Abstract
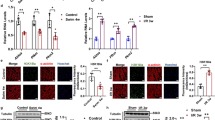
Extracellular vesicles (EVs) serve an important function as mediators of intercellular communication. Exercise is protective for the heart, although the signaling mechanisms that mediate this cardioprotection have not been fully elucidated. Here using nano-flow cytometry, we found a rapid increase in plasma EVs in human subjects undergoing exercise stress testing. We subsequently identified that serum EVs were increased by ~1.85-fold in mice after 3-week swimming. Intramyocardial injection of equivalent quantities of EVs from exercised mice and non-exercised controls provided similar protective effects against acute ischemia/reperfusion (I/R) injury in mice. However, injection of exercise-induced EVs in a quantity equivalent to the increase seen with exercise (1.85 swim group) significantly enhanced the protective effect. Similarly, treatment with exercise-induced increased EVs provided additional anti-apoptotic effect in H2O2-treated H9C2 cardiomyocytes mediated by the activation of ERK1/2 and HSP27 signaling. Finally, by treating H9C2 cells with insulin-like growth factor-1 to mimic exercise stimulus in vitro, we found an increased release of EVs from cardiomyocytes associated with ALIX and RAB35 activation. Collectively, our results show that exercise-induced increase in circulating EVs enhances the protective effects of endogenous EVs against cardiac I/R injury. Exercise-derived EVs might serve as a potent therapy for myocardial injury in the future.







Similar content being viewed by others
Change history
08 October 2019
The original version of this article unfortunately contained a mistake.
References
Aoi W, Ichikawa H, Mune K, Tanimura Y, Mizushima K, Naito Y, Yoshikawa T (2013) Muscle-enriched microRNA miR-486 decreases in circulation in response to exercise in young men. Front Physiol 4:80. doi:10.3389/fphys.2013.00080
Aoi W, Sakuma K (2014) Does regulation of skeletal muscle function involve circulating microRNAs? Front Physiol 5:39. doi:10.3389/fphys.2014.00039
Barile L, Lionetti V, Cervio E, Matteucci M, Gherghiceanu M, Popescu LM, Torre T, Siclari F, Moccetti T, Vassalli G (2014) Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc Res 103:530–541. doi:10.1093/cvr/cvu167
Barile L, Moccetti T, Marban E, Vassalli G (2016) Roles of exosomes in cardioprotection. Eur Heart J. doi:10.1093/eurheartj/ehw304
Bei Y, Fu S, Chen X, Chen M, Zhou Q, Yu P, Yao J, Wang H, Che L, Xu J, Xiao J (2017) Cardiac cell proliferation is not necessary for exercise-induced cardiac growth but required for its protection against ischaemia/reperfusion injury. J Cell Mol Med. doi:10.1111/jcmm.13078
Bei Y, Zhou Q, Sun Q, Xiao J (2015) Exercise as a platform for pharmacotherapy development in cardiac diseases. Curr Pharm Des 21:4409–4416
Bell RM, Botker HE, Carr RD, Davidson SM, Downey JM, Dutka DP, Heusch G, Ibanez B, Macallister R, Stoppe C, Ovize M, Redington A, Walker JM, Yellon DM (2016) 9th Hatter Biannual Meeting: position document on ischaemia/reperfusion injury, conditioning and the ten commandments of cardioprotection. Basic Res Cardiol 111:41. doi:10.1007/s00395-016-0558-1
Cabrera-Fuentes HA, Aragones J, Bernhagen J, Boening A, Boisvert WA, Botker HE, Bulluck H, Cook S, Di Lisa F, Engel FB, Engelmann B, Ferrazzi F, Ferdinandy P, Fong A, Fleming I, Gnaiger E, Hernandez-Resendiz S, Kalkhoran SB, Kim MH, Lecour S, Liehn EA, Marber MS, Mayr M, Miura T, Ong SB, Peter K, Sedding D, Singh MK, Suleiman MS, Schnittler HJ, Schulz R, Shim W, Tello D, Vogel CW, Walker M, Li QO, Yellon DM, Hausenloy DJ, Preissner KT (2016) From basic mechanisms to clinical applications in heart protection, new players in cardiovascular diseases and cardiac theranostics: meeting report from the third international symposium on “New frontiers in cardiovascular research”. Basic Res Cardiol 111:69. doi:10.1007/s00395-016-0586-x
Calvert JW, Condit ME, Aragon JP, Nicholson CK, Moody BF, Hood RL, Sindler AL, Gundewar S, Seals DR, Barouch LA, Lefer DJ (2011) Exercise protects against myocardial ischemia–reperfusion injury via stimulation of beta(3)-adrenergic receptors and increased nitric oxide signaling: role of nitrite and nitrosothiols. Circ Res 108:1448–1458. doi:10.1161/CIRCRESAHA.111.241117
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E, Frisch S, Reed JC (1998) Regulation of cell death protease caspase-9 by phosphorylation. Science 282:1318–1321
Chaturvedi P, Kalani A, Medina I, Familtseva A, Tyagi SC (2015) Cardiosome mediated regulation of MMP9 in diabetic heart: role of mir29b and mir455 in exercise. J Cell Mol Med 19:2153–2161. doi:10.1111/jcmm.12589
Chistiakov DA, Orekhov AN, Bobryshev YV (2016) Cardiac extracellular vesicles in normal and infarcted heart. Int J Mol Sci. doi:10.3390/ijms17010063
Colombo M, Raposo G, Thery C (2014) Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol 30:255–289. doi:10.1146/annurev-cellbio-101512-122326
Danielson KM, Estanislau J, Tigges J, Toxavidis V, Camacho V, Felton EJ, Khoory J, Kreimer S, Ivanov AR, Mantel PY, Jones J, Akuthota P, Das S, Ghiran I (2016) Diurnal variations of circulating extracellular vesicles measured by nano flow cytometry. PLoS One 11:e0144678. doi:10.1371/journal.pone.0144678
Earnest CP, Lupo M, Thibodaux J, Hollier C, Butitta B, Lejeune E, Johannsen NM, Gibala MJ, Church TS (2013) Interval training in men at risk for insulin resistance. Int J Sports Med 34:355–363. doi:10.1055/s-0032-1311594
Emanueli C, Shearn AI, Angelini GD, Sahoo S (2015) Exosomes and exosomal miRNAs in cardiovascular protection and repair. Vasc Pharmacol 71:24–30. doi:10.1016/j.vph.2015.02.008
Forterre A, Jalabert A, Berger E, Baudet M, Chikh K, Errazuriz E, De Larichaudy J, Chanon S, Weiss-Gayet M, Hesse AM, Record M, Geloen A, Lefai E, Vidal H, Coute Y, Rome S (2014) Proteomic analysis of C2C12 myoblast and myotube exosome-like vesicles: a new paradigm for myoblast-myotube cross talk? PLoS One 9:e84153. doi:10.1371/journal.pone.0084153
Fruhbeis C, Helmig S, Tug S, Simon P, Kramer-Albers EM (2015) Physical exercise induces rapid release of small extracellular vesicles into the circulation. J Extracell Vesicles 4:28239. doi:10.3402/jev.v4.28239
Gomes EC, Silva AN, de Oliveira MR (2012) Oxidants, antioxidants, and the beneficial roles of exercise-induced production of reactive species. Oxid Med Cell Longev 2012:756132. doi:10.1155/2012/756132
Gorgens SW, Eckardt K, Jensen J, Drevon CA, Eckel J (2015) Exercise and regulation of adipokine and myokine production. Prog Mol Biol Transl Sci 135:313–336. doi:10.1016/bs.pmbts.2015.07.002
Guescini M, Canonico B, Lucertini F, Maggio S, Annibalini G, Barbieri E, Luchetti F, Papa S, Stocchi V (2015) Muscle releases alpha-sarcoglycan positive extracellular vesicles carrying miRNAs in the bloodstream. PLoS One 10:e0125094. doi:10.1371/journal.pone.0125094
Hausenloy DJ, Barrabes JA, Botker HE, Davidson SM, Di Lisa F, Downey J, Engstrom T, Ferdinandy P, Carbrera-Fuentes HA, Heusch G, Ibanez B, Iliodromitis EK, Inserte J, Jennings R, Kalia N, Kharbanda R, Lecour S, Marber M, Miura T, Ovize M, Perez-Pinzon MA, Piper HM, Przyklenk K, Schmidt MR, Redington A, Ruiz-Meana M, Vilahur G, Vinten-Johansen J, Yellon DM, Garcia-Dorado D (2016) Ischaemic conditioning and targeting reperfusion injury: a 30 year voyage of discovery. Basic Res Cardiol 111:70. doi:10.1007/s00395-016-0588-8
Hausenloy DJ, Yellon DM (2004) New directions for protecting the heart against ischaemia-reperfusion injury: targeting the Reperfusion Injury Salvage Kinase (RISK)-pathway. Cardiovasc Res 61:448–460. doi:10.1016/j.cardiores.2003.09.024
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116:674–699. doi:10.1161/CIRCRESAHA.116.305348
JanssenDuijghuijsen LM, Keijer J, Mensink M, Lenaerts K, Ridder L, Nierkens S, Kartaram SW, Verschuren MC, Pieters RH, Bas R, Witkamp RF, Wichers HJ, van Norren K (2017) Adaptation of exercise-induced stress in well-trained healthy young men. Exp Physiol 102:86–99. doi:10.1113/EP086025
Jeong JJ, Ha YM, Jin YC, Lee EJ, Kim JS, Kim HJ, Seo HG, Lee JH, Kang SS, Kim YS, Chang KC (2009) Rutin from Lonicera japonica inhibits myocardial ischemia/reperfusion-induced apoptosis in vivo and protects H9c2 cells against hydrogen peroxide-mediated injury via ERK1/2 and PI3K/Akt signals in vitro. Food Chem Toxicol 47:1569–1576. doi:10.1016/j.fct.2009.03.044
Jiang X, Guo CX, Zeng XJ, Li HH, Chen BX, Du FH (2015) A soluble receptor for advanced glycation end-products inhibits myocardial apoptosis induced by ischemia/reperfusion via the JAK2/STAT3 pathway. Apoptosis 20:1033–1047. doi:10.1007/s10495-015-1130-4
Khan M, Nickoloff E, Abramova T, Johnson J, Verma SK, Krishnamurthy P, Mackie AR, Vaughan E, Garikipati VN, Benedict C, Ramirez V, Lambers E, Ito A, Gao E, Misener S, Luongo T, Elrod J, Qin G, Houser SR, Koch WJ, Kishore R (2015) Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res 117:52–64. doi:10.1161/CIRCRESAHA.117.305990
Kleindienst A, Battault S, Belaidi E, Tanguy S, Rosselin M, Boulghobra D, Meyer G, Gayrard S, Walther G, Geny B, Durand G, Cazorla O, Reboul C (2016) Exercise does not activate the beta3 adrenergic receptor-eNOS pathway, but reduces inducible NOS expression to protect the heart of obese diabetic mice. Basic Res Cardiol 111:40. doi:10.1007/s00395-016-0559-0
Kowal J, Tkach M, Thery C (2014) Biogenesis and secretion of exosomes. Curr Opin Cell Biol 29:116–125. doi:10.1016/j.ceb.2014.05.004
Li J, Zhang H, Zhang C (2012) Role of inflammation in the regulation of coronary blood flow in ischemia and reperfusion: mechanisms and therapeutic implications. J Mol Cell Cardiol 52:865–872. doi:10.1016/j.yjmcc.2011.08.027
Little JP, Safdar A, Benton CR, Wright DC (2011) Skeletal muscle and beyond: the role of exercise as a mediator of systemic mitochondrial biogenesis. Appl Physiol Nutr Metab 36:598–607. doi:10.1139/h11-076
Little JP, Safdar A, Bishop D, Tarnopolsky MA, Gibala MJ (2011) An acute bout of high-intensity interval training increases the nuclear abundance of PGC-1alpha and activates mitochondrial biogenesis in human skeletal muscle. Am J Physiol Regul Integr Comp Physiol 300:R1303–R1310. doi:10.1152/ajpregu.00538.2010
Liu X, Xiao J, Zhu H, Wei X, Platt C, Damilano F, Xiao C, Bezzerides V, Bostrom P, Che L, Zhang C, Spiegelman BM, Rosenzweig A (2015) miR-222 is necessary for exercise-induced cardiac growth and protects against pathological cardiac remodeling. Cell Metab 21:584–595. doi:10.1016/j.cmet.2015.02.014
Lombardi G, Sanchis-Gomar F, Perego S, Sansoni V, Banfi G (2016) Implications of exercise-induced adipo-myokines in bone metabolism. Endocrine 54:284–305. doi:10.1007/s12020-015-0834-0
Malik ZA, Kott KS, Poe AJ, Kuo T, Chen L, Ferrara KW, Knowlton AA (2013) Cardiac myocyte exosomes: stability, HSP60, and proteomics. Am J Physiol Heart Circ Physiol 304:H954–H965. doi:10.1152/ajpheart.00835.2012
Ostrowski M, Carmo NB, Krumeich S, Fanget I, Raposo G, Savina A, Moita CF, Schauer K, Hume AN, Freitas RP, Goud B, Benaroch P, Hacohen N, Fukuda M, Desnos C, Seabra MC, Darchen F, Amigorena S, Moita LF, Thery C (2010) Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat Cell Biol 12:19–30. doi:10.1038/ncb2000 (sup pp 11–13)
Otani H (2009) The role of nitric oxide in myocardial repair and remodeling. Antioxid Redox Signal 11:1913–1928. doi:10.1089/ARS.2009.2453
Ela S, Mager I, Breakefield XO, Wood MJ (2013) Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov 12:347–357. doi:10.1038/nrd3978
Safdar A, Saleem A, Tarnopolsky MA (2016) The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat Rev Endocrinol 12:504–517. doi:10.1038/nrendo.2016.76
Seldin MM, Peterson JM, Byerly MS, Wei Z, Wong GW (2012) Myonectin (CTRP15), a novel myokine that links skeletal muscle to systemic lipid homeostasis. J Biol Chem 287:11968–11980. doi:10.1074/jbc.M111.336834
Shi J, Bei Y, Kong X, Liu X, Lei Z, Xu T, Wang H, Xuan Q, Chen P, Xu J, Che L, Liu H, Zhong J, Sluijter JP, Li X, Rosenzweig A, Xiao J (2017) miR-17-3p contributes to exercise-induced cardiac growth and protects against myocardial ischemia–reperfusion injury. Theranostics 7:664–676. doi:10.7150/thno.15162
Tao L, Bei Y, Lin S, Zhang H, Zhou Y, Jiang J, Chen P, Shen S, Xiao J, Li X (2015) Exercise training protects against acute myocardial infarction via improving myocardial energy metabolism and mitochondrial biogenesis. Cell Physiol Biochem 37:162–175. doi:10.1159/000430342
Teng X, Chen L, Chen W, Yang J, Yang Z, Shen Z (2015) mesenchymal stem cell-derived exosomes improve the microenvironment of infarcted myocardium contributing to angiogenesis and anti-inflammation. Cell Physiol Biochem 37:2415–2424. doi:10.1159/000438594
Terada K, Kaziro Y, Satoh T (2000) Analysis of Ras-dependent signals that prevent caspase-3 activation and apoptosis induced by cytokine deprivation in hematopoietic cells. Biochem Biophys Res Commun 267:449–455. doi:10.1006/bbrc.1999.1955
Vicencio JM, Yellon DM, Sivaraman V, Das D, Boi-Doku C, Arjun S, Zheng Y, Riquelme JA, Kearney J, Sharma V, Multhoff G, Hall AR, Davidson SM (2015) Plasma exosomes protect the myocardium from ischemia–reperfusion injury. J Am Coll Cardiol 65:1525–1536. doi:10.1016/j.jacc.2015.02.026
Wang Y, Zhang L, Li Y, Chen L, Wang X, Guo W, Zhang X, Qin G, He SH, Zimmerman A, Liu Y, Kim IM, Weintraub NL, Tang Y (2015) Exosomes/microvesicles from induced pluripotent stem cells deliver cardioprotective miRNAs and prevent cardiomyocyte apoptosis in the ischemic myocardium. Int J Cardiol 192:61–69. doi:10.1016/j.ijcard.2015.05.020
Weston CR, Balmanno K, Chalmers C, Hadfield K, Molton SA, Ley R, Wagner EF, Cook SJ (2003) Activation of ERK1/2 by deltaRaf-1:ER* represses Bim expression independently of the JNK or PI3K pathways. Oncogene 22:1281–1293. doi:10.1038/sj.onc.1206261
Xu J, Tang Y, Bei Y, Ding S, Che L, Yao J, Wang H, Lv D, Xiao J (2016) miR-19b attenuates H2O2-induced apoptosis in rat H9C2 cardiomyocytes via targeting PTEN. Oncotarget 7:10870–10878. doi:10.18632/oncotarget.7678
Zhang KR, Liu HT, Zhang HF, Zhang QJ, Li QX, Yu QJ, Guo WY, Wang HC, Gao F (2007) Long-term aerobic exercise protects the heart against ischemia/reperfusion injury via PI3 kinase-dependent and Akt-mediated mechanism. Apoptosis 12:1579–1588. doi:10.1007/s10495-007-0090-8
Acknowledgements
This work was supported by the grants from National Natural Science Foundation of China (81570362 and 91639101 to J.J. Xiao and 81400647 to Y. Bei), the development fund for Shanghai talents (to J.J. Xiao), and the National Institutes of Health (NCATS Grant UH3 TR000901 to S. Das and U01 HL126497 to I.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
395_2017_628_MOESM1_ESM.pdf
Supplementary Figure 1 The mRNA levels of STAM1, TSG101, RAB11, and RAB27A were not changed in H9C2 cells treated with IGF-1 (n=6) (PDF 22 kb)
Rights and permissions
About this article
Cite this article
Bei, Y., Xu, T., Lv, D. et al. Exercise-induced circulating extracellular vesicles protect against cardiac ischemia–reperfusion injury. Basic Res Cardiol 112, 38 (2017). https://doi.org/10.1007/s00395-017-0628-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-017-0628-z