Abstract
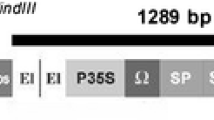
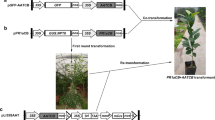
Citrus scab, caused by Elsinoë fawcettii (anamorph Sphaceloma fawcettii), is a common foliar fungal disease affecting many citrus cultivars, including grapefruit. No commercial grapefruit cultivar is resistant to scab, and the disease results in severely blemished fruit which reduces its marketability. Transgenic ‘Duncan’ grapefruit trees expressing the antimicrobial attE gene were produced via Agrobacterium-mediated transformation. In in vitro leaf and greenhouse assays, several transgenic-lines had significantly lower susceptibility to E. fawcettii compared to the non-transformed control (P < 0.0001). In the greenhouse studies, sporulation on all transgenic lines except 1 was significantly reduced (P < 0.0001) but the level of sporulation over time did not correspond to disease severity ratings. Lesion size was also significantly reduced on transgenic lines compared to the non-transformed control (P < 0.0001) and the least susceptible line A-23 had the smallest lesions, but in general there was no correlation between lesion size and disease susceptibility. The level of attE mRNA was inversely related to the number of copies detected by Southern blot. The least susceptible line had a single inserted copy of the attE transgene whereas more susceptible lines had multiple copies. Since the attacin mode of action was thought to be specific to Gram-negative bacteria, it was unexpected to find that there was a significant activity against E. fawcettii.







Similar content being viewed by others
References
Bitancourt, A. A., & Jenkins, A. E. (1936). Elsinoë fawcettii, the perfect stage of citrus scab fungus. Phytopathology, 26, 393–396.
Broekaert, W. F., Cammue, B. P. A., De Bolle, M. F. C., Thevissen, K., De Samblanx, G. W., & Osborn, R. W. (1997). Antimicrobial peptides from plants. Critical Review of Plant Science, 16, 297–323.
Brunner, E., Domhof, S., & Langer, F. (2002). Nonparametric analysis of longitudinal data in factorial experiments. New York: John Wiley & Sons.
Burrow, M. D., Chlan, C. A., Sen, P., & Murai, N. (1990). High frequency generation of transgenic tobacco plants after modified leaf disk co-cultivation with Agrobacterium tumefaciens. Plant Molecular Biology Reporter, 8, 124–139.
Campo, S., Manrique, S., García-Martínez, J., & San Segundo, B. (2008). Production of cecropin A in transgenic rice plants has an impact on host gene expression. Plant Biotechnology Journal, 6, 585–608.
Cardoso, S. C., Mendes, J. M. B., Camargo, R. L. B., Christiano, R. S. C., Filho, A. B., Vieira, M. L. C., et al. (2010). Transgenic sweet orange (Citrus sinensis L. Osbeck) expressing the attacin A gene for resistance to Xanthomonas citri subsp. citri. Plant Molecular and Biological Report, 28, 185–192.
Carlsson, A., Nystrom, T., de Cock, H., & Bennich, H. (1998). Attacin - an insect immune protein - binds LPS and triggers the specific inhibition of bacterial outer-membrane protein synthesis. Microbiology-Society for General Microbiology, 144, 2179–2188.
Carlsson, A., Engstrom, P., Palva, E. T., & Bennich, H. (1991). Attacin, an antibacterial protein from Hyalophora cecropia, inhibits synthesis of outer membrane proteins in Escherichia coli by interfering with omp gene transcription. Infection and Immunity, 59, 3040–3045.
Coca, M., Peñas, G., Gómez, J., Campo, S., Bortolotti, C., Messeguer, J., et al. (2006). Enhanced resistance to the rice blast fungus Magnaporthe grisea conferred by expression of a cecropin A gene in transgenic rice. Planta, 223, 392–406.
DeLucca, A. J., Bland, J. M., Jacks, T. J., Grimm, C., Cleveland, T. E., & Walsh, T. J. (1997). Fungicidal activity of cecropin A. Antimicrobial Agents and Chemotherapy, 4, 481–483.
De Lucca, A. J., Bland, J. M., Grimm, C., Jacks, T. J., Cary, J. W., Jaynes, J. M., et al. (1998). Fungicidal properties, sterol binding, and proteolytic resistance of the synthetic peptide D4E1. Canadian Journal of Microbiology, 44, 514–520.
Dewdney, M. M., & Timmer, L. W. (2010) Citrus scab. In M. E. Rogers, M. M. Dewdney, & T. M. Spann (Eds), 2011 Florida Citrus Pest Management Guide: University of Florida, IFAS. pp. 3. http://edis.ifas.ufl.edu/cg020).
Dutt, M., & Grosser, J. (2009). Evaluation of parameters affecting Agrobacterium-mediated transformation of citrus. Plant Cell, Tissue and Organ Culture, 98, 331–340.
Dutt, M., Orbovic, V., & Grosser, J. W. (2009). Cultivar dependent gene transfer into citrus using Agrobacterium. Proceedings of the Florida State Horticultural Society, 122, 85–89.
Dutt, M., Madhavaraj, J., & Grosser, J. W. (2010). Agrobacterium tumefaciens-mediated genetic transformation and plant regeneration from a complex tetraploid hybrid citrus rootstock. Scientia Horticulturae, 123, 454–458.
Engstrom, P., Carlsson, A., Engstrom, Å., Tao, Z.-J., & Bennich, H. (1984). The antibacterial effect of attacins from the silk moth Hyalophora cecropia is directed against the outer membrane of Escherichia coli. EMBO Journal, 3, 3347–3351.
Gelvin, S. B. (2000). Agrobacterium and plant genes involved in T-DNA transfer and integration. Annual Reviews of Plant Physiology and Plant Molecular Biology, 51, 223–256.
Gmitter, F. G., Jr. (2010). Origin, evolution, and breeding of the grapefruit. In J. Janick (Ed.), Plant breeding reviews, volume 13. Oxford: John Wiley & Sons, Inc. doi:10.1002/9780470650059.ch10.
Grosser, J. W., & Gmitter, F. G., Jr. (2010). Protoplast fusion in the production of tetraploids and triploids: Applications in scion and rootstock breeding. Plant Cell Tissue and Organ Culture, 104, 343–357.
Hobbs, S. L., Kpodar, P., & Delong, C. M. (1990). The effect of T-DNA copy number, position and methylation on reporter gene expression in tobacco transformants. Plant Molecular Biology, 15, 851–864.
Hood, E. E., Gelvin, S. B., Melchers, L. S., & Hoekema, A. (1993). New Agrobacterium helper plasmids for gene transfer to plants. Transgenic Research, 2, 208–218.
Hultmark, D., Engstrom, A., Andersson, K., Steiner, H., Bennich, H., & Boman, H. G. (1983). Insect immunity. Attacins, a family of antibacterial proteins from Hyalophora cecropia. EMBO Journal, 2, 571–576.
Hyun, J. W., Yi, S. H., Mackenzie, S. J., Timmer, L. W., Kim, K. S., Kang, S. K., et al. (2009). Pathotypes and genetic relationship of worldwide collections of Elsinoë spp. causing scab diseases of citrus. Phytopathology, 99, 721–728.
Ieki, H. (1981). Resistance of Citrus to scab. International Citrus Congress (4th : 1981 : Tokyo, Japan) International Society of Citriculture, 1, 340–344.
Jaynes, J. M., Nagpala, P., Destéfano-Beltran, L., Huang, J. H., Kim, J., Denny, T., et al. (1993). Expression of a cercopin B lytic peptide analog in transgenic tobacco confers enhanced resistance to bacterial wilt caused by Pseudomonas solanacearum. Plant Science, 89, 43–53.
Kim, K. W., Hyun, J. W., & Park, E. W. (2004). Cytology of cork layer formation of citrus and limited growth of Elsinoë fawcettii in scab lesions. European Journal of Plant Pathology, 110, 129–138.
Ko, K., Norelli, J. L., Reynoird, J.-P., Boresjza-Wysocka, E., Brown, S. K., & Aldwinckle, H. S. (2000). Effect of untranslated leader sequence of AMV RNA4 and signal peptide of pathogenesis-related protein 1b on attacin gene expression, and resistance to fire blight in transgenic apple. Biotechnology Letters, 22, 373–381.
Kumar, S., & Fladung, M. (2001). Gene stability in transgenic aspen (Populus). II. Molecular characterization of variable expression of transgene in wild and hybrid aspen. Planta, 213, 731–740.
Li, Z., Jayasankar, S., & Gray, D. J. (2001). Expression of a bifunctional green fluorescent protein (GFP) fusion marker under the control of three constitutive promoters and enhanced derivatives in transgenic grape (Vitis vinifera). Plant Science, 160, 877–887.
Martin, D. I., & Whitelaw, E. (1996). The vagaries of variegating transgenes. Bioessays, 18, 919–923.
Mayerhofer, R., Koncz-Kalman, Z., Nawrath, C., Bakkeren, G., Crameri, A., Angelis, K., et al. (1991). T-DNA integration: a model of illegitimate recombination in plants. EMBO Journal, 10, 697–704.
Mitsuhara, I., Matsufuru, H., Ohshima, M., Kaku, H., Nakajima, Y., Murai, N., et al. (2000). Induced expression of sarcotoxin1A enhanced host resistance against both bacterial and fungal pathogens in transgenic tobacco. Molecular Plant-Microbe Interactions, 13, 860–868.
Norelli, J. L., Aldwinckle, H. S., Destéfano-Beltrán, L., & Jaynes, J. M. (1994). Transgenic ‘Mailing 26’ apple expressing the attacin E gene has increased resistance to Erwinia amylovora. Euphytica, 77, 123–128.
Norelli, J. L., Borejsza-Wysocka, E., Reynoird, J.-P., & Aldwinckle, H. S. (2000). Transgenic ‘Royal Gala’ apple expressing Attacin E has increased field resistance to Erwinia amylovora (fire blight). Acta Hortculturae, 538, 631–633.
Osusky, M., Zhou, G., Osuska, L., Hancock, R. E., Kay, W. W., & Misra, S. (2000). Transgenic plants expressing cationic peptide chimeras exhibit broad-spectrum resistance to phytopathogens. Nature Biotechology, 18, 1162–1166.
Pena, L., Cervera, M., Ghorbel, R., Domínguez, A., Fagoaga, C., Juarez, J., et al. (2007). Genetic transformation. In I. A. Khan (Ed.), Citrus genetics, breeding, and biotechnology (pp. 329–344). Wallingford: CABI International.
Rajasekaran, K., Stromberg, K. D., Cary, J. W., & Cleveland, T. E. (2001). Broad-Spectrum Antimicrobial Activity in vitro of the Synthetic Peptide D4E1. Journal of Agricultural and Food Chemistry, 49, 2799–2803.
Reddy, M. S., Dinkins, R. D., & Collins, G. B. (2003). Gene silencing in transgenic soybean plants transformed via particle bombardment. Plant Cell Report, 21, 676–683.
Reynoird, J. P., Mourgues, F., Norelli, J., Aldwinckle, H. S., Brisset, M. N., & Chevreau, E. (1999). First evidence for improved resistance to fire blight in transgenic pear expressing the attacin E gene from Hyalophora cecropia. Plant Science, 149, 23–31.
Shah, D. A., & Madden, L. V. (2004). Nonparametric analysis of ordinal data in designed factorial experiments. Phytopathology, 94, 33–43.
Thevissen, K., Ferket, K. K. A., François, I. E. J. A., & Cammue, B. P. A. (2003). Interactions of antifungal plant defensins with fungal membrane components. Peptides, 24(11), 1705–1712.
Timmer, L. W. (2000). Scab diseases. In L. W. Timmer, S. M. Garnsey, & J. H. Graham (Eds.), Compendium of citrus diseases (2nd ed., pp. 31–32). St. Paul: American Phytopathological Society Press.
Timmer, L. W., & Zitko, S. E. (1993) Techniques for greenhouse evaluation of fungicides for control of citrus scab. In E. Rabe (Ed.), Proceedings of the IV Congress of the International Society of Citrus Nurserymen (pp. 125–129). Johannesburg: The South African Citrus Nurserymen’s Association.
Timmer, L. W., Priest, M., Broadbent, P., & Tan, M.-K. (1996). Morphological and pathological characterization of species of Elsinoë causing scab diseases of citrus. Phytopathology, 86, 1032–1038.
Timmer, L. W., Roberts, P. D., Chung, K. R., & Bhatia, A. (2001). Citrus scab (PP153): University of Florida Institute of Food and Agricultural Sciences. http://edis.ifas.ufl.edu/ch014.
Timmer, L. W., Mondal, S. N., Peres, N. A. R., & Bhatia, A. (2004). Fungal diseases of fruit and foliage of citrus trees. In S. A. M. H. Naqvi (Ed.), Diseases of Fruits and Vegetables – Diagnosis and Management, vol. 1, (pp. 191–227). Dordrecht: Kluwer Academic Publishers.
Tollin, M., Bergman, P., Svenberg, T., Jörnvall, H., Gudmundsson, G. H., & Agerberth, B. (2003). Antimicrobial peptides in the first line defence of human colon mucosa. Peptides, 24, 523–530.
Van Attikum, H., Bundock, P., & Hooykaas, P. J. J. (2001). Non-homologous end-joining proteins are required for Agrobacterium T-DNA integration. EMBO Journal, 20, 6550–6558.
van Hofsten, P., Faye, I., Kockum, K., Lee, J. Y., Xanthopoulos, K. G., Boman, I. A., et al. (1985). Molecular cloning, cDNA sequencing, and chemical synthesis of cecropin B from Hyalophora cecropia. Proceedings of the National Academy of Science USA, 82, 2240–2243.
Whiteside, J. O. (1975). Biological characteristics of Elsinoë fawcettii pertaining to the epidemiology of sour orange scab. Phytopathology, 65, 1170–1177.
Acknowledgments
The authors would like to thank Dr. Herb Aldwinckle, NYSAES Cornell University, USA for providing us with the pCa2Att/121 clone and Drs. Dennis Gray and Zhijian Li, MREC, University of Florida, USA for providing us with a binary vector containing the bifunctional nptII/egfp fusion gene.
Author information
Authors and Affiliations
Corresponding author
Additional information
S. N. Mondal and M. Dutt contributed equally to this work.
Rights and permissions
About this article
Cite this article
Mondal, S.N., Dutt, M., Grosser, J.W. et al. Transgenic citrus expressing the antimicrobial gene Attacin E (attE) reduces the susceptibility of ‘Duncan’ grapefruit to the citrus scab caused by Elsinoë fawcettii . Eur J Plant Pathol 133, 391–404 (2012). https://doi.org/10.1007/s10658-011-9912-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-011-9912-1