2005) (WO/2006/066341) Preparation of Suspensions PCT Patent WO 20006066341 Patent Number: 2004908326, 29 June 2006
Xu ZP, Stevenson G, Lu C-Q, Lu GQ (2006a) Dispersion and size control of layered double hydroxide nanoparticles in aqueous solutions. J Phys Chem B 110(34):16923–16929. doi:10.1021/jp062281o
Xu ZP, Stevenson GS, Lu C-Q, Lu GQ, Bartlett PF, Gray PP (2006b) Stable suspension of layered double hydroxide nanoparticles in aqueous solution. J Am Chem Soc 128(1):36–37. doi:10.1021/ja056652a
Xu ZP, Kurniawan ND, Bartlett PF, Lu GQ (2007) Enhancement of relaxivity rates of Gd-DTPA complexes by intercalation into layered double hydroxide nanoparticles. Chem Eur J 13:2824–2830. doi:10.1002/chem.200600571
Zhuravela NG, Eliseev AA, Lukashin AV, Kynast U, Tret’yakov AYD (2004) Luminescent materials based on Tb- and Eu-containing layered double hydroxides. Dokl Chem 396(1):87–91. doi:10.1023/B:DOCH.0000029538.18156.62
Acknowledgements
The authors would like to acknowledge and thank funding support from the AIBN Challenge Project fund and the ARC Center of Excellence for Functional Nanomaterials. The authors would also like to acknowledge the kind support of the Centre of Microscopy and Microanalysis at UQ
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Musumeci, A.W., Xu, Z.P., Smith, S.V. et al. Layered double hydroxide nanoparticles incorporating terbium: applicability as a fluorescent probe and morphology modifier. J Nanopart Res 12, 111–120 (2010). https://doi.org/10.1007/s11051-008-9583-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-008-9583-9