Abstract
Background
The fatty acid analogue tetradecylthioacetic acid (TTA) is a moderate pan-activator of peroxisome proliferator-activated receptors (PPARs), and has in previous studies showed potential as an antioxidant and anti-inflammatory agent, both through PPAR and non-PPAR mediated mechanisms.
Aims
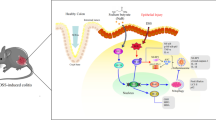
This study aimed to determine whether TTA could alleviate dextran sulfate sodium (DSS)-induced colitis in rats.
Methods
Male Wistar rats were fed a control diet (control- and DSS-group) or a diet supplemented with 0.4 % TTA (TTA + DSS-group) for 30 days, and DSS was added to the drinking water the last 7 days. Ultrasound measurements were performed at day 29. At day 30, rats were sacrificed and the distal colon was removed for histological evaluation and measurement of cytokine levels, oxidative damage, and gene expression.
Results
The disease activity index was not improved in the TTA + DSS-group compared to the DSS-group. However, ultrasound measurements showed a significantly reduced colonic wall thickening in the TTA + DSS-group. TNF-α, IL-1β, and IL-6 were reduced at the protein and mRNA level in the TTA + DSS-group. Moreover, TTA-treated rats demonstrated reduced colonic oxidative damage, while inducible nitric oxide synthase 2 mRNA expression was elevated in both the DSS- and TTA + DSS-groups. PPARγ signaling may be involved in the anti-inflammatory response to TTA, as Pparg mRNA expression was significantly upregulated in colon.
Conclusions
This study demonstrated that the pan-PPAR agonist TTA reduced colonic oxidative damage and cytokine levels in a rat model of colitis, and its potential to ameliorate colitis should be further explored.






Similar content being viewed by others
References
Lomer MC. Dietary and nutritional considerations for inflammatory bowel disease. Proc Nutr Soc. 2011;70:329–335.
Nicholls RJ. Review article: ulcerative colitis-surgical indications and treatment. Aliment Pharmacol Ther. 2002;16:25–28.
Rosenstiel P, Schreiber S. Expression signatures, barriers and beyond: the role of oxidative stress in murine colitis and human inflammatory bowel disease revisited. Eur J Gastroenterol Hepatol. 2008;20:496–499.
Dubuquoy L, Rousseaux C, Thuru X, Peyrin-Biroulet L, et al. PPARgamma as a new therapeutic target in inflammatory bowel diseases. Gut. 2006;55:1341–1349.
Su CG, Wen X, Bailey ST, Jiang W, et al. A novel therapy for colitis utilizing PPAR-gamma ligands to inhibit the epithelial inflammatory response. J Clin Invest. 1999;104:383–389.
Shah YM, Morimura K, Gonzalez FJ. Expression of peroxisome proliferator-activated receptor-gamma in macrophage suppresses experimentally induced colitis. Am J Physiol Gastrointest Liver Physiol. 2007;292:G657–G666.
Celinski K, Dworzanski T, Korolczuk A, Piasecki R, et al. Effects of peroxisome proliferator-activated receptors-gamma ligands on dextran sodium sulphate-induced colitis in rats. J Physiol Pharmacol. 2011;62:347–356.
Pedersen G, Brynskov J. Topical rosiglitazone treatment improves ulcerative colitis by restoring peroxisome proliferator-activated receptor-gamma activity. Am J Gastroenterol. 2010;105:1595–1603.
Bassaganya-Riera J, DiGuardo M, Climent M, Vives C, et al. Activation of PPARgamma and delta by dietary punicic acid ameliorates intestinal inflammation in mice. Br J Nutr. 2011;106:878–886.
Azuma YT, Nishiyama K, Matsuo Y, Kuwamura M, et al. PPARalpha contributes to colonic protection in mice with DSS-induced colitis. Int Immunopharmacol. 2010;10:1261–1267.
Peyrin-Biroulet L, Beisner J, Wang G, Nuding S, et al. Peroxisome proliferator-activated receptor gamma activation is required for maintenance of innate antimicrobial immunity in the colon. Proc Natl Acad Sci USA. 2010;107:8772–8777.
Madsen L, Guerre-Millo M, Flindt EN, Berge K, et al. Tetradecylthioacetic acid prevents high fat diet induced adiposity and insulin resistance. J Lipid Res. 2002;43:742–750.
Berge RK, Madsen L, Vaagenes H, Tronstad KJ, Gottlicher M, Rustan AC. In contrast with docosahexaenoic acid, eicosapentaenoic acid and hypolipidaemic derivatives decrease hepatic synthesis and secretion of triacylglycerol by decreased diacylglycerol acyltransferase activity and stimulation of fatty acid oxidation. Biochem J. 1999;343:191–197.
Berge RK, Skorve J, Tronstad KJ, Berge K, Gudbrandsen OA, Grav H. Metabolic effects of thia fatty acids. Curr Opin Lipidol. 2002;13:295–304.
Dyroy E, Yndestad A, Ueland T, Halvorsen B, et al. Antiinflammatory effects of tetradecylthioacetic acid involve both peroxisome proliferator-activated receptor alpha-dependent and -independent pathways. Arterioscler Thromb Vasc Biol. 2005;25:1364–1369.
Aukrust P, Wergedahl H, Muller F, Ueland T, et al. Immunomodulating effects of 3-thia fatty acids in activated peripheral blood mononuclear cells. Eur J Clin Invest. 2003;33:426–433.
Muna ZA, Gudbrandsen OA, Wergedahl H, Bohov P, Skorve J, Berge RK. Inhibition of rat lipoprotein oxidation after tetradecylthioacetic acid feeding. Biochem Pharmacol. 2002;63:1127–1135.
Vigerust NF, Cacabelos D, Burri L, Berge K, et al. Fish oil and 3-thia fatty acid have additive effects on lipid metabolism but antagonistic effects on oxidative damage when fed to rats for 50 weeks. J Nutr Biochem. 2011. (Epub ahead of print). doi:10.1016/j.bbr.2011.03.031.
Grimstad T, Bjorndal B, Cacabelos D, Aasprong OG, et al. Dietary supplementation of krill oil attenuates inflammation and oxidative stress in experimental ulcerative colitis in rats. Scand J Gastroenterol. 2011;47:49–58.
Spydevold O, Bremer J. Induction of peroxisomal beta-oxidation in 7800 C1 Morris hepatoma cells in steady state by fatty acids and fatty acid analogues. Biochim Biophys Acta. 1989;1003:72–79.
Okayasu I, Hatakeyama S, Yamada M, Ohkusa T, Inagaki Y, Nakaya R. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology. 1990;98:694–702.
Carrier J, Aghdassi E, Platt I, Cullen J, Allard JP. Effect of oral iron supplementation on oxidative stress and colonic inflammation in rats with induced colitis. Aliment Pharmacol Ther. 2001;15:1989–1999.
Fu MX, Requena JR, Jenkins AJ, Lyons TJ, Baynes JW, Thorpe SR. The advanced glycation end product, Nepsilon-(carboxymethyl)lysine, is a product of both lipid peroxidation and glycoxidation reactions. J Biol Chem. 1996;271:9982–9986.
Requena JR, Fu MX, Ahmed MU, Jenkins AJ, et al. Quantification of malondialdehyde and 4-hydroxynonenal adducts to lysine residues in native and oxidized human low-density lipoprotein. Biochem J. 1997;322:317–325.
Requena JR, Chao CC, Levine RL, Stadtman ER. Glutamic and aminoadipic semialdehydes are the main carbonyl products of metal-catalyzed oxidation of proteins. Proc Natl Acad Sci USA. 2001;98:69–74.
Petersson J, Schreiber O, Hansson GC, Gendler SJ, et al. Importance and regulation of the colonic mucus barrier in a mouse model of colitis. Am J Physiol Gastrointest Liver Physiol. 2011;300:G327–G333.
Natividad JM, Petit V, Huang X, de Palma G, et al. Commensal and probiotic bacteria influence intestinal barrier function and susceptibility to colitis in Nod1(−/−);Nod2(−/−) mice. Inflamm Bowel Dis. 2011. (Epub ahead of print). doi:10.1002/ibd.22848.
Yamamoto-Furusho JK, Penaloza-Coronel A, Sanchez-Munoz F, Barreto-Zuniga R, Dominguez-Lopez A. Peroxisome proliferator-activated receptor-gamma (PPAR-gamma) expression is downregulated in patients with active ulcerative colitis. Inflamm Bowel Dis. 2011;17:680–681.
Raspe E, Madsen L, Lefebvre AM, Leitersdorf I, et al. Modulation of rat liver apolipoprotein gene expression and serum lipid levels by tetradecylthioacetic acid (TTA) via PPARalpha activation. J Lipid Res. 1999;40:2099–2110.
Westergaard M, Henningsen J, Svendsen ML, Johansen C, et al. Modulation of keratinocyte gene expression and differentiation by PPAR-selective ligands and tetradecylthioacetic acid. J Invest Dermatol. 2001;116:702–712.
Berge K, Tronstad KJ, Flindt EN, Rasmussen TH, et al. Tetradecylthioacetic acid inhibits growth of rat glioma cells ex vivo and in vivo via PPAR-dependent and PPAR-independent pathways. Carcinogenesis. 2001;22:1747–1755.
Strobel D, Goertz RS, Bernatik T. Diagnostics in inflammatory bowel disease: ultrasound. World J Gastroenterol. 2011;17:3192–3197.
Dietrich CF. Significance of abdominal ultrasound in inflammatory bowel disease. Dig Dis. 2009;27:482–493.
Nylund K, Hausken T, Gilja OH. Ultrasound and inflammatory bowel disease. Ultrasound Q. 2010;26:3–15.
Delerive P, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors in inflammation control. J Endocrinol. 2001;169:453–459.
Marion-Letellier R, Dechelotte P, Iacucci M, Ghosh S. Dietary modulation of peroxisome proliferator-activated receptor gamma. Gut. 2009;58:586–593.
Jiang C, Ting AT, Seed B. PPAR-gamma agonists inhibit production of monocyte inflammatory cytokines. Nature. 1998;391:82–86.
Singh UP, Singh NP, Singh B, Hofseth LJ, et al. Resveratrol (trans-3,5,4′-trihydroxystilbene) induces silent mating type information regulation-1 and down-regulates nuclear transcription factor-kappaB activation to abrogate dextran sulfate sodium-induced colitis. J Pharmacol Exp Ther. 2010;332:829–839.
Verdin E, Hirschey MD, Finley LW, Haigis MC. Sirtuin regulation of mitochondria: energy production, apoptosis, and signaling. Trends Biochem Sci. 2010;35:669–675.
Harel E, Rubinstein A, Nissan A, Khazanov E, et al. Enhanced transferrin receptor expression by proinflammatory cytokines in enterocytes as a means for local delivery of drugs to inflamed gut mucosa. PLoS One. 2011;6:e24202.
Koch TR, Yuan LX, Stryker SJ, Ratliff P, Telford GL, Opara EC. Total antioxidant capacity of colon in patients with chronic ulcerative colitis. Dig Dis Sci. 2000;45:1814–1819.
Pamplona R. Membrane phospholipids, lipoxidative damage and molecular integrity: a causal role in aging and longevity. Biochim Biophys Acta. 2008;1777:1249–1262.
Aoi Y, Terashima S, Ogura M, Nishio H, Kato S, Takeuchi K. Roles of nitric oxide (NO) and NO synthases in healing of dextran sulfate sodium-induced rat colitis. J Physiol Pharmacol. 2008;59:315–336.
Acknowledgments
The authors would like to thank Eline Milde, Marie Sannes Ramsvik, Kari Williams and Randi Sandvik for excellent technical assistance. Ingeborg Kvivik, Kari Espedal and Marita Hanasand are thanked for their help during the cytokine analyses. This study was supported by Nordforsk (grant no. 070010, MitoHealth), and the Research Council of Norway (grant no. 190287/110) to RKB, and the Board of Nutrition Programmes (University of Bergen and Western Norway Regional Health Authority) to TH. DC holds a fellowship from the Spanish Ministry of Health (FI0800707). Work carried out at the Department of Experimental Medicine was supported by grants from the Spanish Ministry of Science and Innovation (BFU2009-11879/BFI), the Autonomous Government of Catalonia (2009SGR735), and the Spanish Ministry of Health (PI081843) to MPO and RP. The study was also supported by the COST B-35 action.
Conflict of interest
The authors have no conflict of interest, financial or otherwise, with respect to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bjørndal, B., Grimstad, T., Cacabelos, D. et al. Tetradecylthioacetic Acid Attenuates Inflammation and Has Antioxidative Potential During Experimental Colitis in Rats. Dig Dis Sci 58, 97–106 (2013). https://doi.org/10.1007/s10620-012-2321-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2321-2